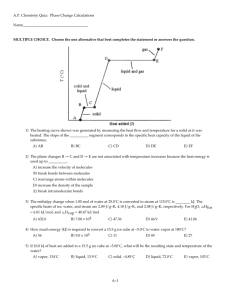
Chapter 12
advertisement

Chapter 12
State of Matter and Intermolecular Forces
Gases, in contrast to liquids and solids, are independent on their chemical
composition, especially at high temperatures. This is because the intermolecular
attractions between gas molecules could be neglected, as they are very small due to
large distances between gas molecules. However, molecules in the liquid and solid
states are close together and intermolecular forces are thus very effective in giving the
liquid or solid substances their unique properties.
Intermolecular forces are generally much weaker than covalent bonds where they are
electrostatic in nature. For instance:
Only 16 kJ/mol of energy is required to overcome the intermolecular attraction
between HCl molecules in the liquid state and change it into vapor. However, 431
kJ/mol of energy is required to break the covalent bond between the H and the Cl
atoms in the HCl molecule. When the state of a molecular substance is changed from
gas to liquid or solid, or vice versa, the bonds within the molecule are unchanged (e.g.
vaporizing HCl does not break the hydrogen-chlorine chemical bond involving a
shared pair of valence electrons)
The temperature required to undergo a phase transition is a consequence of the
strength of the intermolecular attractive forces. The temperature at which a liquid
boils reflects the kinetic energy needed to overcome the attractive intermolecular
forces between liquid molecules. The same is true for melting a solid. In fact, the
strength of the intermolecular forces determines the physical properties of the
substance such as the melting point, vapor pressure, and boiling point.
Attractive forces, which exist between neutral molecules, are electrostatic forces
including:
Dipole-dipole forces
Hydrogen bonding forces
London dispersion forces
Dipole-dipole Interactions
Polar molecules have some atoms of greater electronic clouds than others, according
to their electronegativities. The part of the molecule with a partial negative charge
tends to orient close to the part of the partial positive region of another molecule so as
to decrease potential energy. Since molecules are in continuous motion, this
orientation is far from being a perfect structure and repulsion forces do exist.
However, the overall forces that overwhelm a system are attractive forces. Dipoledipole interactions are about 1% of the usual strength of an ionic bond and are much
stronger in liquids and solids than in gases. Ion-dipole interactions are also important
when solutions contain ions where an ion will attract oppositely charged regions of a
dipole.
Dipole-dipole interactions are most important in polar molecular assemblies.
Boiling points increase for polar molecules of similar mass, but increasing dipole:
Substance
Propane
Dimethyl ether
Methyl chloride
Acetaldehyde
Acetonitrile
Molecular Mass
(amu)
44
46
50
44
41
Dipole moment, u
Boiling Point (°K)
(D)
0.1
231
1.3
248
2.0
249
2.7
294
3.9
355
Hydrogen Bonding
This type of bonding is a special case of dipole-dipole interactions present in
molecules with hydrogen atoms covalently bonded to a small atom of high
electronegativity like N, O, and F. It is the very strong hydrogen bonding between
water molecules that accounts for the high boiling point of water.
Hydrogen Bonding in Water Molecules
London Forces
Fritz London (1930) suggested that the motion of electrons within an atom or nonpolar molecule could result in a transient dipole moment. Non-polar molecules
experience weak attractions called London forces and are primarily due to continuous
change in the distribution of electronic clouds. This results in induced dipoles, which
holds the non-polar molecules together. Even atoms and gases like He and N2
experience such attractive forces due to induced dipoles, which hold these molecules
and atoms together in the liquid state at very low temperatures.
The distribution of electrons around an individual atom, at a given instant in time,
may not be perfectly symmetrical where both electrons may be on one side of the
nucleus and thus the atom would have an apparent dipole moment called a transient
dipole. This will cause the neighboring atoms to be attracted to one another by the so
called the London dispersion force, or just dispersion force, which are significant only
when the atoms are close together.
The ease of distortion of the electron cloud by an induced dipole is referred to as the
polarizability of that molecule where:
The greater the polarizability of a molecule the easier it is to induce a
momentary dipole and the stronger the dispersion forces
Larger atoms tend to have greater polarizability
Large molecules also tend to have greater polarizability, due to the large
number of electrons present. Dispersion forces tend to increase with increasing
molecular mass
Dispersion forces are present between all molecules including polar/non-polar
and polar/polar molecules as well as non-polar/non-polar molecules.
Comparing General Properties of Gases, Liquids, and Solids
Gases are very compressible, have very high diffusion rates, and assume the volume
and shape of the vessel they are placed in. However, liquids and solids can be
compared as in the table below:
Property
1 Compressibility:
Change in volume with
pressure
2 Diffusion:
Relative
movement of molecules
upon collisions
3 Volume and Shape
Liquids
Practically
incompressible
Solids
Incompressible
Slow diffusion
Very slow diffusion
Liquids have same
shape as the container
but their volume is
confined
4 Surface
Tension: Increases
as
the
Energy needed to create intermolecular forces
a unit area of surface
increases
5 Evaporation
Evaporation increases
with surface area and
temperature as well as
when
intermolecular
forces decrease
Solids maintain their
shape and volume when
placed in a container
Some solids sublime
(change from solid to
gas without going to the
liquid state)
Liquids tend to lower their surface area in an attempt to decrease its energy. This is
the reason why water droplets form spherical drops on a wax-coated surface. If water,
on the other hand, is poured on a clean polar surface, water tends to spread since in
this case the attractive forces between polar water molecules and the polar surface is
greater than the surface tension of water.
Heats of Vaporization
The molar heat of vaporization is the amount of energy needed to vaporize one mole
of a liquid at constant pressure. This is also called the enthalpy of vaporization and is
equal to:
Hvaporization = Hvapor - Hliquid
For a molecule in a liquid to vaporize, it should absorb energy from surrounding
molecules and thus the energy of the liquid will fall down and the energy of the
vaporized molecules will increase by the same amount. Recall what happens when
you go out of water after a shower, definitely you will feel cooler as water molecules
continue to evaporate from your body.
The energy needed to cause evaporation will be dependent on the intermolecular
forces between molecules in a liquid. It is obvious that the heat of vaporization will
increase as the intermolecular forces become stronger.
Some Enthalpies of Vaporization at 298 K
Liquid
Hvaporization, kJ/mol
Boiling Point (oC)
C4H10
22.3
0
C6H14
28.6
68
H2S
18.8
-61
Br2
30.7
59
Methyl alcohol, CH3OH
38.0
72
Ethyl alcohol, CH3CH2OH
42.6
78
Water, H2O
44.0
100
HF
30.2
17
NH3
23.6
-33
Sometimes large Hvaporization, are obtained when hydrogen bonding is present, for
example, in compounds with N, O, or F.
Example
The molar heat of vaporization of water is 40.6 kJ/mol. How many kJ of energy are
needed to convert 1.00 L of water (density = 1.00 g/mL) to steam.
Solution
One liter of water = 1000 g water
1 mol of water requires 40.6 kJ to be converted to steam, therefore, find the number of
moles of water present in 1000 g and multiply it by the heat of vaporization.
Energy needed = (Hvaporization kJ/mol) * Number of moles
Energy needed = (40.6 kJ/mol) * (1000 g/18.0 g/mol) = 2260 kJ
Vapor Pressures of Liquids and Solids
In many occasions, one would observe that a liquid in an open container will
evaporate with time and ultimately the container will be empty after some time.
Alcohols evaporate faster than water. However, if a liquid is placed in a closed
container, the evaporated molecules will not escape and a vapor pressure would
accumulate above the liquid. The vapor pressure will continue to increase as more
molecules evaporate till a point is reached where the rate of evaporation exactly
equals the rate of condensation (equilibrium state). This is called the equilibrium
vapor pressure of a liquid and is definitely dependent on temperature.
Patm = PHg + Pvapor
Factors Affecting Vapor Pressure
1. The nature of the attractive forces
As the attractive forces become stronger, a lower vapor pressure is observed as it is
not easy for molecules to overcome these forces of attraction.
2. Temperature
As the temperature of a liquid is increased, its equilibrium vapor pressure is also
increased (in fact so rapidly). The highest temperature at which a distinct liquid
phase exists is called the critical temperature and the vapor pressure at this critical
temperature is called the critical pressure.
Note the Following about Vapor Pressure
1. Changing the volume will have no effect on the vapor pressure provided that
equilibrium is obtained
2. Changing the amount of liquid will have no effect on the vapor pressure
provided that equilibrium is obtained
3. For gases to be liquefied, their temperatures should be brought to at least their
critical temperature since a gas above its critical temperature will occur as a
single phase regardless of the pressure applied
4. When a gas is liquefied, its pressure will remain at the critical pressure as this
pressure is the vapor pressure of the gas
Vapor Pressure of Solids
Molecules vibrate about their equilibrium positions in a solid. These vibrations result
in collisions among neighboring molecules where a small fraction of molecules may
attain enough kinetic energy to overcome intermolecular forces and enter the gaseous
state. This process is called sublimation and the pressure exerted by a vapor in
equilibrium with its solid is called the equilibrium vapor pressure of the solid. An
example of such a phenomenon is the sublimation of solid iodine as in the figure
below:
It should thereby be realized that the vapor pressure of solid ionic compounds (like
NaCl) is much less than the vapor pressures of covalent solids like naphthalene. This
is because ionic bonds are stronger than London forces present in naphthalene.
Boiling Points
The boiling point of a liquid is defined as the point at which its vapor pressure equals
the atmospheric pressure. The temperature will be constant as far as the liquid
continues to boil. Increasing the heat input to the liquid will not affect the boiling
point but rather increase the boiling rate. As the external pressure is increased, the
boiling point is increased as well. The normal boiling point of a liquid is its boiling
point at 1 atm. Boiling points of liquids can indicate how strong the intermolecular
forces are. As the intermolecular forces are increased, boiling points are increased.
Liquid
Hvaporization, kJ/mol
Boiling Point (oC)
Water, H2O
44.0
100
HF
30.2
17
NH3
23.6
-33
Each water molecule is connected to four other molecules (four hydrogen bonds).
While each HF is connected to two other molecules (two hydrogen bonds). Ammonia,
on the other hand, has one pair of nonbonding electrons and can thus be bonded to
two other molecules (one through the nitrogen and another through the nitrogen of
another ammonia molecule.
Water has the highest boiling point of the three compounds as each water molecules is
connected to four hydrogen bonds while HF is connected to only two. Although
fluorine is more electronegative than oxygen, the four hydrogen bonds in water weigh
more than the two bonds in HF. Ammonia is connected to two hydrogen bonds which,
in addition to lower electronegativity of nitrogen, makes ammonia of lowest boiling
point of the three compounds.
Freezing Points
At a given pressure, the temperature at which the liquid and solid forms of a substance
coexist is called the freezing or melting point. In addition, one can define the molar
heat of crystallization (HCryst) as the amount of energy that must be removed from
one mole of a liquid to convert it to a solid at the same temperature. In the same
mannar, the molar heat of fusion (Hfus) is the energy needed to convert 1 mole of the
solid to liquid at the same temperature.
(HCryst) should equal (Hfus) but with opposite sign. Usually, (Hfus) is much smaller
than (Hvap).
Example
Find the energy in kJ necessary to melt 1.00 g of ice. Hfus of ice = 5.98 kJ/mol
Solution
Melting of one mole of ice requires 5.98 kJ, therefore, find moles of ice present in
1.00 g and find energy required.
Energy needed = Hfus * Number of moles
Energy needed = 5.98 kJ/mol * {1.00 g/(18.0 g/mol)} = 0.332 kJ
Heating and Cooling Curves
When heat is added to a solid its temperature will rise till the melting point is reached
where the temperature stays constants till the entire solid is converted to liquid. When
extra heat is added to the system, the temperature of the liquid starts to rise till the
boiling point is reached where the temperature stays constant till all the liquid is
converted to vapor. This can be represented by a heating curve as below:
The inverse of the heating curve is the cooling curve whereby a gas is converted to a
liquid then to a solid.
Sometimes, during the cooling cycle, the temperature may drop below expected
freezing point. This may occur due to fast cooling where molecules may not be
oriented properly to fit into the crystalline lattice. The solution in this case is called a
supercooled solution. Some substances, like glass, never achieve the crystalline state
as their melt is cooled to the freezing point. These substances are called amorphous
solids.
Example
Find the energy needed to convert 36.0 g of ice at -23 oC into vapor at 120 oC. Hfus =
5.98 kJ/mol and Hvap = 44.0 kJ/mol, specific heat of water is 4.184 J g-1 oC-1, specific
heat of ice is 2.06 J g-1 oC-1, and specific heat of steam is 1.99 J g-1 oC-1.
Solution
Number of moles = 36.0/18.0 = 2.00 moles
Following the heating curve above, ice should first be
1. It will require energy to be converted to ice at 0 oC (heat capacity 1)
2. It will require the heat of fusion to convert to liquid at 0 oC
3. It will need energy to be converted to liquid at 100 oC, boiling
temperature for water (heat capacity 2)
4. It will require the heat of vaporization to be converted to water vapor
at 100 oC
5. It will require energy to raise the temperature of the vapor to 120 oC
(heat capacity 3)
Therefore, five energy terms should be summed together. Steps 1, 3, and five can be
summed together as heat capacity term:
Energy required = (heat capacity term) + number of molesHfus + Hvap)
Energy required = {36.0 g * 0.00206 kJ g-1 oC-1 {0 – (-23)} oC * 36.0 g * 0.004184 kJ
g-1 oC-1 {100 – (0)} oC + 36.0 g * 0.00199 kJ g-1 oC-1 {120 – (100)} oC} + 2.00 mol *
(5.98 + 44.0)kJ/mol = 118.2 kJ
Phase diagrams
A phase diagram is a graphical representation of the conditions of temperature and
pressure at which solids, liquids, and gases (vapors) exist, either as single phases or
states of matter or as two or more phases in equilibrium with one another. The
different regions of the diagram correspond to single phases, or states, of matter.
Straight or curved lines where single-phase regions adjoin represent two phases in
equilibrium.
Carbon Dioxide
The pressure at the triple point O (The point O defines the unique temperature and
pressure at which the three states of matter, solid, liquid, and gas, coexist in
equilibrium). A line at P = 1 atm intersects the sublimation curve, not the vapor
pressure curve. If solid CO2 is heated in an open container, it sublimes away at a
constant temperature of –78.5°C. It does not melt at atmospheric pressure (and so is
called "dry ice"). Because it maintains a low temperature and does not produce a
liquid by melting, dry ice is widely used in freezing and preserving foods.
We can obtain liquid CO2 at pressures above 5.1 atm and encounter it most frequently
in CO2 fire extinguishers. All three states of matter are involved in the action of these
fire extinguishers. When the liquid CO2 is released, most of it quickly vaporizes. The
heat required for this vaporization is extracted from the remaining CO2(l), which has
its temperature lowered to the point that it freezes and falls as a CO2(s) "snow." In
turn, the CO2(s) quickly sublimes to CO2(g). All of this helps to quench a fire by (1)
displacing the air around the fire with a "blanket" of CO2(g) and (2) cooling the area
somewhat.
Water
The phase diagram of water presents several new features. One is the fact that the
fusion curve has a negative slope. The melting point of ice decreases with an increase
in pressure, and this is rather unusual behavior for a solid. However, because large
changes in pressure are required to produce even small decreases in the melting point,
we do not commonly observe this melting behavior of ice.
Melting or fusion
(s
l)
Freezing or crystallization
(l
s)
vaporization
(l
g)
condensation
(g
l)
sublimation
(s
g)
deposition
(g
s)
Following are two useful generalizations about the changes that occur when crossing
a two-phase equilibrium curve in a phase diagram.
From lower to higher temperatures along a constant-pressure line (an isobar),
enthalpy increases (Heat is absorbed.)
From lower to higher pressures along a constant-temperature line, volume decreases.
(The phase at the higher pressure has the higher density.)
The latter of these two generalizations helps us to understand why a fusion curve
generally has a positive slope. Typical behavior is for a solid to have a greater density
than the corresponding liquid.
Supercritical Fluids
Because the liquid and gaseous states become identical and indistinguishable at the
critical point, it is difficult to know what to call the state of matter at temperatures and
pressures above the critical point. For example, this state of matter has the high
density of a liquid and the low viscosity of a gas. The term that is now commonly
used is "supercritical fluid " (SCF). Above the critical temperature, no amount of
pressure can liquefy a supercritical fluid. The only way to observe the liquid–vapor
interface is to cross the phase boundary below the critical isotherm. Note that in the
present case we could observe the liquid–vapor interface by lowering the pressure on
the liquid to a point on the vapor pressure curve.