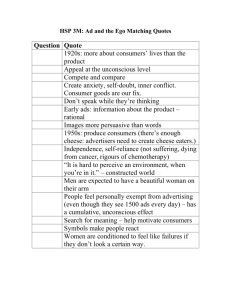
food science experiments
advertisement

FOOD SCIENCE EXPERIMENTS Find yourself a recipe and you can cook one dish. Teach yourself the science of cooking and you can cook creatively forever. Background This resource was created as our implementation project for the Re-Newing the Food Studies Curriculum course at the UBC Summer Institute in August 2005, presented by Dr. Linda Peterat, Dr. Gale Smith, and Dr. Mary Leah de Zwart. Some of the experiments were presented in a workshop session during the course, colleagues contributed some, and some were gathered from the Internet and other sources. Format Most experiments are presented with two handouts - one for students and a second teacher handout that includes background information, the expected results and helpful hints. Where applicable, a student observation chart has been included to save class time. Note: Observations and/or intended results included here are meant as a guide. Before using these experiments teachers are encouraged to pre-test them, and adapt the instructions and questions to suit the course and level of their students. These experiments can be done individually, as class demonstrations, or as part of a station unit grouping those covering a similar topic. Kathy Nicholson Susan Petersen Table of Contents Experiment Page 1. Plastic bag ice cream 2. Digestive system 3. Cooking fresh fruit 4. Eggs #1 5. Eggs #2 6. Visible Fats 7. Invisible Fats 8. Meat packaging 9. McMush 10. Enzyme Break-down of Protein 11. Chameleon in the Kitchen 12. How to get iron out of breakfast cereal 13. Yeast 14. Gluten 15. Baking is a gas 16. Mixing methods 17. Cheese 18. Resource list 3-4 5 6-8 9-12 13-17 18-20 21-23 24-26 27-28 29-31 32-33 34-35 36-38 39-41 42-44 45-50 51-58 59-68 Name: _________________________ Block: ______ Date: ______________ Experiment: Plastic Bag Ice Cream Ingredients/Supplies 1 small zipper-type freezer bag 1 large zipper-type freezer bag 60 mL sugar 125 mL milk 125 mL half-and-half cream 2 mL vanilla 1 plastic spoon 125 mL to 175 mL rock salt 175 mL crushed ice Procedure 1. Place the milk, sugar, vanilla and cream into the small freezer bag. Zip the bag. 2. Place the small bag inside the large bag and add in the crushed ice. 3. Pour some of the salt evenly over the ice. Continue with the ice and salt until the larger bag is completely full. Zip up the large bag. 4. Knead or roll back and forth on a table or desk top. Be careful not to put too much pressure on the bags. 5. After 10 minutes check the mixture to see if it is frozen. If not, continue kneading. 6. When the mixture is frozen, remove the smaller bag, rinse under cold running water to remove all traces of salt, and eat the ice cream directly from the bag. Scientific Principle _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Conclusions __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ Experiment: Plastic Bag Ice Cream TEACHER NOTES Scientific Principle Rock salt forces the ice surrounding the can of ice cream mix to melt. The "brine solution" or liquid that forms in the wooden bucket absorbs heat from the mix and gradually lowers the temperature of the mix until it begins to freeze. If there were no salt added to the ice, it would melt at 32 degrees Fahrenheit and eventually the ice water and mix would come to equilibrium at 32 degrees. The ice cream mix, however, does not begin to freeze until its temperature falls below 27 degrees. Therefore, in order to freeze the mix, we need a salt concentration, or a ratio of 5 cups of ice to 1 cups of salt. At this concentration, our brine temperature should remain constant at 8 to 12 degrees F. This will give the rapid cooling and freezing that is essential to making smooth creamy ice cream. More detailed information provided by David Winer, Bethesda, Maryland For ice to melt into liquid water it must absorb a lot of heat energy. In an ice cream maker this heat needed to melt the ice comes from the mix, and so the mix chills. If the ice were not combined with salt, the mix’s cooling would be slow and would stop when the mix reached the temperature of melting freshwater ice-- at 32 degrees. But salt makes ice melt faster, and drops the temperature to that of freezing salt water. The heat used to melt the ice is drawn much faster from the surroundings, which in this case consist of the melted ice AND the ice cream mix. Since the surroundings lose large quantities of heat to the ice, they cool rapidly and continue to cool until they reach the freezing temperature of melting saltwater ice. This temperature is below the cream mixture's freezing point Debriefing students -presentation of results to class -taste test -compare texture and appearance -complete before and after chart – taste, texture, appearance and flavour -discussion of what happened and why Helpful hints -double bag the ice bag to prevent leaks -squeeze air out of ice bag and milk bag to make kneading easier -knead on towelled surface to prevent slippage and to avoid moisture -using oven mitts helps to insulate hands from the cold and it’s also easier on the bags -this does work nicely with chocolate milk Source: MakeIceCream.com or www.teachnet.com/lesson/science/icecream051999.html Name: _________________________ Block: ______ Date: ______________ Experiment: Digestive System Ingredients/Supplies individually wrapped hard candies (nothing that could melt) a dozen small water guns newspaper a piece of chalk a small trash barrel small piece of paper that read as shown below (try to divide the parts equally with the exception of Anus. Only one student is needed to represent the Anus.) Procedure Place some individually wrapped candies in the centre of some newspaper. Cushion the candy well. Make two or three of these candy balls. These candy balls represent food. This food is passes through the digestive system. Starting in the mouth and ending in the rectum (see below). On some school yard pavement or on the floor, mark the following in a straight line, leaving about 2 feet between each. Pavement reads: Mouth Student slip reads: Saliva Esophagus Props. for students Spray bottles or water guns; students spray the food (enzymes) No student stands here; food just passes through Pancreatic Water guns; these enzymes moisten the food Juice Small Intestine Small Intestine Students use their hands to tear off the paper They distribute nutrients (candy) to entire body , and thus they pass around the Large Intestine Blood candy to all the students participating in the activity Stomach Rectum Anus One student represents the anus, disposing of the paper as it is passes to him/her; this student has the trash barrel. This activity illustrates what part of the digestive system is responsible for each particular duty in the process of digestion. Retrieved August 15, 2005 from http://student.biology.arizona.edu/sciconn/mcmush.html (Yes, it says mcmush, but there are lots of things there including the digestive system.) 6 Name:______________________________ source unknown Block:________ Date:__________________ Experiment: Cooking Fresh Fruit Ingredients/supplies 4 cooking apples 1 litre water spoons for stirring vegetable peeler chopping board tape to label pots 175 ml sugar measures clock/watch/timer knife 4 small saucepans Procedure 1. Prepare 4 cooking apples by washing, peeling, quartering and coring them. Cut each quarter into three or four slices. 2. Prepare 4 cooking solutions in 4 small saucepans as follows: (a) 250 ml water (b) 250 ml water and 25 ml sugar (thin syrup) (c) 250 ml water and 125 ml sugar (heavy syrup) (d) 250 ml water 3. Bring cooking mediums to a boil; add the slices from one apple to each saucepan and cover. Cook over medium to low heat until fruit is tender (8 –10 min.). Make sure all fruit is cooked for the same length of time. 4. To cooking solution (d) add 25ml sugar when the apples have been cooked and stir until dissolved. Scientific Principles _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Conclusions _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ 7 Name:_________________________ _____ Block:________ Date:__________________ Experiment: Cooking Fresh Fruit Apple Treatment 1. Water only 2. Thin syrup 3. Heavy syrup 4. Sugar added after cooking Taste Observation Chart Texture Amount of water 8 Experiment: Cooking Fresh Fruit TEACHER NOTES Scientific Principles Osmosis – the movement of water molecules from an area of low concentration (in this case, of sugar) through a semi-permeable membrane to an area of high concentration (in this case, of sugar) In this experiment the water has a low concentration of sugar and the apple has a high concentration of sugar. Sugar raises the density of the water. Water has a low density, thin syrup has a medium density and the heavy syrup has the highest density. The syrup prevents cell breakdown. Apple Treatment 1. Water only Taste Texture Amount of water -bland -stringy -soft -lots of water -sweet -apple flavour -thick -little water -too sweet -sugar flavour -gel -thick -very little water -bland -soft -lots of water 2. Thin syrup 3. Heavy syrup 4. Sugar added after cooking Cooking fruit Questions: 1. How did the presence of sugar affect the breakdown of the apple? 2. How did the presence of sugar affect the taste of the product? 9 3. How did the presence of sugar affect the thickness of the product? 4. What recipes would you apply this theory to? 5. How did outside variables (room temperature, amount of water, size of pot etc.) affect the results? 10 source: unknown Name________________________________ Block:________ Date:___________________ Experiment: Eggs Ingredients/supplies 4 raw eggs water 2 saucepans Procedure Part 1: Cooking methods of eggs 1. Place two raw eggs in their shells in two saucepans (one in each saucepan) containing enough cold water to cover the eggs by 5 cm. 2. Put the lids on the saucepans and bring the water to a boil in both saucepans. 3. Remove one saucepan from the heat. Do not drain the water or take the lid off the saucepan. Allow the egg to sit in the hot water for 20 minutes. At the end of the 20 minutes, drain off the water and pour cold water over the egg. 4. For the second egg, allow the water to boil rapidly for 10 minutes. Remove from the heat, add the egg, allow it to sit in the hot water for 10 minutes, drain, and pour cold water over the egg. 5. Use these two eggs in part 2 below then peel each egg, being careful to note which egg is which. Cut the eggs in half and make the observations for the chart. Part 2 - Distinguishing between raw and cooked eggs You will need raw and cooked eggs. 1. Use the two cooked eggs from part one of this experiment (before you peel them!). 2. Place each cooked egg on the table and spin them one at a time, making observations for the chart. 3. Place the uncooked egg on the table and spin it, making observations for the chart. Scientific Principle _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Conclusions _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ 11 _______________________________________________________________________________________ _________________________________________________________________________________________ __ Name:_________________________ _____ Block:________ Date:__________________ Experiment: Cooking Eggs Observation Chart Part 1: Cooking eggs Cooking Procedure Appearance of yolk Appearance of white Sit 20 minutes off heat after boiling Cook 10 minutes then sit 10 minutes Part 2 - Distinguishing between raw and cooked eggs Egg type Sit 20 minutes off heat after boiling (hard-cooked) Cook 10 minutes then sit 10 minutes (hard boiled) Raw Spin factor 12 Experiment: Eggs TEACHER NOTES Scientific Principles -raw mass wobbles because it is not of uniform density -cooked mass is solid and of uniform density so it spins uniformly -over-cooked protein has been de-natured and becomes tough -properly cooled egg forces the sulfur to diffuse evenly towards the center of the yolk, while the improperly cooled egg allows the sulfur to slowly diffuse towards the white, therefore causing a grey ring Conclusions There appear to be slight differences in the appearance and spin factor of hard-boiled and hard cooked eggs, and a significant difference between cooked and raw eggs. The hard cooked egg has more tender protein than the hard-boiled egg. The density of the hard cooked egg is uniform and will therefore spin uniformly, while the density of the uncooked yolk and white is unequal, causing it to wobble as it spins. Experiment: Cooking Eggs Observation Chart Part 1: Cooking eggs Cooking Procedure Appearance of yolk Appearance of white Sit 20 minutes off heat after boiling -pale yellow -darker yolk than compared to other egg -no grey tinge -firm but not rubbery Cook 10 minutes then sit 10 minutes -pale yellow -greyish tinge around yolk -drier than other cooked egg -more firm and rubbery than other egg -greyer egg white around yolk Part 2 - Distinguishing between raw and cooked eggs Egg type Spin factor Sit 20 minutes off heat after boiling -spins better -more wobbly Cook 10 minutes then sit 10 minutes -spins longer -less wobbly Raw 13 -slow -off centre Background questions: 1. What is the difference between hard boiled and hard cooked eggs? 2. What is the name of the gas that causes the discolouration of the yolk? 3. What does the heat do (other than cooking the egg) to the egg? 4. What happens to the egg (chemically) when cooling a cooked egg under cold water? Discussion questions: 1. Will the eggs spinning vary? If so, how? 2. Will the consistency of the yolk be the same in the cooked eggs? How? 3. Will the consistency of the egg white be the same in the cooked eggs? How? 4. Describe the differences between the cooked and the uncooked eggs. (i.e. texture, colour, smell) 5. Name 3 products that you can make with each of these egg types? Reference: www.uen.org/Lessonplan/preview.cg?LPid=1179 This site has a wealth of information on eggs including other egg experiments. 14 Food Studies 12 Name:_________________________ _____ Source: M. Gale Smith Block:________ Date:__________________ EGG EXPERIMENT 1 Coagulation of protein Choose identical heavy-bottomed frying pans-small size. Prepare the following recipe for scrambled eggs: 3 eggs 50 mL water 3 mL salt 1 mL pepper Beat lightly with a fork until eggs are broken up and blended with liquid. This beating will begin the denaturation of the eggs. Divide the mixture into two equal portions. Place 20 mL of margarine in each frying pan and heat until melted and hot. Pour one portion of the egg mixture into each frying pan. Record the time. Reduce the heat and allow to cook by lifting the bottom and allowing the uncooked egg mixture to flow under it (you may also stir it.) Cook one mixture for 5 – 8 minutes just until the moister disappears. This is the sample of coagulated protein. EGG EXPERIMENT 2 Hard-boiled versus hard-cooked eggs Place two eggs in their shells in two saucepans (one in each saucepan) containing enough cold water to cover the eggs by 5 cm. Cover the saucepans and bring the water to a boil in both saucepans. (a) Remove one saucepan from the heat. Do not drain the water or take the cover off the saucepan. Allow the eggs to sit in the hot water for 20 minutes. At the end of the 20 minutes, drain off the water and pour cold water over the egg. (b) For the second egg, allow the water to boil rapidly for 10 minutes. Remove from the heat, allow to sit in the hot water for 10 minutes, drain, and pour cold water over the egg. Peel each egg, being careful to note which egg is which. Cut the eggs in half and record your observations on your record sheet. EGG EXPERIMENT 3 Volume and stability of beaten egg whites Prepare 5 whites in the following manner: 15 (a) Beat 1 egg white in a small bowl to stiff peak stage. Place in a pyrex measuring cup for display. (b) Beat 1 egg white in a small bowl to foamy stage. Add 15 mL water and beat to stiff peak stage. Display. (c) Beat 1 egg white in a small bowl to foamy stage. Gradually sprinkle in 30 mL sugar and continue beating to stiff peak stage. Display. (d) Beat 1 egg white in a small bowl to foamy stage. Add 15 mL cream of tartar and beat to stiff peak stage. Display. (e) Beat 1 egg white in a small bowl to foamy stage. Add 15 mL egg yolk and beat to the stiff peak stage. Display. Record your observation on your record sheet. EGG EXPERIMENT 4 Poach 3 eggs in the following manner: (a) Break one egg into a pan of simmering water. Cook for 3 minutes. Remove from water with a slotted spoon and place on a small plate for display. (b) Break one egg into a pan of boiling water. Cook for 3 minutes. Remove from water with a slotted spoon and place on a small plate for display. (c) Break one egg into a pan of simmering water which has had 15 mL vinegar added. Cook for 3 minutes. Remove from water with a slotted spoon and place on a small plate for display. Record your observation on your record sheet. EGG EXPERIMENT 5 Distinguish between raw and cooked eggs. You will need two eggs. (a) Set one egg aside. Place the other one in a saucepan, cover with water and bring to a boil. Turn heat off and let sit 20 minutes. Cool by immersing in cold water. Remove from water and dry. (b) Place the cooked egg on the table and spin it. (c) Place the uncooked egg on the table and spin it. Record your observation on your record sheet. EGG EXPERIMENT 6 Exploring the insulating power of egg white foam. You will need: 3 egg whites 125 mL sugar graham wafers 250 mL ice cream baking sheet aluminum foil Heat oven to 500 ‘F. Cover baking sheet with aluminum foil. Beat egg whites until they form soft peaks. Add sugar to beaten egg whites, 15 mL at a time, beating egg whites after each addition. Continue beating the egg whites and sugar until the mixture is thick and glossy looking (meringue) Put the graham wafer on the baking sheet. Place some frozen ice cream on the top of the wafer, so that it fits without hanging over the edge of the water. (g) Spread meringue thickly all over the ice cream like icing on a cake, covering it completely. Make sure there are no exposed spots. (h) Bake on the lowest rack of the oven for 3 to 5 minutes or until the meringue is light brown. (i) Take out of the oven and cut in half for display. (a) (b) (c) (d) (e) (f) 16 Food Studies 12 EGG EXPERIMENTS – SUTDENT RECORD SHEET 1. Coagulated vs. over coagulated protein Record your observations of the two samples of scrambled eggs on the following chart: Appearance including presence of water Texture Tenderness/ toughness Flavour Coagulated protein Overcoagulated protein Conclusions: How does the amount of heat affect the quality of cooked protein products? Give a general rule that could be used for protein cookery. 2. Hard cooked vs. hard boiled eggs Colour of yolk Hard cooked egg Hard boiled egg Conclusion: Texture of yolk Texture of white 17 What is the difference between a hard-boiled and a hard-cooked egg? Explain the effect of heat on proteins. 3. Volume and stability of beaten egg whites Stability Volume Egg white Egg white + water Egg white + sugar Egg white + cream of tartar Egg white + yolk Conclusion: Which substances stabilize an egg white foam? Why? Which substance prevents egg whites from foaming? Why? 4. Poaching Methods: Appearance and spread of white Simmered Texture of egg 18 Boiled Simmered with vinegar Which of the three methods produces the better product? Why? What is the effect of vinegar on poached eggs? 5. Distinguishing fresh from hard-cooked eggs Describe the spin of the raw egg: Describe the spin of the cooked egg: Conclusion: A hard cooked egg is and spins as a . Therefore it spins . A raw egg, the shell has to start to move the inside. This uses more so the egg spins slowly. 6. The insulating power of egg white foam Did the ice cream melt in the 500 ‘F oven? Conclusion: Beating the egg traps many small spaces in the meringue. These act as so that the heat can’t get in during the short time it is the oven. 19 Name:_________________________ Block:________ Date:__________________ Experiment: Visible Fats source: unknown Ingredients/Supplies Chocolate chips (semi-sweet) Sunflower seeds Potato chips Microwave Paper towels Foil Rolling pin Scale Procedure Part 1 – Chocolate Chips 1. 2. 3. 4. Measure out 2 grams of chocolate chips and place on a paper towel. Microwave for 40 seconds on high Fold the paper towel over the chocolate chips and gently press the chocolate chips flat with your fingers. Allow it to sit for 5 minutes. Open up the paper towel. Record your results. Part 2 – Potato Chips 1. Measure out 2 grams of potato chips and place on a paper towel. 2. Microwave for 25 seconds on high 3. Fold the paper towel over the potato chips and crush the chips with a rolling pin. Use equal, constant pressure. 4. Allow it to sit for 5 minutes. Open up the paper towel. Record your results. Part 3 – Sunflower Seeds 1. Measure out 2 grams of sunflower seeds and place on a paper towel. 2. Microwave for 25 seconds on high 3. Fold the paper towel over the sunflower seeds and crush the seeds with a rolling pin. Use equal, constant pressure. 4. Allow it to sit for 5 minutes. Open up the paper towel. Record your results. 20 Scientific Principle _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Conclusions _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ 21 Name:_________________________ Block:________ Date:__________________ Experiment: Visible Fats Food Item Chocolate chips Potato chips Roasted sunflower seeds Observation Chart Before heating Immediately after heating 5 minutes after heating 22 Experiment: Visible Fats TEACHER NOTES Background information : Equipment- review how to use the micro-wave - review or demonstrate how to use the scale (weighing food) Appropriate food to use : - dry, moisture free foods - variations of “chips” and “seeds” (different types of potato & chocolate chips) - various methods of processing (baked, fried etc.) Observation descriptors - concentration - lightly, moderately, heavily soaked - size of stain Questions : 1. What foods contain fat? 2. How can you determine if there is fat in a food? Discussion : Observing fat and moisture in various food they have eaten or been served(i.e. chocolate chip cookies, fish & chips etc. Heat releases fat in food yet in some cases heat puts fat into the food. (i.e. in deep frying fat replaces the moisture) Observation Chart Food Item Before heating Immediately after heating 5 minutes after heating and pounding Chocolate chips -no stain -small, moist stain Potato chips -no stain -very small Roasted sunflower seeds -no stain -very small -stain was larger and dark brown -the size did not increase after 5 minutes -approx. 6 cm -largest of the 3 food items -clear -approx. 10 cm -the size increased after the 5 minutes was up -approx. 8 cm -the size did not increase after 5 minutes 23 Name:_________________________ Block:________ Date:__________________ Experiment: Invisible Fats Equipment/Supplies 4 - 8 foods Paper (e.g. paper towel, brown bag, writing paper) Procedure 1. Cut 2-inch squares from a brown paper grocery bag, paper towel or writing paper. 2. Add a drop of cooking oil to the centre of one square and rub the oil in with your finger. Allow to dry. 3. Add a drop of water to the centre of another square and rub it in with your finger. Allow to dry. 4. Hold each square to a light source. Observe which square is translucent (allows some light to pass through) and which square is opaque (does not allow light to pass through). 5. Repeat the test using available food samples (salad dressing, mayonnaise, ketchup, mustard, honey, peanut butter, cheese, etc.) 6. Let the paper dry for about a half hour then make your observations Scientific Principle _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Conclusions __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ 24 Invisible Fats Observation Chart Food item Oil Water Black licorice Peanut butter Mustard Ketchup Mayonnaise Parmesan cheese Avocado Marble cheese Cracker Cookie Ice cream Nesquick Translucent Opaque Pre-observation guess: fat or no fat? Observation fat or no fat 25 Invisible Fats Observation Chart Background information: TEACHER NOTES -Knowledge about oils and water; solubility on various surfaces/materials -Labelling information; terminology i.e. “partially-hydrogenated” -Tran fats/ saturated/ polyunsaturated fatty acids (PUFA) types use in prolonging shelf life. Questions: 1. How many of you have had a croissant or potato chips that have left a stain on a napkin &/or your clothes? What part of the food left a stain? 2. What foods here might leave a stain? 3. What foods here might contain fats? 4. Which products did not leave a stain on the paper bag or paper towel? 5. What conclusions can be drawn from the products that left or did not leave a mark? 6. What does this exercise tell you about the fat content of food products? 7. What is another way of finding out the fat content of a particular food? 8. Are all fats equal? How are they hidden in food? Food item Translucent Oil Water Black licorice Peanut butter Mustard Ketchup Mayonnaise Parmesan cheese Avocado Marble cheese Cracker Cookie Ice cream Nesquick yes Opaque yes yes yes yes yes yes yes yes yes yes yes yes yes reference: http://agexted.cas.psu.edu/FCS/4hfl/Fat.html Pre-observation guess: fat or no fat? Observation fat or no fat 26 Name:_________________________ Block:________ Date:__________________ source: unknown Experiment: Meat packaging Equipment/Supplies 1 tightly packed package of fresh ground beef cutting board knife white plate Saran® wrap and Handiwrap® plastic wrap refrigerator optional – rubber/latex gloves for handling meat Procedure Place the ground beef on a cutting board and slice across the length in the centre of the package. Make another cut about 1 inch (2.5 cm.) from the first to form a hamburger patty. Place the patty on a white plate and observe the colour near the edge of the patty and near the centre. Cut off two more 1-inch thick patties. Wrap one patty using Saran® wrap which is oxygen impermeable. Use oxygen permeable Handiwrap® to wrap the second patty. Place each of the patties on a white plate. Refrigerate the patties and observe colour changes over the next 1 to 2 hours. Scientific Principle _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Conclusions ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ______________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ __________ 27 Name:_________________________ Block:________ Date:__________________ Experiment: Packaging Type of packaging Saran® wrap (or other oxygen impermeabl e) Handiwrap® (or other oxygen permeable) Observation Chart Patty before wrapping Patty after wrapping Patty after 1 hour Patty after 2 hours 28 Experiment: Meat packaging TEACHER NOTES Observation Chart Type of packaging Saran® wrap (or other oxygen impermeabl e) Handiwrap® (or other oxygen permeable) Patty before wrapping Patty after wrapping Patty after 1 hour Patty after 2 hours Bright red Bright red Bright red Bright red, but goes brown with more time Bright red Purple “Yucky” greyish brown Brown Teacher information Background Information: 1. muscle meat (i.e. ground beef) myoglobin – when exposed to oxygen it goes brownmeta. myoglobin 2. plastic wrap -oxygen permeable (Handiwrap® ) -allows oxygen in -stays red for a time -oxygen non-permeable (Saran® wrap) -keeps oxygen away -colour stays brown 3. cooking temperature: If you start with red meat it goes RED to PINK to BROWN as liquid and gat are removed. If you start with brown meat it looks done at 131 F. so you have to cook it well – (firm and elastic, liquid and fat removed) 29 References: Kitchen Science by Howard Hillman 1989 http://www.fsis.usda.gov/oa/pubs/colortech.htm Related information on packaging and bone colour in meat: www.oznet.ksu.edu/news/sty/2004/beef_products121504 . Name:_________________________ Block:________ Date:__________________ Experiment: McMush Materials/Supplies 1 quarter pound hamburger meal (with medium fries 1 blender 1 hot plate 1 - 500 ml beaker 1 - 200 ml graduated cylinder 100 ml of water 2 oven mitts 1 wooden spoon 1 refrigerator to cool McMush mixture Procedure Overview 1. The complete meal will be blended. 2. A large sample of the meal will be heated. 3. From the heated sample, 100 ml will be taken to be cooled . 4. Results from the 100 ml sample are representational of the complete meal. Procedure 1. Preheat the hot plate. 2. Break up meal into small pieces and blend. 3. Pour part of the blended McMush into a 500 ml beaker. 4. Add 100 ml of water to the McMush and stir well. 5. Boil McMush mixture gently for 15 minutes. 6. Use oven mitts to protect your hands and pour the hot McMush mixture into a graduated cylinder. Then cool in the refrigerator for 5 minutes. 7. Remove McMush from the refrigerator and measure the amount of accumulated fat at the top of the graduated cylinder. 8. Record results. 9. Clean glassware with warm soapy water. 30 Retrieved August 16, 2005 from http://student.biology.arizona.edu/sciconn/mcmush.html Scientific Principle _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Conclusions __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ Experiment: McMush Concept: TEACHER NOTES Fat is less dense than other components of food. After homogenizing a food (i.e., blending it up to make a uniform "mush") and letting it separate out, the fat, being less dense, will rise to the top. When you do this in a graduated cylinder, you can determine how much fat was in the food. Notes: The fat, being less dense, forms a layer at the top and will solidify as it cools. You can calculate the percent fat in the meal by dividing the total mL of fat by the total mL of your sample. For example, if you find 40 mL of fat in a 100 mL sample, this would indicate that the total meal contained 40% fat. It is important to note that this is 40% volume/volume, not 40% of the total calories in the meal Nutrition recommendations for the macronutrients (fat, carbohydrates, protein) are often given as a recommendation for the % of total calories that should come from each macronutrient -- e.g., the current Acceptable Macronutrient Distribution Range (AMDR) (which 31 is the recommendation from the Dietary Reference Intake, the official nutrient recommendations for Canada and the US) for fat is that between 20-35% of energy intake. Saying that the meal is 40% volume/volume is not the same thing as 40% of energy intake To calculate the % of total energy coming from fat, you would need to know: o the total energy intake (try using our Measuring Calories activity!) and o you would need to convert the volume of fat (in mL) to calories: fat contains 9 Calories per gram students could weigh a sample of the fat to figure out the density (in order to convert mL to grams) Extension: Have several different foods available and get the students to compare the amount of fat in the different foods. Have students develop a hypothesis about which food will have the most fat and which will have the least before they start. Review the hypothesis at the end of the experiment -- was the hypothesis correct? * Note: When blending the meal together, be sure not to blend too much or a smooth emulsion results and the fat does not rise to the top. Name:_________________________ Block:________ Date:__________________ source: unknown Experiment: Enzyme Break-Down of Protein Equipment/Supplies 1 package of light coloured gelatin dessert powder (e.g.Jello®) 10 ml each of fresh pineapple and kiwi juice 3 dark coloured marbles 3 - 100 mL graduated cylinders Procedure 1. Prepare the gelatin dessert according to the package directions and fill 3 100 mL cylinders to the 90 mL level 2. Place in a refrigerator. After the gel is formed, add 10 mL of fresh pineapple, to one cylinder and 10 ml and 10 ml kiwi juice to another cylinder. Label each cylinder. 3. Place a marble on the gelatin in each cylinder and observe over time. 32 4. Read the preparation instructions for a package of gelatin desert. Comment on the precaution statement: To add fruit or vegetables: Refrigerate gelatin 1 ½ hours or until thickened. Stir 1 ½ to 3 cups chopped fruit or vegetables (well-drained, if using canned). Refrigerate 4 hours or until firm. Note: DO Not use fresh or frozen pineapple, kiwi, gingerroot, papaya, figs or guava. Gelatin will not set. Scientific Principle _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Conclusions _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _____________________ Observation Chart Drop in mm Time 30 seconds 1 minute 1 ½ minutes 2 minutes 2 ½ minutes Control Kiwi Pineapple 33 etc. Experiment: Enzyme Break-Down of Protein TEACHER NOTES Scientific Principle Enzymes Breaking Down Protein Background information: - gelatin – composed of protein (isolecine, threonine and methionine) - animal proteins – a variety of methods are used for tenderizing: - aging -pounding - enzymes - proteolytic i.e. pineapple – bromelio, figs – ficin, papaya – papain) -tenderizing helps denature the elastin connective tissue in meat Questions: 1. What happened to the gelatin in the 3 cylinders? 2. Why did the gelatin in some cylinders breakdown? 3. What is in the fruit juices that cause this reaction to occur? 4. What are the roles of enzymes in food and during cooking? 5. How does this information relate to cooking? 34 Conclusions Pineapple and kiwi juice cause the gelatin to break down and the solid changes back to a liquid. Observation Chart TEACHER NOTES Drop in mm Time Control Kiwi Pineapple 30 seconds The marble took the longest to get to the bottom of the cylinder.* The marble took less time to get to the bottom of the cylinder.* The marble took less time to get to the bottom of the cylinder.* 1 minute 1 ½ minutes 2 minutes 2 ½ minutes etc. * At the Summer Institute we did not have enough time to let the gelatin set. Be sure to set the gelatin well in advance when conducting this experiment. Name:________________________ Block:________ Date:__________________ source:unknown Experiment: Chameleon in the Kitchen Equipment/Supplies Shake #1 125 mL frozen blueberries 125 mL orange juice 10 mL honey (optional) 5 mL lemon juice In blender, combine blueberries, orange juice, honey and lemon juice; blend at high speeduntil smooth. Makes 250 mL. Shake #2 125 mL frozen blueberries 50 mL club soda 1 large scoop vanilla ice cream (or substitute yogurt) 10 mL granulated sugar 35 2 mL vanilla In blender, combine blueberries, club soda, ice cream or yogurt, sugar and vanilla; blend at high speed until smooth. Makes 250 mL. Scientific Principle _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Conclusions _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _____________________ Experiment: Chameleon in the Kitchen TEACHER NOTES Background information: - what effect does an acid/base have on the colour of an ingredient? - what are anthocyanins? A purple pigment found in foods like red cabbage/berries/plums. It is an “indicator” (colour changes when in the presence of an acid or base) Questions Predict what colour change will occur when an acid/base is added to an anthocyanin ? Note: This experiment did not perform as well as expected. The suggestion was made that the red cabbage and acid combination would work better. related experiment: http://agexted.cas.psu.edu/FCS/4hfl/Color.html 36 technical background: http://lpi.oregonstate.edu/ssO1/anthocyanin.html 37 Name_______________________________ Block:________ Date:__________________ Experiment: How to Get Iron Out of Breakfast Cereal Equipment/Supplies 2-3 cups fortified cereal Magnet Bowl Spoon or other utensil Water Blender (optional) Napkin Procedure 1. Pour the cereal into the bowl or blender. 2. Add sufficient water to completely cover the cereal. (It’s not an exact measurement – you can add as much as you want as iron doesn’t dissolve in water) 3. Mash the cereal with a spoon or mix it with the water using a blender. The more finely ground the cereal is, the easier it will be to get the iron. 4. Stir the magnet through the crushed cereal. Iron is heavy and will sink, so be sure to pay attention to the bottom of the bowl. If you used a blender, make sure you can get to the particles at the bottom of the jar. 5. Look for the black “fuzz” or iron on the magnet. It’s easiest to see the iron if you wipe the iron on a white napkin or paper towel. Scientific Principle _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Conclusions _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _______________________________________________________________________________ _____________________ 38 Experiment: How to Get Iron Out of Breakfast Cereal TEACHER NOTES Background Information: Cereals are traditionally fortified with iron. Iron is found in the Bran of the cereal kernel. Parts of a cereal kernel. Knowledge about the processing involved in fortify a cereal. Read cereal packages for iron content Show how to use a magnet Questions: 1. Do cereals contain iron? 2. Where does the iron come from in cereals? 3. Why is iron important for the human body? 4. Is there away of “seeing” the iron in a cereal besides reading the label? Follow-up experiment Have students compare a) a heated solution b) a cold solution c) a soaked solution with a variety of cereals (both whole grain and fortified) to see which is a better source of iron. Can a person increase the absorption level of the iron in a cereal? Note: research shows that in a heated solution the iron adheres better. (see cited website) Reference: http://chemistry.about.com/cs/howtos/ht/ironfromcereal.htm 39 Source: Eda Favaro Name: ______________________ Block: ____ Date: ____________ Experiment: Yeast Ingredients/Supplies 3 pyrex custard cups 1 candy thermometer 15 mL yeast water 1 cake pan 1 mL salt 8 mL sugar Procedure (pre-heat oven) 1. 2. 3. 4. 5. 6. Label each cup with masking tape #1, #2, & #3. Measure 5 mL of yeast into each custard cup. Add 3 mL sugar to each of the 3 custard cups. To custard cup #1 add 1 mL salt To custard cup #1 and #2 – add 50 mL water (45-50 degrees) to each To custard cup #3 add 125 mL boiling water . Place this custard cup in the cake pan and pour boiling water into the pan. Place the pan and contents into the oven at 350 degrees for 10 minutes. 7. Record observations Scientific Principle __________________________________________________________________________ __________________________________________________________________________ __________________________________________________________________________ __________________________________________________________________________ __________________________________________________________________________ __________________________________________________________________________ Conclusion __________________________________________________________________________ __________________________________________________________________________ __________________________________________________________________________ __________________________________________________________________________ __________________________________________________________________________ __________________________________________________________________________ __________________________________________________________________________ __________________________________________________________________________ 40 ________________________________________________________________________ __________________________________________________________________________ __ Name: ______________________ Block: ____ Date: ____________ Experiment: Yeast Observations Custard cup #1 Custard cup #2 Custard cup #3 Questions 1. How does heat affect yeast? _____________________________________________ 2. How does salt affect yeast? ______________________________________________ 3. How does sugar affect yeast? _____________________________________________ 4. Explain the chemical reaction that takes place when yeast ferments._______________ ________________________________________________________________________ 5. Explain how yeast acts as a leavener. _________________________________________ __________________________________________________________________________ 41 Experiment: Yeast TEACHER NOTES Observations Custard cup #1 -little or no action as salt retards yeast growth Custard cup #2 -bubbles and puffs up nicely because it has food for growth and the required warm temperature Custard cup #3 -does not react because the high temperature has killed the yeast References: Management and Foods p. 296 genome~www.stanford.edu/VL-yeast.html www.Fleischmann's.yeast Related experiment "Yeast Breath" at www.kidwizard.com/Spells/Yeast 42 Source: Eda Favaro Name: ______________________________ Block: _________ Date: _______________ Experiment: Gluten Ingredients/Supplies 125 mL flour 30 mL water Procedure 1. Place 125 mL of flour into a small bowl. 2. Slowly add approximately 30 mL of water, stirring with a fork until a stiff, but sticky dough is formed. 3. Knead dough for 5 minutes. 4. Note the stiffness and elastic qualities of the dough. 5. Place dough in a sieve in a bowl of cold water and allow to sit for 10-20 minutes. 6. Wash away the starch from the dough by kneading it in a sink under cold water. Work it until the water runs clear. 7. Squeeze the water out of the wet GLUTEN. Shape it into a ball. Place the ball in the appropriate space on the supply table. Note the relative sizes of the gluten balls. Scientific Principle _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Conclusion _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ 43 _______________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ __ Observations Flour Appearance of dry flour Relative size of wet gluten Relative size of baked gluten Colour & structure of gluten 44 Experiment: Gluten TEACHER NOTES Why is gluten so important? Without it, there would be nothing to hold the gas that makes bread rise. Think of gluten as the rubber of a balloon: The stronger it is, the more gas it can hold. But stronger isn’t always better. For many baked goods, like pastries and pie crusts, it’s important to avoid gluten development. That’s why different flours contain different amounts of protein, depending on how they are meant to be used. A high-protein flour will make a dough with strong gluten, good for hearty yeast breads. Pastry chefs, on the other hand, prefer low-protein flours that yield delicate, tender doughs. The gluten experiment outlined on the previous page is explained, with accompanying pictures, at www.exploratorium.edu/cookingbread/activity-gluten.htm/ Another gluten-related experiment: The reason why bread bakes up so airy is because the bread dough captures and holds the carbon dioxide that the yeast produces. It does this because flour contains a protein called gluten. To see gluten in action, try this experiment: 1. 2. 3. 4. Mix 1/2 cup water and 1/2 cup flour in a bowl. Stir the mixture with a fork to wet the flour. What you will have initially is a lumpy, grainy mass. Lift the fork out of this mass. You will find that the mass is quite watery. Now keep stirring for about five minutes (set a timer for five minutes -- it is a long time when you are stirring!). Over time, the batter will smooth out. 5. Keep stirring, and a funny thing will happen when you lift the fork slowly from the bowl: The batter will have become quite elastic! Not elastic like a rubber band, but elastic enough that you'll be able to pull away up to a 1-inch-long thread of batter with the fork. This mixture is now extremely smooth and not watery at all. 45 That elasticity is caused by the gluten in the flour. Gluten is a protein that forms thread-like chains. By stirring (or more commonly, kneading) the dough, the gluten develops into long, interlaced chains. Kneading is better for developing these chains because kneading is gentle -- it does not cut the chains up. When you knead bread dough, you are creating gluten chains. If you were to skip the kneading part, your bread would not rise very well -- all the carbon dioxide in the yeast would bubble up to the top and escape, rather than being captured inside the elastic dough. Source: http://science.howstuffworks.com/bread4.htm Name:_________________________ Block:________ Date:__________________ source: unknown Experiment: Baking is a Gas! Equipment/Supplies small and liquid measures funnel baking soda baking powder lemon juice balloon plastic bottle cool and warm water custard cups or glasses Procedure Baking soda: 1. Mix 10 mL baking soda with 125 mL water. Watch carefully. 2. Now add 15 mL lemon juice. Baking powder: 1. Mix 15 mL baking powder with 125 mL cool water and listen. 2. Using a funnel, put 25 mL baking powder into a balloon. 3. Then put 250 mL warm water into a pop bottle. 4. Fit the balloon over the neck of the bottle and let the baking powder fall into the water. Shake the bottle and watch the balloon grow. Scientific Principle _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ 46 _______________________________________________________________________________________ _________________________________________________________________________________________ __ Conclusions ________________________________________________________________________________ ________________________________________________________________________________ ________________________________________________________________________________ ________________________________________________________________________________ ________________________________________________________________________________ ________________________________________________________________________________ ________________________________________________________________________________ _______________________________________________________________ Name:_________________________ Block:________ Date:__________________ Experiment: Baking is a Gas! Ingredient Baking soda and water Lemon juice added to baking soda and water Baking powder and cool water Baking powder and warm water Observation chart Results 47 Experiment: Baking is a Gas! TEACHER NOTES Scientific Principle Background Information Baking powder: mixture of baking soda and acids or acid salts that are separated by an inert filler. Inert filler: usually cornstarch or powdered calcium carbonate acts as a buffer between active ingredients and prevents their going into action when exposed to moisture. Baking soda: one of the earliest chemical leavening agents used was sodium bicarbonate. Leaveners make a product rise. (air,steam etc.) NaHCO3 + Baking soda HKC4H4O6 → Cream of tartar 2NaSO4Al2(SO4)3 NaKC4H4O6 + Potassium acid tartrate + 6H2O H2O + CO2 water → NA2SO4 carbon dioxide + 2Al(OH)3 + H2SO4 heat Sodium aluminium Sulfate water sodium sulfate Observation chart Ingredient Baking soda and water Lemon juice added to baking soda and water Results -some dissolved -a bit floated on top -overall nothing -bubbled up aluminium hydroxide sulphuric acid 48 Baking powder and cool water -bubbled and dissolved -made popping sounds Baking powder and warm water -balloon slowly expands -if you keep shaking it keeps expanding Background Information Baking powder: mixture of baking soda and acids or acid salts that are separated by an inert filler. Inert filler: usually cornstarch or powdered calcium carbonate acts as a buffer between active ingredients and prevents their going into action when exposed to moisture. Baking soda: one of the earliest chemical leavening agents used was sodium bicarbonate. Leaveners make a product rise. (air,steam etc.) Questions: 1. What is baking powder and baking soda? 2. What are leaveners and what do they do ina recipe? 3. What chemical reaction is occurring? 4. What was the gas released? 5. Why did it bubble? 6. What about the lemon juice caused it to bubble? 7. What will this do in a recipe? 8. Can you interchange baking soda and baking powder in a recipe? Source: C. Thomson Name: ____________________ Block: ____ Date: ___________ Experiment: Mixing Methods Introduction This experiment is intended as a review of the mixing methods for muffins, biscuits and cakes. Students work in groups of four to prepare a product from the list of given ingredients. They must figure out the mixing method from the ingredients and prepare the product. The questions should be answered first before they proceed with preparation. The final products are evaluated. Ingredients/Supplies Station 1 Station 2 Station 3 Station 4 Station 5 250 mL flour 7 mL baking powder 1 mL salt (optional) 45 mL cocoa 60 mL margarine(room temp.) 125 mL sugar 1 egg 7 mL vanilla 80 mL milk 250 mL flour 7 mL baking powder 2 mL salt (optional) 60 mL margarine (cold) 50 mL raisins 90 mL milk 250 mL flour 10 mL baking powder 50 mL sugar 1 small egg (beaten) 125 mL milk 30 mL oil 125 mL grated cheese 250 mL flour 7 mL baking powder 60 mL margarine (room temp.) 125 mL sugar 1 egg 7 mL vanilla 80 mL milk 25 mL raisins 25 mL walnuts 250 mL flour 10 mL baking powder 50 mL sugar 1 egg beaten 125 mL milk 30 mL oil 125 mL chocolate chips Procedure 1. Divide unit into four tasks: * head chef * food supervisor (assists chef) 49 * food manager(consults with the rest of the group about lab sheet & assists recorder) * recorder (records units answers on the lab sheet & hands in completed lab sheet) 2. Groups read through the ingredients and answer the questions. 3. Groups prepare the flour mixture. 4. Clean up while product is baking. 5. Set out product for evaluation and hand in lab sheet. Scientific Principle How different forms of fat and different ingredients are best mixed and prepared in a certain way for best results. Questions 1. What type of fat is used in this recipe? How is it added? 2. Is there an egg in this recipe? 3. Does this recipe follow the cake method, muffin method or biscuit method when mixing the ingredients? 4. Explain the steps you will use to mix your product? 5. What equipment is used to mix? 6. What exactly is your finished product? 7. Do you use a greased or ungreased pan ? Rate your unit on the success of your product from 1 bad/ 2 O.K/ 3 good/ 4 very good/ 5 excellent Names: ____________________________ ____________________________ Block: ______ Date: _________________ Experiment: Flour Mixtures Ingredients/Supplies Station 1 250 mL flour 7 mL baking powder 1 mL salt (optional) 45 mL cocoa 60 mL margarine(room temp.) 125 mL sugar 1 egg 7 mL vanilla 80 mL milk Procedure 1. Divide unit into four tasks: * head chef * food supervisor (assists chef) * food manager(consults with the rest of the group about lab sheet & assists recorder) * recorder (records units answers on the lab sheet & hands in completed lab sheet) 2. Groups read through the ingredients and answer the questions. 3. Groups prepare the flour mixture. 4. Clean up while product is baking. 5. Set out product for evaluation and hand in lab sheet. Questions 50 1. What type of fat is used in this recipe? How is it added?_________________________________________________________________________________________ _________________________________________________________________ 2. Is there an egg in this recipe?_____________________ 3. Does this recipe follow the cake method, muffin method or biscuit method when mixing the ingredients?________________________________________________________ 4. Explain the steps you will use to mix your product? _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________ 5. What equipment is used to mix?__________________________________________________ 6. What exactly is your finished product?_____________________________________________ 7. Do you use a greased or ungreased pan ?_________________________________________ Rate your unit on the success of your product from 1 bad/ 2 O.K/ 3 good/ 4 very good/ 5 excellent _________________________________________________________________________________________ _________________________________________________________________ Conclusion _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Names: ____________________________ ____________________________ Block: ______ Date: _________________ Experiment: Flour Mixtures Ingredients/Supplies Station 2 250 mL flour 7 mL baking powder 2 mL salt (optional) 60 mL margarine (cold) 50 mL raisins 90 mL milk Procedure 1. Divide unit into four tasks: * head chef * food supervisor (assists chef) * food manager(consults with the rest of the group about lab sheet & assists recorder) * recorder (records units answers on the lab sheet & hands in completed lab sheet) 2. Groups read through the ingredients and answer the questions. 3. Groups prepare the flour mixture. 4. Clean up while product is baking. 5. Set out product for evaluation and hand in lab sheet. Questions 51 1. What type of fat is used in this recipe? How is it added?_________________________________________________________________________________________ _________________________________________________________________ 2. Is there an egg in this recipe?_____________________ 3. Does this recipe follow the cake method, muffin method or biscuit method when mixing the ingredients?________________________________________________________ 4. Explain the steps you will use to mix your product? _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________ 5. What equipment is used to mix?__________________________________________________ 6. What exactly is your finished product?_____________________________________________ 7. Do you use a greased or ungreased pan ?_________________________________________ Rate your unit on the success of your product from 1 bad/ 2 O.K/ 3 good/ 4 very good/ 5 excellent _________________________________________________________________________________________ _________________________________________________________________ Conclusion _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Names: ____________________________ ____________________________ Block: ______ Date: _________________ Experiment: Flour Mixtures Ingredients/Supplies Station 3 250 mL flour 10 mL baking powder 50 mL sugar 1 small egg (beaten) 125 mL milk 30 mL oil 125 mL grated cheese Procedure 1. Divide unit into four tasks: * head chef * food supervisor (assists chef) * food manager(consults with the rest of the group about lab sheet & assists recorder) * recorder (records units answers on the lab sheet & hands in completed lab sheet) 2. Groups read through the ingredients and answer the questions. 3. Groups prepare the flour mixture. 4. Clean up while product is baking. 5. Set out product for evaluation and hand in lab sheet. 52 Questions 1. What type of fat is used in this recipe? How is it added?_________________________________________________________________________________________ _________________________________________________________________ 2. Is there an egg in this recipe?_____________________ 3. Does this recipe follow the cake method, muffin method or biscuit method when mixing the ingredients?________________________________________________________ 4. Explain the steps you will use to mix your product? _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________ 5. What equipment is used to mix?__________________________________________________ 6. What exactly is your finished product?_____________________________________________ 7. Do you use a greased or ungreased pan ?_________________________________________ Rate your unit on the success of your product from 1 bad/ 2 O.K/ 3 good/ 4 very good/ 5 excellent _________________________________________________________________________________________ _________________________________________________________________ Conclusion _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Names: ____________________________ ____________________________ Block: ______ Date: _________________ Experiment: Flour Mixtures Ingredients/Supplies Station 4 250 mL flour 7 mL baking powder 60 mL margarine (room temp.) 125 mL sugar 1 egg 7 mL vanilla 80 mL milk 25 mL raisins 25 mL walnuts Procedure 1. Divide unit into four tasks: * head chef * food supervisor (assists chef) * food manager(consults with the rest of the group about lab sheet & assists recorder) * recorder (records units answers on the lab sheet & hands in completed lab sheet) 53 2. Groups read through the ingredients and answer the questions. 3. Groups prepare the flour mixture. 4. Clean up while product is baking. 5. Set out product for evaluation and hand in lab sheet. Questions 1. What type of fat is used in this recipe? How is it added?_________________________________________________________________________________________ _________________________________________________________________ 2. Is there an egg in this recipe?_____________________ 3. Does this recipe follow the cake method, muffin method or biscuit method when mixing the ingredients?________________________________________________________ 4. Explain the steps you will use to mix your product? _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________ 5. What equipment is used to mix?__________________________________________________ 6. What exactly is your finished product?_____________________________________________ 7. Do you use a greased or ungreased pan ?_________________________________________ Rate your unit on the success of your product from 1 bad/ 2 O.K/ 3 good/ 4 very good/ 5 excellent _________________________________________________________________________________________ _________________________________________________________________ Conclusion _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Names: ____________________________ ____________________________ Block: ______ Date: _________________ Experiment: Flour Mixtures Ingredients/Supplies Station 5 250 mL flour 10 mL baking powder 50 mL sugar 1 egg beaten 125 mL milk 30 mL oil 125 mL chocolate chips Procedure 1. Divide unit into four tasks: * head chef * food supervisor (assists chef) * food manager(consults with the rest of the group about lab sheet & assists recorder) * recorder (records units answers on the lab sheet & hands in completed lab sheet) 2. Groups read through the ingredients and answer the questions. 54 3. Groups prepare the flour mixture. 4. Clean up while product is baking. 5. Set out product for evaluation and hand in lab sheet. Questions 1. What type of fat is used in this recipe? How is it added?_________________________________________________________________________________________ _________________________________________________________________ 2. Is there an egg in this recipe?_____________________ 3. Does this recipe follow the cake method, muffin method or biscuit method when mixing the ingredients?________________________________________________________ 4. Explain the steps you will use to mix your product? _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________ 5. What equipment is used to mix?__________________________________________________ 6. What exactly is your finished product?_____________________________________________ 7. Do you use a greased or ungreased pan ?_________________________________________ Rate your unit on the success of your product from 1 bad/ 2 O.K/ 3 good/ 4 very good/ 5 excellent _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Conclusion _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ _________________________________________________________________________________________ Food and Nutrition 9/10 Experiment: Cooking and Storing Properties of Cheese Source: C. Thomson Procedure: This is an experimental lesson which encourages inductive reasoning. Students present findings and conclusions to rest of class. Group students into six groups, each group taking one of the following guide sheets for their experiment. Prepare cheese and supplies for students in advance. Experiments take approximately 10 minutes. Students can work in pairs and more than one group can do each experiment. This would allow for comparison of findings and conclusions. Conclude lesson with worksheet or oral questions following “Properties of (some) Cheeses.” Have students complete worksheet or summarize findings and conclusions in their notebook. 55 Food and Nutrition 9/10 Names ___________ Group #1 Cheddar cheese vs. processed cheese (melting properties) Follow the directions below and report your findings to the class. You have received two types of cheese. One type is processed cheese and the other one is cheddar cheese. Your mission is to determine what happens to each when they melt. Direction: You have two portions of each type of cheese. Use only one portion of cheddar cheese and one portion of processed cheese to carry out your experiment to determine what will happen. You will do this experiment only once so be accurate. The remaining two portions you will use to conduct a class demonstration with and teach the class what you discovered about your cheese. 56 Choose only two people from your group to do the class demonstration, making sure you explain the properties of your discovery. Procedure: Place one piece cheddar cheese in the microwave for 10 sec. Then place the processed cheese in the microwave for 10 sec. Questions to explain during your presentation: What happens to the processed cheese when it melts? What happens to the cheddar cheese when it melts? Do either of these cheeses separate into a liquid and solid mass? Food and Nutrition 9/10 Names Group #2 Refrigerated Cheddar cheese vs. previously frozen cheddar cheese (storage properties) Follow the directions below and report your findings to the class. You have received two types of cheese. One has been stored in the refrigerator and the other has been stored in the freezer. Your mission is to determine what happens to each when you try to slice these two products. 57 Direction: You have two portions of each type of cheese. Use only one slice of refrigerated cheddar cheese and one slice of preciously frozen cheddar cheese to carry out your experiment to determine what will happen. You will do this experiment only once so be accurate. The remaining two slices you will use to conduct a class demonstration with and teach the class what you discovered about your cheese. Choose only tow people from your group to do the class demonstration, making sure you explain the properties of your discovery. Procedure: Slice one piece of cheddar cheese off the refrigerated block and then one slice off the previously frozen block. Questions to explain during your presentation: What happens when slicing cheddar cheese that has been stored in the refrigerator and slicing cheddar cheese that has been previously frozen? Food and Nutrition 9/10 Names Group #3 Cheddar cheese vs. Cheddar cheese (cooking properties) Follow the directions below and report your findings to the class. You have received two types of cheese. One piece is to be just melted and the other is to be cooked longer. Your mission is to determine what happens to each when they are cooked for different lengths of time. Directions: 58 You have two portions of cheese. Use only two portions of cheddar cheese to carry out your experiment to determine what will happen. You will do this experiment only once so be accurate. The remaining two portions you will use to conduct a class demonstration with and teach the class what you discovered about your cheese. Choose only tow people from your group to do the class demonstration, making sure you explain the properties of your discovery. Procedure: Place one piece cheddar cheese in the microwave for 10 sec. Then place the other piece of cheese in the microwave for 15 sec. Questions to explain during your presentation: Using a toothpick, what is the consistency of each? If cheese is considered cooked when it is just melted, what happens to it when it is cooked too long? Food and Nutrition 9/10 Names Group #4 Cheddar cheese vs. Mozzarella cheese (melting properties) Follow the directions below and report your findings to the class. You have received two types of cheese. One piece is mozzarella cheese and the other one is cheddar cheese. Your mission is to determine what happens to each when they melt. 59 Directions: You have two portions of each type of cheese. Use only one portion of cheddar cheese and one portion of mozzarella cheese to carry out your experiment to determine what will happen. You will do this experiment only once so be accurate. The remaining two portions you will use to conduct a class demonstration with and teach the class what you discovered about your cheese. Choose only two people from your group to do the class demonstration, making sure you explain the properties of your discovery. Procedure: Place one piece cheddar cheese in the microwave for 10 sec. Then place the mozzarella cheese in the microwave for 10 sec. Questions to explain during your presentation: What are the two properties of the melted mozzarella cheese that are different from the melted cheddar cheese? Food and Nutrition 9/10 Names Group #5 Cheddar cheese vs. Parmesan cheese (melting properties) Follow the directions below and report your findings to the class. You have received two types of cheese. One type is parmesan cheese and the other one is cheddar cheese. Your mission is to determine what happens to each when they melt. 60 Directions: You have two portions of each type of cheese. Use only one portion of cheddar cheese and one portion of parmesan cheese to carry out your experiment to determine what will happen. You will do this experiment only once so be accurate. The remaining two portions you will use to conduct a class demonstration with and teach the class what you discovered about your cheese. Choose only two people from your group to do the class demonstration, making sure you explain the properties of your discovery. Procedure: Place one piece cheddar cheese in the microwave for 10 sec. Then place the parmesan cheese in the microwave for 10 sec, then 10 more sec. and for 10 more sec. Questions to explain during your presentation: What happens to the parmesan cheese when you try to melt it like cheddar cheese? Does it melt in this way? Why or why not? From this experiment, would you conclude that this is a soft cheese or a hard cheese? Food and Nutrition 9/10 Names Group #6 Cottage cheese vs. previously frozen cottage cheese (storing properties) Follow the directions below and report your findings to the class. You have received two types of cheese. One type is cottage cheese and the other one is previously 61 frozen cottage cheese. Your mission is to determine what happens to cottage cheese when it is been frozen. Directions: You have two portions of each type of cheese. Use only one portion of cottage cheese and one portion of previously frozen cottage cheese to carry out your experiment to determine what will happen. You will do this experiment only once so be accurate. The remaining two portions you will use to conduct a class demonstration with and teach the class what you discovered about your cheese. Choose only two people from your group to do the class demonstration, making sure you explain the properties of your discovery. Procedure: Compare your two examples of cottage cheese by both sight and taste test. Questions to explain during your presentation: Does the one previously frozen have more liquid then regular refrigerated cottage cheese? By tasting, is the texture the same for each or is one more granular then the other? Food and Nutrition 9/10 Name Properties of (some) Cheeses 1. What happens to cheddar cheese and processed cheese when each is melted? 62 2. What happens to previously frozen cheddar cheese when you try to slice it? 3. If cheese is considered cooked when it is just melted, what happens to cheddar cheese if it is cooked too long? 4. What are the two melted properties of mozzarella cheese that are different from cheddar cheese? 5. Why does parmesan cheese not melt like cheddar cheese? (two points) 6. Can you store cottage cheese in the freezer and end up with a product that is equally acceptable as the product stored in the refrigerator? Resources Books 1. Glencoe/McGraw-Hill Food for Today – Food Science Resources ISBN 0-07-820700-2 This resource booklet, which accompanies the Food for Today textbook, has 25 food science experiments. There is an excellent introduction that covers objectives of food science, essential skills, overcoming science phobias, scientific method and safety. 2. McGraw Hill Ryerson. Food and Nutrition Sciences Lab Manual ISBN 0-07-095238-8 This manual is similar to the one above and is intended for use with the Food Today textbook 63 Don’t be put off by the fact that these are textbook companions. They are great standalone resources. 3. Wolke, R. (2002). What Einstein Told His Cook: Kitchen Science Explained. New York: W.W. Nortan & Co. Internet Note: Most of the information listed here was compiled by Dr. Luke LaBorde from the Department of Food Science at Penn State University. We have added some other sites and given some explanations where they were not provided. The URL is: www.foodscience.psu.edu/outreach/fun_food_science.html Suggested search methods Instead of general browsing with words like "food experiments" or "food science experiments" we found that by going into a search engine such as Google or Askjeeves and putting in very specific requests such as "gluten balls" or "cooking egg experiments", we often had very good success. Experiments with Food Additives http://www.saskschools.ca/curr_content/science10/unitc/expemul.html There is a lot of good information here. The science unit on additives has experiments on emulsifiers and oxidation of fruits. Look in Unit C: Food Additives and Nutrition. Lesson 4: Experiments with Food Additives Disease transmission http://scienceforfamilies.allinfo-about.com/features/feelingrotten.html This is a very simple apple experiment to show how microorganisms are transmitted between foods or people. Testing for vitamin C http://www.agriscience.ca/pages/e_s_exper.html Students are taught how to make a simple indicator solution and then how to test various juices to check the vitamin C content. Food & Cooking Chemistry http://chemistry.about.com/od/foodcookingchemistry/ Learn about the chemistry of foods and cooking. Get information about vitamins, minerals, processes, herbs, ingredients, and other kitchen chemistry. This site gives lots of background information that is useful to explain food processes. Science Fair Food Experiments 64 http://chemistry.about.com/cs/sciencefairideas/a/aa041503a.htm Lots of ideas are suggested for science fair projects using food. Instructions are not given, but the ideas could be turned into classroom experiments. Testing for hidden sugar http://www.healthyteeth.org/experiments/hiddenSugar.html A very simple process is outlined for students to test foods for sugar. The idea is to choose some foods they may not think about that contain sugar. Food preservatives http://www.discoverykids.ca/gross/experiments/experimentsDetail.asp?id=67 A very simple experiment with bread to show the effectiveness of calcium propionate as a preservative. What is Food Science and Technology? http://www.foodscience.psu.edu/Explore/explore.html Learn about the science of food at Penn State University Academic curricula and learning sets Introduction To The Food Industry http://www.ift.org/cms/?pid=1000411 A self-study learning tool designed to assist high school students in their exploration into the food industry and its career opportunities. Institute of Food Technologists Food Safety Lessons for Middle School Students http://foodsafety.cas.psu.edu/nie/nie.html These lessons were designed to complement the Newspapers in Education (NIE) Supplement called Food Safety: From Farm to Table. The supplement is available as a PDF on this website in two alternative formats for your use. The centerpiece of the supplement is also here as a single PDF for your use. Bring Food Science into Your Middle and High School Classrooms http://www.nsta.org/288/ A curriculum used to engage students in inquiry-based science – The Food and Drug Administration (FDA) in collaboration with the National Science Teachers Association (NSTA) Fun Food Stuff http://www.biotech.wisc.edu/Education/FunFoodStuff/default.htm Science projects teachers can do with their students, at minimal cost, to illustrate some of the principles of biotechnology and of science experimentation in general. 65 Food and Science http://www.uen.org/utahlink/lp_res/nutri375.html Volume IV in a series of Food Nutrition and Science Curriculum projects developed for the Utah State Board of Education. School Sciences and Food http://www.ifst.org/school.htm Experiments and projects to assist teachers in developing their own programs for secondary school children. Institute of Food Science and Technology. FDA Food Safety Resources for Kids, Teens, & Educators http://www.cfsan.fda.gov/~dms/educate.html Games coloring books, songs, quizzes, puzzles and more that teach principles of safe handling of foods. Food Science Lesson Plans for K-12 students http://www.umaine.edu/NSFGK-12/lessons.html National Science Foundation GK-12 Fellows Program, University of Maine. Resources for teachers Marketplace for the Mind http://www.marketplaceforthemind.state.pa.us/ As you explore this site you will find the Agricultural Resource Library full of information on quality, non-biased references, books, lesson plans and teaching tools that are aligned to Pennsylvania's Science & Technology and Environment & Ecology teaching standards. The library can be searched in a multitude of convenient ways. 4-H Leaders' and Teachers' Guides Food and Nutrition http://www.nal.usda.gov/Kids/4hfood.htm P rogram and curricula guides developed by the USDA's Cooperative Extension Service to support youth education and programs. National Chemistry Week 2000: Journal of Chemical Education Resources in Food Chemistry, J. Chem. Educ. 2000, 77, 1256. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=food&-find=&format=detail. html&-skip=2&-max=1&-token.2=2&-token.3=25 Food chemistry information and activities at the college, high school, and elementary school levels (many of these are listed below). Chocolate: A Marvelous Natural Product of Chemistry http://www.jce.divched.org/Journal/Issues/2004/Aug/abs1131.html Chocolate is a natural product as ubiquitous as television. Of course, it is eaten, but it is also found in air fresheners, marking pens, flavoring in a multitude of products including soda pop, and as an aroma in "chocolate-dyed" T-shirts. Journal of Chemical Education August 2004 Vol. 81 No. 8 p. 1131 66 Artificial Sweeteners - No-calorie sugar substitutes provide options for enjoying the sweet life http://pubs.acs.org/cen/whatstuff/stuff/8225sweeteners.html "Most days my coffee is accompanied by packets of artificial sweetener instead of spoonfuls of sucrose--the compound we call sugar. But what exactly are the no-calorie sugar substitutes that help me keep my figure while satisfying my sweet tooth?..." Foods under the Microscope http://www.magma.ca/~scimat/ A collection of sub-microscopic images of common foods that visually illustrates why some foods have properties such as elasticity, firmness, and grittiness. The Accidental Scientist – Science of Cooking http://www.exploratorium.com/cooking/ Learn about the science behind candy, bread, eggs, pickles, meat and seasonings from the Exploratorium Science Museum in San Francisco. Where Candy Comes From - Resource guide http://www.agintheclassroom.org/candy/where%20candy%20booklet%20form.pdf Illinois Farm Bureau - Ag in the Classroom Simple experiments and activities young kids can do for school or science fair projects Fruit Juice Mystery http://www.sciencenter.org/chemistry/documents/fruitjuicemystery1a_000.pdfttp://www.exploratorium.com/cooki ng/ Challenge: Can you discover which juice is a fake made from food coloring, water, and sugar? National Science Teachers Association Testing Foods for Glucose and Starch http://school.discovery.com/curriculumcenter/chemistry/activity2.html Students practice safe laboratory methods while learning how to interpret results of chemical tests. They determine the foods to test and interpret and record their results. Based on the amount of glucose or starch present in the food, both tests will provide varying results. This gives students the chance to make decisions about results and helps them understand that scientists must repeat tests to confirm results. Countertop Chemistry http://www2.ncsu.edu/ncsu/pams/science_house/learn/CountertopChem/index.html Chemistry Activities that use chemicals you can find at the grocery or the hardware store. NC State University. Edible/Inedible Experiments Archive http://www.madsci.org/experiments Science should be fun….and science should be edible! Food batteries, cabbage juice pH indicator, generating light by chewing, and more! How Stuff Works – Foods 67 http://www.howstuffworks.com/category.htm?cat=Food Beer, coffee, antioxidants, food preservation, etc. Food Chemistry Experiments http://www.gomilpitas.com/homeschooling/explore/chemistryfood.htm A to Z Home's Cool Homeschooling Web Site Plastic Bag Ice Cream I http://www.sme.org/memb/neweek/actice.htm Students learn how to lower the freezing point of water and how ice cream forms as a solution freezes. Plastic Bag Ice Cream II http://www.agintheclassroom.org/resources/agziploc.pdf Illinois Farm Bureau - Ag ZipLocks & You - 10 plastic bag activities for kids Plastic Bag Ice Cream III http://www.agintheclassroom.org/resources/agziploc.pdf Wisconsin Ag in the Classroom Kitchen Chemistry http://www.discoveryeurope2.com/kitchen/index.php The Discovery Channel Lab Dad http://www.geocities.com/CapeCanaveral/Hall/1410/index.html Laboratory experiments for young people to do at home, as well as a reference for science teachers. Most of the labs are intended for grades 7-12, but many can easily be done by younger scientists. Introductory Electrochemistry for Kids - Food for Thought, and Human Potential. J. Chem. Educ. 1998, 75, 178. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex.fp5&-lay=wwwform&title=food&token.2=11&-token.3 =25&-find=&-format=detail.html&-skip=15&-max=1&-token.2=15&-token.3=25 &-skip=11&max=1&-token.2=11&-token.3=25 A simple, inexpensive experiment using fruit, a voltmeter, and wires made of various metals allows children to discover many electrochemical principles including: circuits, series connections, the chemical nature of batteries, electrodes, and potential/voltage Microwave Ovens What is the science behind microwave cooking? http://www.reachoutmichigan.org/funexperiments/agesubject/lessons/newton/micrwave.html Newtons Apple Making Ginger Ale at Home http://biology.clc.uc.edu/fankhauser/Cheese/Ginger_Ale_Ag0.htm Making Root Beer at Home http://biology.clc.uc.edu/fankhauser/Cheese/ROOTBEER_Jn0.htm David B. Fankhauser, Ph.D. Professor of Biology and Chemistry U.C. Clermont College Batavia OH 45103 Food Preservation http://www.sciencenetlinks.com/lessons.cfm?BenchmarkID=8&DocID=396 Grade 3-5 lessons to help students understand how various food preservation techniques help to slow down the spoiling of food by microscopic organisms 68 Food safety http://www.enc.org/resources/records/0,1240,025548,00.shtm Science in a technical world. Grades: 9 10 11 12 Food chemistry http://www.enc.org/resources/records/0,1240,001491,00.shtm Science and Technology for Children (STC). Grades: 4 Classroom experiments, demonstrations, and resources for beginning food scientists Student experiments with food www.micrecolde/food.html A short description and conclusion for each experiment. Shows photos of results for a variety of experiments, but no detailed explanations. Food Science experiments for K-12 students. http://www.clemson.edu/foodscience/teachers.htm Hydrogen bond formation, pH and titratable acidity, rancidity of foods, enzymatic denaturation of proteins, color changes in chlorophyll, etc. Finding Science in Ice Cream - An Experiment for Secondary School Classrooms http://www.foodsci.uoguelph.ca/dairyedu/findsci.html As the hot weather approaches and students minds begin to drift from the rigors of the school classroom or laboratory, a fun afternoon might be spent making ice cream and in so doing, introducing several aspects of the science and technology "behind the scenes". This web site is a classroom experiment for school teachers on ice cream making. Anthocyanins: A Colorful Class of Compounds. J. Chem. Educ. 1997, 74, 1176A. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=anthocyanins&find=&-format=detail.html &-skip=0&-max=1&-token.2=0&-token.3=25 The red colors of leaves in the fall, and the colors of radish skins, and some cabbages result from pigments known as anthocyanins. This activity makes use of the fact that these substances are also acid-base indicators. Tick Tock, a Vitamin C Clock J. Chem. Educ. 2002, 79, 40A. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=tock&-find=&format=detail.html&- skip=0&-max=1&-token.2=0&-token.3=25 This Activity can be used to explore reaction kinetics, and in particular the effect of reactant concentrations on the apparent rate of a reaction. It can also be used in a discussion of redox chemistry, the descriptive chemistry of iodine, and the chemistry of vitamin C. Fizzy Drinks: Stoichiometry You Can Taste. J. Chem. Educ. 2000, 77, 1608A. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=taste&-find=&format=detail.html&-skip=0&-max=1&-token.2=0&-token.3=25 Students create their own Fizzie-style carbonated beverage. They find that using too much baking soda or too much citric acid ruins the taste. In the final steps, they use stoichiometry to calculate the correct mix of these two ingredients. Cabbage Patch Chemistry. J. Chem. Educ. 2000, 77, 1432A. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=cabbage&-find=&- 69 format=detail.htm l&-skip=0&-max=1&-token.2=0&-token.3=25 Students make sauerkraut and also investigate the effect of changing one variable in the sauerkraut-making process. Put an Enzyme to Work. http://gslc.genetics.utah.edu/units/activities/proteins/ This is a simple experiment using pineapple and gelatin to show how enzymes work. Soup or Salad - Investigating the Action of Enzymes in Fruit on Gelatin. J. Chem. Educ. 1999, 76, 624A. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=gelatin&-find=&format=detail.html &-skip=0&-max=1&-token.2=0&-token.3=25 Students observe gelatin samples treated with substances that may or may not have an enzymatic effect on the protein in the gelatin. Substances used are fresh pineapple, canned pineapple, fresh pineapple that has been frozen and microwaved, and meat tenderizer. Apple Fool! An Introduction to Artificial Flavors. J. Chem. Educ. 2003, 80, 408A. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=apple fool&-find=&format=detail.html &-skip=0&-max=1&-token.2=0&-token.3=25 Students investigate flavorings by making artificial “cooked apples” from a mixture of crackers, sugar, cream of tartar, and water, as is done for the filling in recipes for Mock Apple Pie. Flat As a Pancake? Exploring Rising in Baked Goods. J. Chem. Educ. 2000, 77, 1264A. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=Pancake&-find=&format=detail.html &-skip=0&-max=1&-token.2=0&-token.3=25 Baked goods have a wide range of characteristics. The ingredients and the relative amounts of each along with mixing and baking techniques determine the properties (appearance, texture, taste, and nutritive value) of the product. This activity investigates the action of baking powder. The Alginate Demonstration: Polymers, Food Science, and Ion Exchange . J. Chem. Educ. 1998, 75, 1430. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=alginate&-find=&format=detail.htm l&-skip=0&-max=1&-token.2=0&-token.3=25 A polymer demonstration involving the calcium crosslinking of Sodium alginate. Detection of Catalysis by Taste. J. Chem. Educ. 1998, 75, 315. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=taste&-find=&format=detail.html&-skip=1&-max=1&-token.2=1&-token.3=25 Sensory demonstration of the action of the enzyme b-galactosidase (or lactase) which catalyzes the hydrolysis of lactose into Dglucose and D-galactose. The Heat Content of Nuts and Snack Foods. http://www.woodrow.org/teachers/chemistry/institutes/1988/foodheat.html In this experiment, several types of nuts and snack foods are burned in order to determine their heat content per gram. More advanced experiments and demonstrations in food science 70 Chromatography of Food Dyes: http://www.ift.org/publications/jfse/jfsev1n1p0018-0022ms20010622.pdf A Simple Demonstration for Actively Engaging High School Students in the Chemistry of Foods. Journal of Food Science Education. Bioanalytical Experiments for the Undergraduate Laboratory - Monitoring Glucose in Sports Drinks. J. Chem. Educ. 2001, 78, 788. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?db=jceindex.fp5&-lay =wwwform&title=bioanalytical&-token.2=0&-token.3=25&-find=&-format=detail.html&-skip =1&-max=1&-token.2=1&-token.3=255 A solution-based enzyme assay for the analysis of glucose concentrations in sports drinks and fabrication of an enzyme electrode. Hot Chili Peppers: Extraction, Cleanup, and Measurement of Capsaicin. J. Chem. Educ. 2000, 77, 1630. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=chili&-find=&format=detail.html &-skip=0&-max=1&-token.2=0&-token.3=25 Determination of the Scoville Heat Value for Hot Sauces and Chilies: An HPLC Experiment. J. Chem. Educ. 2000, 77, 266. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=chili&-find=&format=detail.html &-skip=1&-max=1&-token.2=1&-token.3=25 Pungency Quantitation of Hot Pepper Sauces Using HPLC. J. Chem. Educ. 1999, 76, 240. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=pepper&-find=&format=detail.html &-skip=1&-max=1&-token.2=1&-token.3=25 Laboratory activities to acquaint students with the active ingredients of hot chili pepper (capsaicin and dihydrocapsaicin) and the extraction, cleanup, and HPLC determination of Scoville heat value (SHV). Pungency Assessment in Onions. http://www.zoo.utoronto.ca/able/volumes/vol-19/11-randle/11-randle.htm A common assessment of pungency is made by measuring pyruvate, which is formed as a stable primary compound from the enzymatic decomposition of each of the flavor precursors. Pyruvate is produced in a mole for mole relationship with the flavor precursors Chemical residues in food http://terrificscience.org/freeresources/lessonpdfs/What_You_Eat.pdf Simulated chemical residues experiment and follow-up discussion ideas on consumer decisions regarding risks of food Integrating Introductory Biology and Chemistry Laboratories: Human Metabolism of Vitamin C and Fruit Juice Analysis—an Example http://www.zoo.utoronto.ca/able/volumes/vol-18/11-godrick.pdf Three and a half week module called the Examination of Vitamin C (ascorbic acid), its Sources, Properties, and Metabolism. Boston University Enzymatic Spectrophotometric Reaction Rate Determination of Glucose in Fruit Drinks and Carbonated Beverages. J. Chem. Educ. 2000, 77, 1327. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=carbonated &find=&-format=detail.html &-skip=1&-max=1&-token.2=1&-token.3=25 71 This paper describes an undergraduate analytical chemistry laboratory for the kinetic determination of glucose in fruit drinks and carbonated beverages using glucose oxidase (GOX) and horseradish peroxidase (HRP). Determination of Aspartame and Caffeine in Carbonated Beverages Utilizing Electrospray Ionization-Mass Spectrometry. J. Chem. Educ. 2000, 77, 1325. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=carbonated&-find=&format=detail.html &-skip=0&-max=1&- token.2=0&-token.3=25 Procedures can be used to quantify the content of caffeine and aspartame in beverages or to understand the capabilities of electrospray ionization. Enzyme Kinetics http://www.zoo.utoronto.ca/able/volumes/vol-19/6-stiefbold.pdf A laboratory exercise for assaying b-galactosidase enzyme activity - part of a sophomore level core curriculum course required of University of Oregon biology majors. The Other Double Helix—The Fascinating Chemistry of Starch. J. Chem. Educ. 2000, 77, 988. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=helix&-find=&format=detail.html&-skip=0&-max=1&-token.2=0&-token.3=25 The chemistry of starch; the most abundant biomolecule on earth after cellulose and a major part of the diet of all human beings. The Arrhenius Law and Storage of Food in a Freezer. J. Chem. Educ. 1999, 76, 504. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title =freezer&-find=&format=detail.html &-skip=0&-max=1&-token.2=0&-token.3=25 Application of the Arrhenius law for temperature dependency of chemical reactions Showing Properties of Food Foams with Common Dairy Foods. J. Chem. Educ. 1997, 74, 1133. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title =foams&-find=&format=detail.html &-skip=0&-max=1&-token.2=0&-token.3=25 Demonstration of how food properties can be visualized with the aid of simple and inexpensive experiments using dairy products that can be found in any kitchen Demonstration of the exponential decay law using beer froth http://ej.iop.org/links/q91/qM08CBJiaBJEiYuaZh2NIg/ej2104.pdf The volume of beer froth decays exponentially with time. This property is used to demonstrate the exponential decay law in the classroom. The decay constant depends on the type of beer and can be used to differentiate between different beers. Eur. J. Phys. 23 (2002) 21–26 A Simple Laboratory Exercise in Food Structure/Texture Relationships Using a Flatbed Scanner. J. Chem. Educ. 2000, 77, 368. http://jchemed4.chem.wisc.edu:8081/JCEIndex/FMPro?-db=jceindex&-lay=wwwform&title=flatbed&-find=&format=detail.html &-skip=0&-max=1&-token.2=0&-token.3=25 A demonstration a simple laboratory exercise designed to demonstrate structure/texture relationships in a common food product using a common flatbed scanner.. Characterization of Organic Illumination Systems. A study on the science of Lifesavers that shoot sparks when you chew them and the glowing pickle phenomenon * http://www.research.compaq.com/wrl/techreports/abstracts/TN-13.html *really! 72 Sauerkraut Fermentation. http://www.splammo.net/foodapplmicro/applkraut.html Bacteriology/Food Science 324 at the University of Wisconsin – Madison. For more experiments visit the Journal of Food Science Education (http://www.ift.org/publications/jfse/) and the Journal of Chemical Education (http://jchemed.chem.wisc.edu/index.html)