Post-lab
advertisement

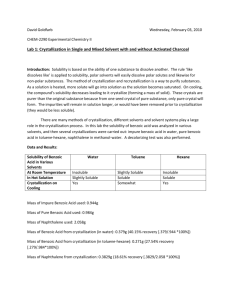
© Professor Kathleen V. Kilway, Department of Chemistry, University of Missouri – Kansas City, 2007 Name Lab Section GTA 1. Station Crystallization Post-lab report Fill out the appropriate sections below. Show all work. All answers are to be typed, including calculations and correct spelling and grammar is required. All calculations must be shown for credit and correct significant figures are required. Results Amounts and units Initial weight of salicylic acid Final weight of salicylic acid Volume of water used to recrystallize salicylic acid Initial weight of benzoic acid Final weight of benzoic acid Volume of water used to recrystallize benzoic acid Initial weight of phthalic acid Final weight of phthalic acid Volume of water used to recrystallize phthalic acid Compound you took melting point of for Part 1 melting point of compound above (°C) Initial weight of naphthalene Final weight of naphthalene Initial weight of biphenyl Final weight of biphenyl Compound you took melting point of for Part 2 melting point of compound above (°C) Crystallization 1 © Professor Kathleen V. Kilway, Department of Chemistry, University of Missouri – Kansas City, 2007 Complete the following questions and submit with your report. 1. The solubility of phthalic acid in 100 mL of water is given in The Handbook of Chemistry and Physics as 0.54 g at 14 C and 18 g at 99 C. a. What volume of water was theoretically required to dissolve your phthalic acid? b. How much phthalic acid should you have theoretically recovered (you may assume that ice-bath temperature is 14 ºC)? c. Calculate the % difference for the volume. d. Calculate the % difference for the recovered mass. 2. Determine the solubility of benzoic acid at 99 ºC and 14 ºC 3. Determine the solubility of salicylic acid at 99 ºC and 14 ºC 4. What is your percent recovery for a. naphthalene? b. biphenyl? Crystallization 2 © Professor Kathleen V. Kilway, Department of Chemistry, University of Missouri – Kansas City, 2007 5. You are given a mixture of benzoic acid and sand. Write a short procedure to recover pure benzoic acid. 6. Explain why one would recrystallize a solid from a solvent pair. Explain how it works in terms of solubilities. What were you observing when your ethanol – water solutions became cloudy? Crystallization 3