isotopes and average atomic mass
advertisement

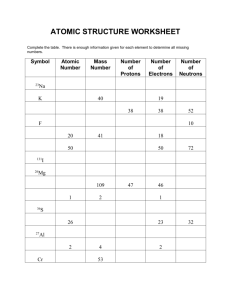
ISOTOPES AND AVERAGE ATOMIC MASS Isotopic Abundance: the relative amount in which each isotope is present in an element Atomic mass unit: inconvenient to measure the mass of subatomic particles. Chemists use atomic mass unit (amu) which is equivalent to 1.66x10-24, the mass of a proton. Average Atomic Mass: the average mass of all the element isotopes Calculating Average Atomic Mass Example 1: Naturally occurring silver has two isotopes Ag – 107 with a atomic mass of 106.9 u and a relative abundance of 51.8% and Ag -109, with an atomic mass of 108.9 amu and a relative abundance of 48.2 %. Calculate the average atomic mass of silver. Average atomic mass = amu x percent isotope abundance (decimal) Average atomic mass = 106.9 amu (0.518) + 108.9 amu (0.482) = 107.9 Example 2: Boron exists as two naturally occurring isotopes 105B (10.1 amu) and 115B (11.01 amu) Calculate the relative abundance of each isotope of boron. Let x = the percent abundance of Boron -10. Which isotope you choose to represent x is irrelevant. We could have chosen Boron -11 as x. Average atomic mass = 10.81 (from the periodic table) Average atomic mass = x(atomic mass B-10) + (1-x)(atomic mass B-11) 10.81 = x (10.01) + (1-x) (11.01) x = 0.2000 Percent abundance of B-10 is 0.200 And B-11 = 1 – 0.200 = 0.8000 1. Isotopes and Average Atomic Mass Practice Problems Determine the number of protons, neutrons, and electrons for the following a. 28 b. 131 p_____ n_____ e_____ c. 207 p_____ n_____ e_____ d. 127 -1 p_____ n_____ e_____ Si Xe Pb+2 I p_____ n_____ e_____ 2. Silver exists as 51.84% 107Ag and 48.16% 109Ag. The mass of 107Ag is 106.90509 u and the mass of 109Ag is 108.90476 u. What is the average atomic mass of silver? 3. Natural potassium consists of 93.1% K-39 and 6.90% K-41. Do these relative values confirm the accepted average atomic mass for natural potassium (from the periodic table)? 4. The average atomic mass of copper is 63.55 u. If the only two isotopes of copper have masses of 62.94 amu and 64.93 u, what are the percentages of each? 5. Naturally occurring chlorine consists of chlorine-35 with an isotopic mass of 34.968852 u and chlorine-37 with an isotopic mass of 36.965903 u. The average atomic mass of chlorine is 35.4527 u. Calculate the percent abundance of each isotope.