Terms
advertisement

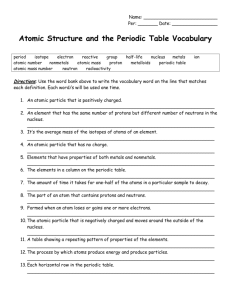
Chemistry Unit Vocabulary Terms 1. Proton – positively charged subatomic particle found in the nucleus 2. Neutron – neutral (no charge) subatomic particle found in the nucleus 3. Electron – negatively charged subatomic particle found in the electron cloud 4. Atom - the smallest particle of an element that still has the properties of that element 5. Element - An element is a substance made up of only one kind of atom 6. Atomic number – the number that identifies an element as the number of protons in the nucleus 7. Mass number - the mass of a specific isotope of an element (number of protons + number of neutrons) 8. Average mass - the weighted average of the mass numbers of all the isotopes of an element. ( a.k.a. average atomic mass) 9. Nucleus - dense core of an atom that carries a positive charge and composed protons & neutrons 10. Electron cloud - a cloud of negative charge that surrounds the nucleus of an atom composed of electrons. 11. Isotopes - Atoms of the same element with different numbers of neutrons 12. Metalloids or semi-metallics or semi-conductors – elements the have properties of both metals & non metals that are found along the stair step that divides the periodic table 13. Transition metals – mildly reactive metal elements located in the “B” groups or 3-12 14. Alkali metals – very reactive metal elements located in group 1A or 1 15. Alkaline earth metals – active metal elements located in group 2A or 2 16. Noble gases – inert (non reactive) gas elements located in group 8A or 18 17. Halogens – very active nonmetallic elements located in group 7A or 17 18. Metals – elements having 3 or fewer electrons in their outer energy level 19. Nonmetals – elements having 5 or more electrons in their outer energy level 20. Periodic Table Group or Family – the elements in a column on the periodic table. The group number relates the number of valence electrons in each of the elements in the group 21. Periodic Table Period - the elements in a row on the periodic table. The period number gives the number of energy levels of the elements in that period.