surface chemistry in one page
advertisement

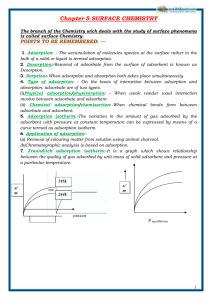
SURFACE CHEMISTRY Adsorption Adsorbate & Adsorbent. Desorption. Adsorption and Absorption Sorption Enthalpy of adsorption Surface area & Adsorption Physisorption Chemisorption Adsorption Isotherm Freundlich isotherm Applications of Adsorption Promoters and Poisons Homogeneous catalysis Heterogeneous catalysis Activity of a solid catalyst Selectivity of a catalyst Shape- Selective Catalysis by Zeolites Characteristics of enzyme catalysis Catalysts used in industrial process. Colloids Lyophilic colloids Lyophobic colloids Multimolecular colloids Macromolecular colloids Associated colloids (Micelles) Kraft Temperature (Tk) CMC Cleansing action of soaps Electrical disintegration or Bredig’s Arc method Peptization Dialysis: Tyndall effect Conditions effect for Tyndall Accumulation of molecular species at the surface rather than in the bulk of a solid or liquid. The substance adsorbed is called adsorbate. And the surface provider is called adsorbent. The process of removing an adsorbed substance from a surface. In adsorption, the substance is concentrated only at the surface while in absorption, the substance is uniformly distributed throughout the bulk of the solid. Both adsorption and absorption can take place simultaneously. Adsorption is exothermic ΔH is -ve. There is decrease in entropy, ΔS is -ve. For a process to be spontaneous, ΔG must be negative, so ΔH should have sufficiently high-ve value. Adsorption increases with the increase of surface area of the adsorbent. Thus, finely divided metals and porous substances having large surface areas are good adsorbents. van der Waals’ forces / not specific /reversible /easily liquefiable gases adsorbed readily./ Enthalpy of adsorption is low/ Low temperature is favourable / Multimolecular layers. chemical bond /highly specific / irreversible/Enthalpy of adsorption high / High temperature is favourable / / unimolecular layer. Variation of amount of gas adsorbed by the adsorbent with pressure at constant temperature can be expressed by means of a curve termed as adsorption isotherm. X / m = k.P1/n (n > 1) log x/m = log k + 1/n log P High vacuum/masks/Control of humidity/ Removal of colour/ catalysis/ curing diseases/ N2(g) + 3H2(g) ………… 2NH3(g) Reactants and the catalyst are in the same phase. Reactants and the catalyst are in different phases. Depends upon strength of chemisorption .Reactants must get adsorbed reasonably strongly. The selectivity of a catalyst is its ability to direct a reaction to yield a particular product. Depends upon the pore structure of the catalyst and the size of the reactant and product molecules. Example:- ZSM-5 (a zeolite) converts alcohols directly into gasoline (petrol). Highly efficient/ Highly specific / active under optimum temperature& pH/ Increasing activity in presence of activators and co-enzymes/ Influence of inhibitors and poisons Haber’s process (NH3) Finely divided Fe, Mo - promoter; Ostwald’s process(HNO3) Pt Contact process(H2SO4) Pt or vanadium pentoxide (V2O5); Diameters of the particles is between 1 and 1000 nm. Liquid-loving/ can be reconstituted/ reversible/easily prepared/stable/ eg- gum, starch. Liquid-hating/ not be reconstituted/ irreversible/ prepared by special methods/ precipitated on addition of electrolytes/heating/shaking/ not stable./ eg –metal sols , sulphide sols. Large number of atoms or molecules of a substance aggregate eg- gold sol , sulphur sol. Macromolecules in suitable solvents. Eg-starch, cellulose, proteins and enzymes; polythene. Substances at low conc behave as strong electrolytes, but at high conc exhibit colloidal behaviour due to the formation of aggregates(micelles.) eg- soaps and detergents. Formation of micelles takes place only above a particular temperature. Formation of micelles takes place only above a particular conc. Soap molecules form micelle around the oil droplet in such a way that hydrophobic part of the stearate ions is in the oil droplet and hydrophilic part projects out of the grease droplet like the bristles . Since the polar groups can interact with water, the oil droplet surrounded by stearate ions is now pulled in water and removed from the dirty surface. This process involves dispersion as well as condensation. Colloidal sols of metals such as gold, silver, platinum, etc., can be prepared. Process of converting a ppt into colloid by shaking it with dispersion medium in the presence of a small amount of electrolyte. The electrolyte used for this purpose is called peptizing agent. Removal of dissolved substance from colloidal by diffusion through a suitable membrane. Colloidal particles scatter light in all directions. Scattering of light illuminates the path of beam of the light. (i) Diameter of particles is not much smaller than wavelength of the light used; (ii) Refractive indices of the phase and medium differ greatly in magnitude +ve charged sols Hydrated metallic oxides,/methylene blue sol. /Haemoglobin (blood) -ve charged sols Metals,/ sulphides/ eosin, congo red / starch, gum, gelatin, clay,charcoal. Preferential adsorption of ions . The sol particles acquire positive or negative charge by preferential adsorption of +ve or –ve ions. When two or more ions are present in the dispersion medium, preferential adsorption of the ion common to the colloidal particle usually takes place. AgI/I– Zeta potential Potential difference between the fixed layer and the diffused layer. Electrophoresis The movement of colloidal particles under an electric potential is called electrophoresis. Electroosmosis If electrophoresis is prevented , it is observed that the dispersion medium begins to move in an electric field. Coagulation(ppt) The process of settling of colloidal particles is called coagulation or precipitation of the sol. Reason of Coagulation Electrophoresis/mix oppositely charged sols/boiling/persistent dialysis/addition of electrolytes. Hardy-Schulze rule The greater the valence of the flocculating ion added, the greater is its power to cause precipitation. For coagulation of a negative sol, : Al3+>Ba2+>Na+ For coagulation of a negative sol :[Fe(CN)6]4– > PO4 3– > SO4 2– > Cl– Coagulating value Min. conc of an electrolyte in millimoles/L required to cause precipitation of a sol in 2 hours Emulsions Liquid-liquid colloida (i) Oil dispersed in water (O/W type) - milk and vanishing cream . (ii) Water dispersed in oil (W/O type).- butter and cream. Demulsification Emulsions can be broken into constituent liquids by heating, freezing, centrifuging, etc. Artificial Rain By throwing electrified sand , spraying a sol of opposite charge to the clouds from aeroplane Formation of Delta River water is a colloidal solution of clay. Sea water contains a number of electrolytes. When river water meets the sea water, the electrolytes present in sea water coagulate the colloidal solution of clay resulting in its deposition. Electrical precipitation of The smoke, before it comes out from the chimney, is led through a chamber containing smoke(Cottrellppter) plates having a charge opposite to that carried by smoke particles. The particles on coming in contact with these plates lose their charge and get precipitated. Charge on colloidal particles Origin of charge on colloidal particles