Chemical Bonding Homework: Lewis Structures & More
advertisement

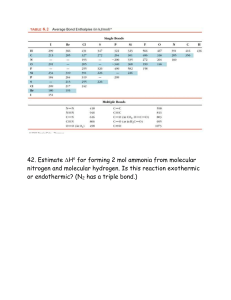
HOMEWORK:CHAPTER 9 CHEMICAL BONDING I 9.16 Give the empirical formulas and names of the compounds formed from the following pairs of ions: (a) Rb+ and I- (b) Cs+ and SO42- , (c) Sr2+ and N3- (d) Al3+ and S29.18 Write the Lewis dot symbols of the reactants and products in the following reactions. (First balance the equations.) (a) Sr + Se → SrSe (b) Ca + H2 → CaH2 (c) Li + N2 → Li3N (d) Al + S → Al2S3 9.20 For each of the following pairs of elements, state whether the binary compound they form is likely to be ionic or covalent. Write the empirical formula and name of the compound: (a) B and F, (b) K and Br. 9.26 Calculate the lattice energy of calcium chloride given that the heat of sublimation of Ca is 121 kJ/mol and ∆Hf° (CaCl2) = -795 kJ/mol. (See Tables 8.2 and 8.3 for other data.) 9.36 Arrange the following bonds in order of increasing ionic character: carbon to hydrogen, fluorine to hydrogen, bromine to hydrogen, sodium to chlorine, potassium to fluorine, lithium to chlorine. 9.38 List the following bonds in order of increasing ionic character: cesium to fluorine, chlorine to chlorine, bromine to chlorine, silicon to carbon. 9.40 Classify the following bonds as ionic, polar covalent, or covalent, and give your reasons: (a) the SiSi bond in Cl3SiSiCl3 , (b) the SiCl bond in Cl3SiSiCl3 , (c) the CaF bond in CaF2 , (d) the NH bond in NH3 . 9.44 Write Lewis structures for the following ions: (a) O22- (b) C22- , (c) NO+ , (d) NH+4. Show formal charges. 9.45 The skeletal structure of acetic acid shown below is correct, but some of the bonds are wrong. (a) Identify the incorrect bonds and explain what is wrong with them. (b) Write the correct Lewis structure for acetic acid. 9.50 Draw three resonance structures for the chlorate ion, ClO-3. Show formal charges. 9.52 Draw tow resonance structures for diazomethane. CH2N2 . Show formal charges. The skeletal structure of the molecule is H C N N H 9.54 Draw three resonance structures for the molecule N2O in which the atoms are arranged in the order NNO. Indicate formal charges. 9.60 In the vapor phase, beryllium chloride consists of discrete BeCl2 molecules. Is the octet rule satisfied for Be in this compound? If not, can you form an octet around Be by drawing another resonance structure? How plausible is this structure? 9.62 Write a Lewis structure for SbCl5. Does this molecule obey the octet rule? 9.64 Write Lewis structures for the reaction AlCl3 + Cl- → AlCl-4 What kind of bond joins Al and Cl in the product? 9.68 For the reaction ∆H° = -107.2 kJ O(g) + O2(g) → O3(g) Calculate the average bond energy in O3 . 9.70 For the reaction 2C2H6(g) + 7O2(g) → 4CO2(g) + 6H2O(g) (a) Predict the enthalpy of reaction from the average bond energies in Table 9.4. (b) Calculate the enthalpy of reaction from the standard enthalpies of formation (see Appendix 3) of the reactant and product molecules, and compare the result with your answer for part (a). 9.72 Which of the following are ionic compounds? Which are covalent compounds? RbCl, PF5, BrF3, KO2, Cl4 9.74 The formulas for the fluorides of the third-period elements are NaF, MgF2, AlF3, SiF4, PF5, SF6, and CLF3. Classify these compounds as covalent or ionic. 9.76 Describe some characteristics of an ionic compound such as KF that would distinguish it from a covalent compound such as benzene (C6H6). 9.78 Write three reasonable resonance structures for the azide ion N-3 in which the atoms are arranged as NNN. Show formal charges. 9.79 Give an example of an ion or molecule containing Al that (a) obeys the octet rule, (b) has an expanded octet, and (c) has an incomplete octet. 9.82 Attempts to prepare the compounds listed below as stable species under atmospheric conditions have failed. Suggest possible reasons for the failure. CF2, LiO2, CsCl2, PI5 9.84 Are the following statements true or false? (a) Formal charges represent actual separation of charges. (b) ∆H°rxn can be estimated from the bond energies of reactants and products. (c) All second-period elements obey the octet rule in their compounds. (d) The resonance structures of a molecule can be separated from one another. 9.86 Using the following information and the fact that the average C−H bond energy is 414 kJ/mol, estimate the standard enthalpy of formation of methane (CH4). C(s) → C(g) ∆H°rxn = 716 kJ ∆H°rxn = 872.8 kJ 2H2(g) → 4H(g) 2 9.88 Which of the following molecules has the shortest nitrogen-to-nitrogen bond? Explain. N2H4, N2O, N2, N2O4 9.92 The amide ion, NH-2 , is a Bronsted base, Represent the reaction between the amide ion and water. 9.94 The triiodide ion (I-3) in which the I atoms are arranged in a straight line is stable, but the corresponding F-3 ion does not exist. Explain. 9.96 Methyl isocyanate (CH3NCO) is used to make certain pesticides. In December 1984, water leaked into a tank containing this substance at a chemical plant, producing a toxic cloud that killed thousands of people in Bhopal, India. Draw Lewis structures for CH3NCO, showing formal charges. 9.98 Several resonance structures for the molecule CO2 are shown below. Explain why some of them are likely to be of little importance in describing the bonding in this molecule. (a) O C O (c) O C O (b) O C O (d) O C O 2+ 9.100 Draw Lewis structures for the following chlorofluorocarbons (CFCs), which are partly responsible for the depletion of ozone in the stratosphere: (a) CFCl3, (b) CF2Cl2, (c) CHF2Cl, (d) CF3CHF29.102 Calculate ∆H° for the reaction H2(g) + I2(g) → 2HI(g) 9.104 Write Lewis structures for the following four isoelectronic species: (a) CO, (b) NO+ , (c) CN- , (d) N2, Show formal charges. 9.106 Comment on the correctness of the statement, “All compounds containing a noble gas atom violate the octet rule.” 9.108 (a) From the following data calculate the bond energy of the F-2 ion. ∆H°rxn = 156.9 kJ F2(g) → 2F(g) ∆H°rxn = 333 kJ F-(g) → 2F(g) + e∆H°rxn = 290 kJ F2-(g) → 2F2(g) + e(b) Explain the difference between the bond energies of F2 and F-2 . 9.110 What are the other two reasons for choosing (b) in Example 9.7? 9.112 The N − O bond distance in nitric oxide is 115 pm, which is intermediate between a triple bond (106 pm) and a double bond (120 pm). (a) Draw two resonance structures for NO and comment 3 9.114 9.116 9.118 9.120 9.122 on their relative importance. (b) Is it possible to draw a resonance structure having a triple bond between the atoms? Another possible skeletal structure for the CO2-3 (carbonate) ion besides the one presented in Example 9.5 is O C O O, Why would we not use this structure to represent CO2-3? In the gas phase, aluminum chloride exists as a dimer (a unit of two) with the formula Al2Cl6. Its skeletal structure is given by Cl Cl Cl Al Al Cl Cl Cl Complete the Lewis structure and indicate the coordinate covalent bonds in the molecule. Experiments show that it takes 1656 kJ/mol to break all the bonds in methane (CH4) and 4006 kJ/mol to break all the bonds in propane (C3H8). Based on these data, calculate the average bond energy of the C−C bond Vinyl chloride (C2H2Cl) differs from ethylene (C2H4) in that one of the H atoms is replaced with a Cl atom. Vinyl chloride is used to prepare poly(vinyl chloride), which is an important polymer used in pipes. (a) Draw the Lewis structure of vinyl chloride. (b) The repeating unit in poly(vinyl chloride) is−CH2−CHCl− . Draw a portion of the molecule showing three such repeating units. (c) Calculate the enthalpy change when 1.0 x 103 kg of vinyl chloride forms poly(vinyl chloride). The American chemist Robert S.Mulliken suggested a different definition for the electronegativity (EN) of an element, given by EN = IE + EA 2 where IE is the first ionization energy and EA the electron affinity of the element. Calculate the electronegativities of O, F, and Cl using the above equation. Compare the electronegativities of these elements on the Muliken and Pauling scale. (To convert to the Pauling scale, divide each EN value by 230 KJ/mol.) 4