Steve's Musculoskeletal
advertisement

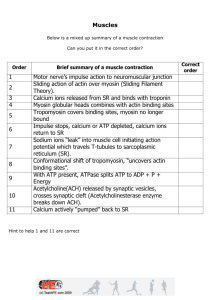
S Tran Musculoskeletal System Lecture 1: Molecules of Movement Learning Objectives Appreciate that there are a large number of molecular motors, each with its assigned role. Linear molecular motors are associated with polymeric filaments. Which filaments are myosins and kinesins associated with? Which molecular motor is that found predominantly in muscle cells? Describe the structure of myosin II. Describe the structure of myosin filaments. Describe the structure of actin filaments. What are sarcomeres? Draw a sarcomere in longitudinal section. Activation of the skeletal muscle cell results from a cascade of events: (i) What causes depolarisation of the muscle cell membrane? (ii) How does cell membrane depolarisation propagate: (a) along the cell surface? And (b) into the core of the muscle cell? (iii) Describe how calcium ions are released from intracellular calcium stores (sarcoplasmic reticulum). Which membrane-bound proteins are involved in this process? (iv) Describe the fate of calcium ions during activation of the myofilaments. (v) How is the thin filament affected by calcium? What are the consequences? What is relaxation? What happens to calcium ions during relaxation? Muscles The molecules of movement are protein assemblies which convert chemical energy into mechanical work. Energy for biological motors is usually derived directly from the hydrolysis of ATP or from an ionic gradient (e.g. H+ or Na+ gradient). Motors: Linear Motors 1. Actin filaments for Myosins 2. Microtubules for Dynein, Kinesins and NCD 3. DNA or RNA for helicases & topoisomerases 4. Other linear motors rely on filament polymerisation (actin polymerisation in listeria) Rotary Motors - require stators 1. E.g. Flagellar motor, ATP-hydrolysing F1 portion of the F1F0-ATPase Oscillatory Motors 1. Found in Cilia & require cross-linked microtubules bundles (axonemes) & powered by dynein pg. 1 S Tran Intracelluar Transport Kinesins: Move towards the plus end of a microtubule (towards the axon terminal) Dyneins: Move towards the minus end of a microtubule (towards the soma) Microtubule Motor Proteins that move in opposite directions. Have two globular ATP-binding heads and a tail. o The tail binds stably to some cell components such as a vesicle or an organelle. o Heads have ATP-hydrolysing (ATPase) activity, which provide the energy for a cycle of conformational changes in the head that enables it to move along the microtubule by a cycle of binding, release and rebinding to the microtubule - (‘Walking’ along the filament). Kinesin pulls ER towards the cell edge, whilst dynein pulls the Golgi Apparatus towards the cell centre. Cilia and Flagella 9+2 arrangement of microtubules. Movement is produced by teh bending of its core as the microtubules slide against each other - Instead of sliding, the microtubules bend due to the attached linking proteins (Nexin) Ciliary Dynein is like cytoplasmic dynein, it attaches to the microtubule by its tail whilst its head interacts with adjacent microtubules. pg. 2 S Tran Myosin II – Thick Filament has 3 main functions in the body: 1. cytoplasmic- cytokinesis, phagocytosis, cell shape and/or polarity 2. smooth muscle- smooth muscle contraction 3. skeletal muscle- skeletal muscle contraction 294 Myosin molecules bind together to make a myosin filament. The myosin tails form the shaft of the thick filament, whereas the myosin heads (Cross-bridges) project outwards towards thin filaments (actin) Actin (Thin) Filaments Contain actin, troponin and tropomyosin. Extend from anchoring points of Z discs. Two actin filaments twist into a helix. Each actin molecule has a myosin binding site. But small amounts of troponin and tropomyosin regulate the cross-linking. In relaxed state, the binding sites are blocked. pg. 3 S Tran Skeletal Muscle Activation A. Initiation: Depolarisation of Muscle Cell 1. 2. 3. 4. An action potential arrives at the nerve terminal The voltage-activated Ca2+ channels open, allowing Ca2+ influx. Ca2+ influx attracts pre-synaptic vesicles to the active zones. vesicles fuse to the pre-synaptic membrane and release Acetylcholine in packets by the process of exocytosis 5. Acetylcholine diffuses across the synaptic cleft to bind to the nicAChR (receptors) on the junctional folds. There is activation of channels and an influx of Na+ 6. Na+ influx depolarizes the muscle. This is called an end plate potential (e.p.p) B. Propagation of Action Potential Along the Sarcolemma: o Bidirectional propagation of the action potential occurs along the sarcolemma. o This causes excitation of muscle fibre along its whole length so that all sarcomeres contract simultaneously Into Core of the Muscle Cell: o The action potential is propagated into the muscle fibre via the T tubule. o There are 2 T tubules per sarcomere; these encircle the myofibril at the AI junction. o T tubules communicate with the extracellular space C. Result of Action Potential: Calcium Release Sarcoplasmic reticulum on both sides of the T tubules communicates with the T tubule via junctional feet. Depolarisation of the T-tubules results in a signal to the sarcoplasmic reticulum terminal cisternae. Ca2+ channels open in the sarcoplasmic reticulum and Ca2+ move along a concentration gradient into the sarcoplasm around the myofibrils D. Activation of Myofilaments Ca2+ ions bind to troponin and cause the troponin-tropomyosin complexes to move away from the myosin binding sites on actin. pg. 4 S Tran Once these sites are few, the contractile cycle begins: 1. ATP Hydrolysis Myosin heads have an ATP binding pocket and ATPase. This hydrolysis reaction energises the myosin head. (ADP & Pi still attached) 2. Cross-Bridging of the myosin to actin. Energised myosin head attaches to the myosin-binding site on actin & releases the hydorlysed phosphate group 3. Power Stroke Release of the phosphate group triggers the power stroke of contraction. During this, the ATP/ADP pocket on the myosin head is opened, which causes the myosin head to rotate and release the ADP. Myosin head generates force as it rotates towards the centre of the sarcomere, sliding the thin filament past the thick towards the M line. 4. Detachment of Myosin from Actin. At the end of the power stroke, the myosin head remains firmly attached to the actin until an ATP molecule binds. Hence an ATP causes the release of the actin filament. 5. This contractile cycle continue for as long as ATP is available and Ca2+ level near the thin filament is sufficiently high. (No ATP = Rigor Mortis) E. Termination: Relaxation Relaxation of the muscle is Ca2+ dependent. Upon repolarisation, Ca2+ is actively pumped back into the sarcoplasmic reticulum. o Concentration of Ca2+ drops and is no longer bound to troponin o Myosin binding sites become covered by tropomysosin again: prevents ‘walking’ / further contractions. pg. 5 S Tran Lecture 2: Molecules Working Together: Force & Movement Learning Objectives Appreciate that the myosin cross-bridge cycle is the elementary process resulting in force generation and shortening by all muscle cells. Draw a diagram of the main steps in the cross-bridge cycle, showing the changes in cross-bridge structure and how ATP is involved. List 3 key features of the cycle (ATP used in each cycle, one-way sliding, and small step size). Know that the following factors control the amount of force produced in vivo, and briefly explain the mechanisms involved. - recruitment of motor units - frequency of activation (covered earlier) - sarcomere length and filament overlap - speed and direction of movement List the metabolic sources of ATP and briefly outline the conditions in which each is important. Smooth Muscle: Describe its main structural features. Give examples of tissues contain smooth muscle and state the nature of the signal that triggers contraction. Describe the intracellular events during activation of contraction and during relaxation. Be able to explain how this is different from the corresponding events in skeletal and cardiac muscle. Cardiac Muscle: Describe its main structural features. Be aware that the crossbridge cycle is like that in skeletal muscle. Describe the sources of Ca for activation and how this differs from skeletal muscle. Types of Contractions: Isometric: force without length change Isovelocity: Muscle shortens or is stretehced at constant velocity Isotonic: constant force during shortening or lengthening pg. 6 S Tran Cross-Bridging Cycle: Key Features 1. ATP is used in each cycle to provide energy. 0 ATP = Rigor Mortis 2. Direction of filament sliding is one-way (Thin filament towards the centre) a. Shorterning only. Not Lengthening 3. Step size is small: sliding produced is only ~1% of sarcomere length a. Successional cycles cause large movements Variation in Force 1) Recruitment Vary the number of motor units that are active and thus vary force. Motor unit: a motor neuron & all associated muscle fibres Motor Unit size varies (muscle fibres per motoneuron) Scattered distribution within muscle. 2) Frequency of Stimulation Force increase with stimulation frequency Force summates, however AP doesn’t Unfused tetanus: individual stimuli detected (Wibbly Line) Fused tetanus: no detection of single stimulus (Straight Line) The higher stimulation frequencies, more Ca2+ is released into the space around the actin filaments. Hence more Ca2+ bound troponin causes more cross-bridges to attach increased muscle force. pg. 7 S Tran 3) Force is proportional to filament overlap 4) Force depends on velocity & direction of movement Large number of cross-bridges acting together. Force depends on the: o Proportion of bridges that are attached o Force each attached bridge produces ATP Supply of Muscles ATP = ADP + Inorganic phosphate + work + Heat 1. Creatine Phosphate (PCr) Buffers ATP concentration Occurs in every contraction involving twitches PCR is dedicated to the above reaction and stays in the muscle fibres. 2. Glycolysis No oxygen required. Products: Pyruvate or Lactic Acid produce more ATP with oxygen. 3. Oxidation Requires oxygen adn makes more ATP per carbon when metabolised Substrates: Glucose, glycogen and fatty acids. Products: ATP, carbon dioxide and water. ACTIVITY FUEL SUPPLY DURING ACTIVITY Sprint Sustained, low intensity Sustained, high intensity ATP, PCr ATP, PCr, fatty acids (glycogen and glucose) ATP, PCr, glucose and glycogen (fatty acids) pg. 8 S Tran Smooth Muscle Appear ‘smooth’ – not striated Contain filament but not in an arranged way Contractile mechanism similar to skeletal and cardiac but activation by Ca2+ is different Normally arranged in sheet forming walls of tubular organs, e.g. blood vessels, the gastrointestinal tract and reproductive tract. Autonomic nervous system controlled. 6a) Describe its main structural features of smooth muscle The cells appear ‘smooth’ – not striated They contain filament but not in an arranged way The contractile mechanism (cross bridge cycle) is similar to skeletal and cardiac muscle. However activation by Calcium is by a different mechanism to that of skeletal and cardiac muscle. 6b) Give examples of tissues contain smooth muscle and state the nature of the signal that triggers contraction Smooth muscle is normally arranged in sheet forming walls of tubular organs, such as blood vessels, the gastrointestinal tract and the reproductive tract The nature of the signal that triggers contraction of smooth muscle is that of the autonomic nervous system 6c) Describe the intracellular events during activation of contraction and during relaxation. Be able to explain how this is different from the corresponding events in skeletal and cardiac muscle. Calcium ions flow into the smooth muscle cytosol from both the interstitial fluid sarcoplasmic reticulum. Because there are no transverse tubules in smooth muscle fibers, it takes longer for calcium ions to reach the filaments in the centre of the fiber and trigger he contractile process. This accounts for the slow onset and prolonged contraction of smooth muscle. Several mechanisms regulate contraction and relaxation of smooth muscle cells. A regulatory protein called calmodulin binds to calcium ions in the cytosol (recall that troponin has this role in striated muscle fibers). After the binding of calcium calmodulin activates an enzyme called myosin light chain kinase. The myosin light chain kinase uses ATP to phosphorylate a portion of the myosin head. Once the phosphate group is attached the myosin can attach to the actin and contraction can occur. Myosin light chain kinase works rather slowly, also contributing to the slowness of smooth muscle contraction. 7a) Describe its main structural features of cardiac muscle Compared to skeletal fibers, cardiac muscle fibers are shorter in length and less circular in transverse section pg. 9 S Tran The ends of cardiac muscle fibers connect to neighbouring fibers by irregular transverse thickenings of the sarcolemma called intercalated discs. The disc contains desosomes which hold the fibers together and gap junctions which allow muscle action potentials to conduct from one fiber to the next. Mitochondria are bigger and more numerous in cardiac muscle cells Cardiac muscle fibers have the same bands, zones as skeletal muscle The sarcoplasmic reticulum of cardiac muscle is somewhat smaller than that of the skeletal muscle. As a result, cardiac muscle has a smaller intracellular reserve of Calcium ions. 7b) Be aware that the crossbridge cycle is like that in skeletal muscle. 7c) Describe the sources of Ca for activation and how this differs from skeletal muscle. Timing of ventricular action potential and isometric force Force is already relaxing during the membrane’s refractory period AP scale (mV) Force scale (N) Therefore cardiac muscle CANNOT produce a fused tetanus Sources of Ca: from SR and entry into the cell during the plateau of the action potential via voltage-gated Ca channels Designed for pumping 0 Time (ms) 20 0 Normally not enough Ca to bind to all troponins. Anything that changes Ca changes force pg. 10 S Tran Lecture 3: Molecules of Movement Learning Objectives You should be able to define the following terms: - Twitch - Tetanus - Summation - Electromyogram You should know (in qualitative terms at least) how long a muscle action potential lasts compared to how the force response lasts. Defintion: Twitch: Force and/or movement response to a single stimulus Tetanus: Force and/or movement response to stimuli in quick succession More force is produced during repetitive stimulation (tetanus) than during a twitch (single stimulation) During titanic stimulation, the force response summate but the action potentials do not Summation Increased strength of muscle contraction that results when stimuli follow one another in quick succession Electromyogram (EMG) Technique used to record the electrical activity in muscle both at rest and during contraction. Method: o During EMG, a needle electrode is inserted into muscle. o Electrical activity in the muscle is displayed on a cathode ray oscilloscope or heard on a speaker. o Records action potential produced in skeletal muscle fibres Extracellular recording- measures electrical potential between 2 locations, both on the outside of the muscle fibre. Duration of a nerve impulse is 0.5-2.0 msec, ut a muscle action potential is considerably longer – about 1-5 msec for skeletal muscle and 10-300 msec for cardiac and smooth muscle fibers. Percutaneous Electrical Nerve Stimulation (PENS) An electrical stimulation of the peripheral nerve by a needle electrode inserted through the skin. Used to relieve pain pg. 11