Chemistry 2A Sample Midterm Exam
advertisement

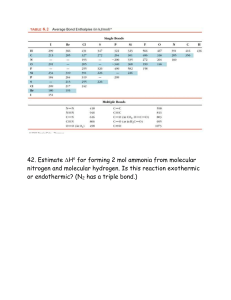
Sample Midterm #3 These are problems that I (Rolf Unterleitner) made up for all the 2A sections. This covers only the material since the last midterm. The answers are on the last pages. I’ll go over the solutions Sunday (3/15) evening starting at 5:30 PM in 26 Wellman it is likely to last about two hours. Please do not ask the Prof. or the TA's for the answers they do not have them thank you. 1) Which is not a reasonable resonance structures for the same molecule? a) O S O b) O S c) O O S O d) O 2 S O 2) Draw the lewis structure for each of the following molecules or ions (in case of resonance, one form is enough). Deduce the formal charge, if any, on each atom. Which of the following assignments is wrong? a) SCN- 0 on central C b) NO2- 0 on N c) NO3- 0 on central N d) CO2 0 on C e) N2O +1 on Central N 3) The lewis formula for BrF2+ has how many lone pairs of electrons? a) 2 b) 3 c) 7 d) 8 e) 9 4) In the best set of resonance forms for sulfate there are ___ sigma bonds and _____ pi bonds respectively around the central S. a) 1, 3 b) 2, 1 c) 4, 2 d) 2, 2 e) 3, 1 5) The minimum total number of sigma bonds in the C4H6 is a) 7 b) 13 6) c) 9 d) 15 e) 11 Which of the following statements is false for drawing Lewis structures? a) Octet can never be expanded on C, N, O and F. b) Formal charge is found by subtracting the number of electrons right around a given atom from the number of valence electrons the atom should have to be neutral. c) When counting octet some of the electrons counted are shared by two atoms. d) If every atom has an octet then there is no need to check the formal charges on the atoms. e) The sum of the formal charges must add up to the net charge on the molecule or the number of electrons on the molecule is wrong. 1 7) In which of the following must the octet rule be violated on second period elements? 1. BrF4- 2. SO2-2 3. BH3 a) 2 and 3 b) All of them c) 1 and 3 d) None of them e) 3 only 8) Which of the following lewis formulas is incorrect? a) C O For CO - b) N H C For NO O O c) O O For HCO2 - d) O S O For SO3 -2 O e) O 9) S 2 For SO3 The lewis formula for CH3COOF has how many lone pairs of electrons? a) 2 10) O b) 7 c) 4 d) 9 What is the bond order for the C-O bonds in carbonate? a) 2 b) 1 c) 1 1/3 d) 1 2/3 e) 1 1/2 11) All of the following have a dipole moment except a) O3 b) SH5F c) CF2H2 d) ClF2e) BrCl3 2 e) 5 12) Which of the following species is see-saw? a) SiF4 b) SCl3O2c) BrF4d) XeF2O2 e) NH3 13) Which of the following species has only a bond angle of 180°? a) H2O b) PCl2+ c) SeF6 d) SF4 e) BrH214) Which of the following species has a molecular geometry of tetrahedral? a) S2O2-2 15) b) SeH3- d) BrI4- c) PH3 e) SO2F2 Which of the following species has bond angles of exactly 109.5°? a) SO4-2 b) PI4- c) H2CO d) PO3-3 e) SO2H 16) Which of the molecules would have the shortest bond length? a) N2 b) N2- c) N2+ d) N2-2 e) N2+2 17) Which of the following combinations of wave functions would give a Ψ*σ2p if the sigma bonds are found to lie on the y axis. a) Ψ2py.Ψ*σ2py b) Ψ*2pz + Ψ*2pz d) Ψ2px - Ψ2px e) Ψ2py + Ψ2py c) Ψ*2py - Ψ*2py 3 18) Which of the following is the CORRECT reaction for the second ionization of Gold (Au). a. Au(s) Au+2(g)+ 2eb. Au(g) Au+2(g) + 2ec. Au+(g) Au+2(g) + ed. Au+(s) Au+2(s) + ee. Au+3(g) Au+2(g) + e- 19) Which is the CORRECT reaction for the second electron affinity of oxygen, and what would the sign on EA be? a. O(s) + 2e- O-2(g) (positive) b. O2(g) + 2e- O2-2(g) (negative) c. O- (g) + e- O-2(g) (positive) d. O- (g) + e- O-2(g) (negative) e. O- + e- (g) O-2 (positive) 20) Which of the following species would be paramagnetic? i) B2 ii) BC iii) NO+ iv) CF- a) ii only b) all are c) i , ii, iv, v d) ii, v e) i , v 4 v) O2 Fill in the blanks 1) Answer the following questions by about the molecule below by filling in the blanks after first completing the lewis structure. H H N C1 O C3 C2 H i) The O-C3 σ bond involves overlap of a _____ orbital on the O and a _____ orbital on the C3. ii) The C2-C3 σ bond involves overlap of a _____ orbital on the C2 and a _____ orbital on the C3. iii) The C1-C2 σ bond involves overlap of a _____ orbital on the C1 and a _____ orbital on the C2. iv) The N-H bond involves overlap of a _____ orbital on the H and a _____ orbital on the N. v) The N-C1-C2 bond angles is approximately _____ °. vi) The H-N-H bond angles is approximately _____ °. vi) The O-C-H bond angles is approximately _____ °. 5 2) Fill in the following table (see the example at the top). For the determination of formal charge(s) consider the Lewis formula with the smallest formal charges (lowest energy) molecule Electron Group Geometry (central atom) ClF3 TrigonalBipyramidal Molecular Geometry Ideal Bond Angles T-Shaped 90°, 180° Polar or Nonpolar Molecule polar BrCl5 SF3+ CH3- NO3- 6 Hybridization on central atom sp3d Formal Charge(s) On underlined element 0 Oxidation Number of the underlined element -1 3) For carbon dioxide CO2. a) Draw the lewis structure. b) Using VBT give the orbital involved in the sigma bonding on each atom. Also give the ground state electron configuration for valence electrons on each atom as it is found in the molecule. C O Orbital/ Hybrid Ground state e- conf. (with hybridization) c) Draw the sigma bond frame work (orbitals overlapping). d) Dram the π bond framework (orbitals how they would overlap). 4) For NH2CH2COOCP a. Draw the correct lewis structure and use valence bond theory to describe all the bonds that do not involve hydrogen. b. How many sigma bonds (total) are there?______________ c. How many π bonds are there?________________ d. How many lone pairs are there?__________________ e. How many valence electrons are there? 7 5) a) Sketch the Molecular Orbital energy diagrams for molecules formed by second period elements including the atomic orbitals. b) Give the ground state electron configuration for the molecules CN, ON- and OF+ and calculate the bond order and the number of unpaired electrons and the magnetism (para/ dia). Which is more stable c) Draw the lewis structure for each CN, ON- and OF+. Use the lewis structure to give bond order and predict the magnetism for each. 8 6) Arrange each of the following in the correct order. a. increasing ionization energy. K, Ca, Ga (same as strongest reducing agent) < < < b. increasing electron affinity. K, Ca, Ga (same as strongest oxidizing agent) < < < c. increasing atomic radii. K, Ca, Ga < < < d. increasing radii K+, Ca+2, Se-2, Br-, Kr < < < < 7) Draw the lewis dot structure for sodium carbonate Na2CO3 8) Draw the set of the best resonance structures for BrO4-. 9 9) Draw the lewis structure for H3PO4. 10) What would the best arrangement be for a compound with 2 N's and one O. (Show all your reasoning) 11) What would the best arrangement be for a compound with one H and one N and one C. (Show all your reasoning) 10 Answers: 1) d 2) c 3) d 4) c 5) c 6) d 7) e 8) b 9) b 10) c 11) d 12) d 13) e 14) e 15) a 16) a 17) e 18) c 19) c 20) c H H N C i) 2p, sp2 ii) sp, sp2 iii) sp, sp iv) 1s, sp3 v) 180° vi) <109.5 ° vii) >120° 1) O C C H 2) molecule ClF3 Electron Group Geometry (central atom) Molecular Geometry Ideal Bond Angles T-Shaped 90°, 180° Square pyramidal Trigonal Pyramidal 90°, 180° Polar sp3d2 0 +5 <109.5° Polar sp3 +1 +4 Trigonal bipyramidal See-saw 90°, 120°, 180° polar sp3d 0 +4 Trigonal planar Trigonal planar 120° Non-polar sp2 +1 +5 TrigonalBipyramidal octahedral BrCl5 Tetrahedral + SF3 SF4 NO 3 Polar or Nonpolar Molecule polar 11 Hybridization on central atom Formal Charge(s) On underlined element sp3d 0 Oxidation Number of the underlined element -1 6) a) K<Ga<Ca b) Ca<K<Ga c) Ga<Ca<K d) Ca+2<K+<Kr<Br-<S-2 12 7) O C O Na O Na 8) Best set puts the neg charge on the most electronegative atom (in this case O). O O O O O Br O O O 9) H Br O O O O O H P O Br O O Br O O O H oxy acids O's around the central atom and H's off the O's O 10) N N O N N O N best lowest formal charges 11) H C N best lowest formal charges H N C 13 2 O N