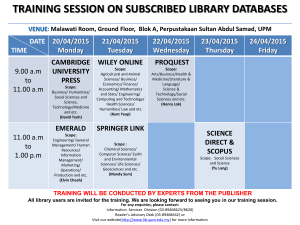
Thermal engineering
advertisement

Master in Space Science and Technology Thermal engineering Isidoro Martínez Master in Space Science and Technology UPM. Isidoro Martínez 1 Thermal engineering •Thermodynamics – Basics • Energy and entropy • Temperature and thermometry • Variables: state properties, process functions • Equations of state, simple processes • Phase change – Applied: • Mixtures. Humid air (air conditioning) • Thermochemistry (combustion) • Heat engines (power generation) • Refrigeration (cold generation) • Thermal effects on materials and processes • Thermofluiddynamic flow 1D… •Heat transfer (conduction, convection, radiation, heat exchangers) Master in Space Science and Technology, UPM. Isidoro Martínez 2 Thermodynamics • Basic thermodynamics – The science of heat and temperature. Work. Energy. Thermal energy. – Energy and entropy. The isolated system. The traditional Principles – Generalisation (mass, momentum, energy): the science of assets (conservatives do not disappear) and spreads (conservatives tend to disperse) – Type of thermodynamic systems (system, frontier, and surroundings) • Isolated system: Dm=0, DE=0 • Closed system : Dm=0, DE0 • Open system : Dm0, DE0 – Type of thermodynamic variables • Intensive or extensive variables • State or process variables – Type of thermodynamic equations • Balance equations (conservation laws); e.g. DEclose-sys=W+Q • Equations of state (constitutive laws); e.g. pV=mRT • Equilibrium laws: S(U,V,ni)iso-sys(t) Smax e.g. dS/dU|V,ni=uniform… • (Kinetics is beyond classical thermodynamics; e.g. q kT ) • Applied thermodynamics Master in Space Science and Technology, UPM. Isidoro Martínez 3 Thermodynamics (cont.) • Basic thermodynamics • Applied thermodynamics – – – – – – Energy and exergy analysis (minimum expense and maximum benefit) Non-reactive mixtures (properties of real mixtures, ideal mixture model…) Hygrometry (humid air applications: drying, humidification, air conditioning…) Phase transition in mixtures (liquid-vapour equilibrium, solutions…) Reactive mixtures. Thermochemistry. Combustion Heat engines • Gas cycles for reciprocating and rotodynamic engines • Vapour cycles (steam and organic fluid power plants) – Refrigeration, and heat pumps • Cryogenics (cryocoolers, cryostats, cryopreservation…) – Thermal analysis of materials (fixed points, calorimetry, dilatometry…) – Non-equilibrium thermodynamics (thermoelectricity, dissipative structures…) – Environmental thermodynamics (ocean and atmospheric processes…) Master in Space Science and Technology, UPM. Isidoro Martínez 4 Balance equations Magnitude Accumul . mass dm = momentum d(m v ) = Production energy d(me) = 0 mg dt 0 entropy exergy d(ms) d(m) = = dSgen T0dSgen Impermeable flux +0 + FA dt +dW+dQ +dQ/T +dWu+(1T0/T)dQ Permeable flux +dme + ve dmepeAe ne dt e+htedme +sedme +edme with e=u+em=u+gz+v2/2 dW =IFFdx =IFMdq, Wu=W+ p0DV h=u+pv, ht=h+em ds=(du+pdv)/T =(dq+demdf)/T, demdf 0, dSgen0 =e+p0vT0s, =htT0s Master in Space Science and Technology, UPM. Isidoro Martínez 5 Substance data • Perfect gas model – Ideal gas: pV=mRT or pV=nRuT (R=Ru/M, Ru=8.3 J/(mol·K)) – Energetically linear in temperature: DU=mcvDT – • Air data: R=287 J/(kg·K) and cp=cv+R=1000 J/(kg·K), or M=0.029 kg/mol and g=cp/cv=1.4 Perfect solid or liquid model – Incompressible, undilatable substance: V=constant (but beware of dilatations!) – Energetically linear in temperature: DU=mcDT – • Water data: r=1000 kg/m3, c=4200 J(kg·K) Perfect mixture (homogeneous) v xi vi , u xiui , s xi si R xi ln xi – Ideal mixture – Energetically linear in temperature: DU=mcvDT * • * * Heterogeneous systems – Phase equilibria of pure substances (Clapeyron’s equation) xV 1 p1* (T ) – Ideal liquid-vapour mixtures (Raoult’s law): x p L1 – Ideal liquid-gas solutions (Henry’s law): cL,s K sdis,cc (T ) • dp dT sat Dh T Dv cG , s Real gases. The corresponding state model, and other equations of state. Master in Space Science and Technology, UPM. Isidoro Martínez 6 Thermodynamic processes • Adiabatic non-dissipative process of a perfect gas: dE dQ dW • • mcv dT mRT dV V dT R dV 0 Tvg 1 cte., pvg cte., T/p T cv V g 1 g cte. Fluid heating or cooling – – • dU pdV At constant volume: Q=DU At constant pressure: Q=DH=D(U+pV) Adiabatic gas compression or expansion – Close system: w=Du=cv(T2-T1) – Open system: w=Dh=cp(T2-T1) C ws h2ts h1t w h2t h1t PGM p2t g 1 g p1t 1 w PGM 1 T1t T2t T g 1 T2t T1t 1 ws 1 p1t p2t g Internal energy equation (heating and cooling processes) DU DE DEm Q Emdf pdV • One-dimensional flow at steady state min mout r vA rV • Dh w q Thermodynamic processes in engines w dp r Dem emdf Master in Space Science and Technology, UPM. Isidoro Martínez 7 Phase diagrams (pure substance) • Normal freezing and boiling points (p0=100 kPa) • Triple point (for water TTR=273.16 K, pTR=611 Pa) • Critical point (for water TCR=647.3 K, pTR=22.1 MPa) • Clapeyron’s equation (for water hSL=334 kJ/kg, hLV=2260 kJ/kg) dp dT sat hV hL T (vV v L ) FG p IJ h FG 1 1 IJ H p K R HT T K V v L , vV RT / p , hLV const v ln LV 0 Master in Space Science and Technology, UPM. Isidoro Martínez 0 8 Thermometry • Temperature, the thermal level of a system, can be measured by different primary means: – – – – – T pV lim The ideal-gas, constant-volume thermometer TTPW p 0 pV TPW The acoustic gas thermometer 1/ 4 The spectral radiation thermometer T M lim The total radiation thermometer TTPW 1 M TPW The electronic noise thermometer • The temperature unit is chosen such that TTPW 273.16 K • The Celsius scale is defined by T/ºC T/K-273.15 • Practical thermometers: – Thermoresistances (e.g. Pt100, NTC) – Thermocouples (K,J…). Master in Space Science and Technology, UPM. Isidoro Martínez 9 Piezometry • Pressure (normal surface force per unit normal area), is a scalar magnitude measured by difference (in non-isolated systems; recall free-body force diagrams). • Gauge and absolute pressure: • • Pressure unit (SI) is the pascal, 1 Pa1 N/m2 (1 bar100 kPa) PLM Hydrostatic equation: p p0 r g z z0 dp r g PGM dp p dz g dz RT Vacuum (practical limit is about 10-8 Pa) Pressure sensors: U-tube, Bourdon tube, diaphragm, piezoelectric… • • Master in Space Science and Technology, UPM. Isidoro Martínez 10 Questions (Only one answer is correct) 1. The mass of air in a 30 litre vessel at 27 ºC and a gauge pressure of 187 kPa is about? a) b) c) d) 2. When a gas in a 30 litre rigid vessel is heated from 50 ºC to 100 ºC, the pressure ratio: (final/initial): a) b) c) d) 3. Cannot be compressed Cannot be heated by compression Heat a little bit when compressed, but volume remains the same Heat up and shrink when compressed The critical temperature of any gas is: a) b) c) d) 5. Doubles Is closer to 1 than to 2. Depends on initial volume Depends on heating speed Liquids: a) b) c) d) 4. 1g 10 g 100 g 1000 g.. The temperature below which the gas cannot exist as a liquid -273.16 ºC The temperature above which the gas cannot be liquefied The temperature at which solid, liquid, and gas coexist In a refrigerator, the amount of heat extracted from the cold side: a) b) c) d) Cannot be larger than the work consumed Cannot be larger than the heat rejected to the hot side Is inversely proportional to the temperature of the cold side Is proportional to the temperature of the cold side. Master in Space Science and Technology, UPM. Isidoro Martínez 11 Questions (Only one answer is correct) 6. Which of the following assertions is correct? a) b) c) d) 7. The variation of entropy in a gas when it is compressed in a reversible way is: a) b) c) d) 8. Is always positive Is dimensionless Is different in the Kelvin and Celsius temperature scales Is three times the linear coefficient value. It is not possible to boil an egg in the Everest because: a) b) c) d) 10. Less than zero Equal to zero Greater than zero It depends on the process. The volumetric coefficient of thermal expansion: a) b) c) d) 9. Heat is proportional to temperature Heat is a body’s thermal energy Net heat is converted to net work in a heat engine The algebraic sum of received heats in an interaction of two bodies must be null. The air is too cold to boil water Air pressure is too low for stoves to burn Boiling water is not hot enough Water cannot be boiled at high altitudes. When a combustion takes place inside a rigid and adiabatic vessel: a) b) c) d) Internal energy increases Internal energy variation is null Energy is not conserved Heat flows out. Master in Space Science and Technology, UPM. Isidoro Martínez 12 Exercises 1. A U-tube is made by joining two 1 m vertical glass-tubes of 3 mm bore (6 mm external diameter) with a short tube at the bottom. Water is poured until the liquid fills 600 mm in each column. Then, one end is closed. Find: 1. 2. 2. 3. 4. An aluminium block of 54.5 g, heated in boiling water, is put in a calorimeter with 150 cm3 of water at 22 ºC, with the thermometer attaining a maximum of 27.5 ºC after a while. Find the thermal capacity of aluminium. How many ice cubes of 33 g each, at -20 ºC, are required to cool 1 litre of tea from 100 ºC to 0 ºC? Carbon dioxide is trapped inside a vertical cylinder 25 cm in diameter by a piston that holds internal pressure at 120 kPa. The plunger is initially 0.5 m from the cylinder bottom, and the gas is at 15 ºC. Thence, an electrical heater inside is plugged to 220 V, and the volume increases by 50% after 3 minutes. Neglecting heat losses through all walls, and piston friction, find: 1. 2. 5. The change in menisci height due to an ambient pressure change, (∂z/∂pamb), with application to Dp=1 kPa. The change in menisci height due to an ambient temperature change, (∂z/∂Tamb), with application to DT=5 ºC. The energy balance for the gas and for the heater. The final temperature and work delivered or received by the gas. Find the air stagnation temperature on leading edges of an aircraft flying at 2000 km/h in air at -60 ºC. Master in Space Science and Technology, UPM. Isidoro Martínez 13 Master in Space Science and Technology Thermal engineering Isidoro Martínez Master in Space Science and Technology UPM. Isidoro Martínez 14 Thermal engineering •Thermodynamics –Basic (energy and entropy, state properties, state equations, simple processes, phase changes) –Applied (mixtures, liquid-vapour equilibrium, air conditioning, thermochemistry, power and cold generation, materials processes) •Heat transfer – – – – – – – Thermal conduction (solids…) Thermal convection (fluids…) Thermal radiation (vacuum…) Heat exchangers Heat generation (electrical heaters…) Thermal control systems Combined heat and mass transfer (evaporative cooling, ablation…) Master in Space Science and Technology, UPM. Isidoro Martínez 15 Heat transfer • What is heat (i.e. heat flow, heat transfer)? – First law: heat is non-work energy-transfer through an impermeable surf. Q DE W DE pdV Wdis DH Vdp Wdis mcDT PIS,non-dis – Second law: heat tends to equilibrate the temperature field. sgen • T q T2 What is heat flux (i.e. heat flow rate, heat transfer rate)? dQ dT Q mc dt dt • KADT PSM,non-dis Heat transfer is the flow of thermal energy driven by thermal nonequilibrium (i.e. the effect of a non-uniform temperature field), commonly measured as a heat flux (vector field). Master in Space Science and Technology, UPM. Isidoro Martínez 16 Heat transfer modes • How is heat flux density modelled? conduction q k T Q q K DT convection q h T T A 4 4 radiation q T T – The 3 ways to change Q: K, A, and DT. – K is thermal conductance coeff. (or heat transfer coeff.), k is conductivity, h is convective coeff., is emissivity. – Field or interface variables? – Vector or scalar equations? – Linear or non-linear equations? – Material or configuration properties? – Which emissivity? This form only applies to bodies in large enclosures. Master in Space Science and Technology, UPM. Isidoro Martínez 17 Heat conduction • Physical transport mechanism –Short-range atomic interactions (collision of particles in fluids, or phonon waves in solids), supplemented with free-electron flow in metals. • Fourier’s law (1822) q kT • Heat equation dH dt Q p rc V T dV q ndA dV q dV dV t A V V V k 0 T rc q k 2T , or t V 0 T a 2T t rc –with the initial and boundary conditions particular to each problem. Master in Space Science and Technology, UPM. Isidoro Martínez 18 Thermal conductivity Table 1. Representative thermal conductivity values k [W/(m·K)] Comments Order of magnitude for solids 10 (good conductors) In metals, Lorentz's law (1881), k/(T)=constant 1 (bad conductors) Aluminium 200 Duralumin has k=174 W/(m·K), increasing to k=188 W/(m·K) at 500 K. Iron and steel 50 (carbon steel) Increases with temperature. 20 (stainless steel) Decreases with alloying Order of magnitude for liquids 1 (inorganic) Poor conductors (except liquid metals). 0.1 (organic) Water 0.6 Ice has k=2.3 W/(m·K), 2 Order of magnitude for gases 10 Very poor thermal conductors. KTG predicts k/(rc)a=Di=10-5 m2/s Air 0.024 Super insulators must be air evacuated. 2 Master in Space Science and Technology, UPM. Isidoro Martínez 19 Simple heat conduction cases • One-dimensional steady cases – Planar – Cylindrical – Spherical • • Q kA T1 T2 L12 Q k 2 L Q k 4 R1R2 T1 T2 R2 R1 T1 T2 R ln 2 R1 Composite wall (planar multilayer) T T T T T T q K DT k12 2 1 k23 3 2 ... n 1 Li L12 L23 k i Unsteady case. Relaxation time: Bi 1 r cL2 Dt k hL Dt mcDT / Q Bi Bi 1 r cV kS Dt hA Master in Space Science and Technology, UPM. Isidoro Martínez K 1 Li k i 20 Multiple path in heat conduction • Multidimensional analysis – Analytical, e.g. separation of variables, conduction shape factors, – Numerical, finite differences, lumped network, finite elements • Parallel thermal resistances – Example: honeycomb panel made of ribbon (thickness d), cell size s: DT Qx kFx Ax x Lx Qy kFy Ay Qz kFz Az DTy Ly DTz Lz Master in Space Science and Technology, UPM. Isidoro Martínez 3d with Fx 2 s d with Fy s 8d with Fz 3 s 21 Heat convection • Newton’s law and physical mechanism q h T T knT • • hDT k DT d Nu hL f Re,Pr... k W/(m2·K) e.g. in air flow, h=a+bvwind, with a=3 and b=3 J/(m3·K) e.g. plate (<1 m)at rest, h=a(T-T)1/4, with a2 W/(m2·K5/4) (1,6 upper, 0.8 lower, 1.8 vert.) • Classification of heat convection problems – – – – – By time change: steady, unsteady (e.g. onset of convection) By flow origin: forced (flow), natural (thermal, solutal…) By flow regime: laminar, turbulent By flow topology: internal flow, external flow By flow phase: single-phase or multi-phase flow • Heat exchangers (tube-and-shell, plates…) • Heat pipes Master in Space Science and Technology, UPM. Isidoro Martínez 22 Heat radiation • Heat radiation (thermal radiation) – It is the transfer of internal thermal energy to electromagnetic field energy, or viceversa, modelled from the basic black-body theory. Electromagnetic radiation is emitted as a result of the motion of electric charges in atoms and molecules. • Blackbody radiation – Radiation within a vacuum cavity • • • • Radiation temperature (equilibrium with matter) Photon gas (wave-particle duality, carriers with zero rest mass, E=h, p=E/c) Isotropic, unpolarised, incoherent spatial distribution Spectral distribution of photon energies at equilibrium (E=const., S=max.) – Radiation escaping from a hole in a cavity • Blackbody emmision 2 hc 2 M hc 5 exp k T 1 W 4 8 M M d T 5.67· 10 0 m2 K 4 C C 0.003 m K M max T Master in Space Science and Technology, UPM. Isidoro Martínez 23 Thermo-optical properties • Propagation through real media – Attenuation by absorption and scattering (Rayleigh if d<<, Mie if d) • Properties of real surfaces – Partial absorption (a), reflectance (r), emissivity (), and, in some cases, transmittance (t). Energy balance: art=1. – Directional and spectral effects (e.g. retroreflective surfaces, selective glasses…) – Detailed equilibrium: Kirchhoff's law (1859), aqT=qT, but usually a≠ • Spectral and directional modelling – Two-spectral-band model: – Diffuse (cosine law) or specular models • Radiative coupling • T14 T24 – e.g. planar infinite surfaces: q12 1 1 1 2 1 1 2 cos 1 cos 2 1 dA1dA2 View factors F12 A1 A2 A1 r122 Master in Space Science and Technology, UPM. Isidoro Martínez 24 Heat transfer goals • Analysis –Find the heat flux for a given set-up and temperature field e.g. Q kA T T / L 1 2 –Find the temperature corresponding to a given heat flux and set-up e.g. T T QL / kA) 1 • 2 Design –Find an appropriate material that allows a prescribed heat flux with a given T-field in a given geometry e.g. k QL / ADT –Find the thickness of insulation to achieve a certain heat flux with a given T-field in a prescribed geometry e.g. L kA T T / Q 1 • 2 Control -To prevent high temperatures, use insulation and radiation shields, or use heat sinks and coolers. –To prevent low temperatures, use insulation and radiation shields, or use heaters. –To soften transients, increase thermal inertia (higher thermal capacity, phase change materials). Master in Space Science and Technology, UPM. Isidoro Martínez 25 Application to electronics cooling • All active electrical devices at steady state must evacuate the energy dissipated by Joule effect (i.e. need of heat sinks). • Most electronics failures are due to overheating (e.g. for germanium at T>100 ºC, for silicon at T>125 ºC). • At any working temperature there is always some dopant diffusion at junctions and bond-material creeping, causing random electrical failures, with an event-rate doubling every 10 ºC of temperature increase. Need of thermal control. • Computing power is limited by the difficulty to evacuate the energy dissipation (a Pentium 4 CPU at 2 GHz in 0.18 mm technology must dissipate 76 W in an environment at 40 ºC without surpassing 75 ºC at the case, 125 ºC at junctions). • Modern electronic equipment, being powerfull and of small size, usually require liquid cooling (e.g. heat pipes). Master in Space Science and Technology, UPM. Isidoro Martínez 26 Thermal modelling • Modelling the geometry • Modelling the material properties • Modelling the transients • Modelling the heat equation • Mathematical solution of the model – Analytical solutions – Numerical solutions • Analysis of the results • Verification planning (analytical checks and testing) • Feedback Master in Space Science and Technology, UPM. Isidoro Martínez 27 Questions (Only one answer is correct) 1. The steady temperature profile in heat transfer along a compound wall: a) b) c) d) 2. If the temperature at the hot side of a wall is doubled: a) b) c) d) 3. It loses and gains heat at the same rate The heat absorbed equals its thermal capacity It reflects all the energy that strikes it No more heat is absorbed. A certain blackbody at 100 ºC radiates 100 W. How much radiates at 200 ºC? a) b) c) d) 5. Heat flow through the wall doubles Heat flow through the wall increases by a factor of 4 Heat flow through the wall increases by a factor of 8 None of the above. When a piece of material is exposed to the sun, its temperature rises until: a) b) c) d) 4. Has discontinuities Must have inflexion points Must be monotonously increasing or decreasing Must have a continuous derivative. 200 W 400 W 800 W None of the above. When two spheres, with same properties except for their radius, are exposed to the Sun and empty space: a) b) c) d) The larger one gets hotter The larger one gets colder The larger one gets hotter or cooler depending on their emissivity-to-absorptance ratio None of the above. Master in Space Science and Technology, UPM. Isidoro Martínez 28 Exercises 1. 2. 3. 4. 5. A small frustrum cone 5 cm long, made of copper, connects two metallic plates, one at 300 K in contact with the smallest face, which is 1 cm in diameter, and the other at 400 K, at the other face, which is 3 cm in diameter. Assuming steady state, quasi-one-dimensional flow, and no lateral losses, find: – The temperature profile along the axis. – The heat flow rate. An electronics board 100·150·1 mm3 in size, made of glass fibre laminated with epoxy, and having k=0.25 W/(m·K), must dissipate 5 W from its components, which are assumed uniformly distributed. The board is connected at the largest edges to high conducting supports held at 30 ºC. Find: 1. The maximum temperature along the board, if only heat conduction at the edges is accounted for (no convection or radiation losses). 2. The thickness of a one-side copper layer (bonded to the glass-fibre board) required for the maximum temperature to be below 40 ºC above that of the supports. 3. The transient temperature field, with and without a convective coefficient of h=2 W/(m2·K). Find the required area for a vertical plate at 65 ºC to communicate 1 kW to ambient air at 15 ºC. Consider two infinite parallel plates, one at 1000 ºC with =0.8 and the other at 100 ºC with =0.7. Find: – The heat flux exchanged. – The effect of interposing a thin blackbody plate in between. Find the steady temperature at 1 AU, for an isothermal blackbody exposed to solar and microwave background radiation, for the following geometries: planar one-side surface (i.e. rear insulated), plate, cylinder, sphere, and cube. (END) Master in Space Science and Technology, UPM. Isidoro Martínez 29 Referencias • http://webserver.dmt.upm.es/~isidoro/ • Martínez, I., "Termodinámica básica y aplicada", Dossat, 1992. • Wark, K., "Thermodynamics", McGraw-Hill, 1999. Versión española Edit. McGraw-Hill, 2000. • Holman, J.P., "Heat transfer", McGraw-Hill, 2010. • Gilmore, D.G., “Satellite Thermal Control Handbook”, The Aerospace Corporation Press, 1994. • www.electronics-cooling.com Master in Space Science and Technology, UPM. Isidoro Martínez 30