Chemistry Reference Tables - Constants, Formulas, Periodic Table
advertisement

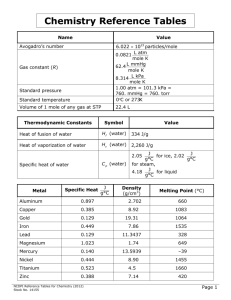
Chemistry Reference Tables Name Value Standard temperature 6.022 × 1023 particles/mole 0.0821 L atm mole K L mmHg 62.4 mole K 8.314 L kPa mole K 1.00 atm = 101.3 kPa = 760. mmHg = 760. torr 0°C or 273K Volume of 1 mole of any gas at STP 22.4 L Avogadro’s number Gas constant (R) Standard pressure Thermodynamic Constants Symbol Value Heat of fusion of water H f (water) 334 J/g Heat of vaporization of water Hv (water) 2,260 J/g J for ice, 2.02 J for steam, g°C g°C J 4.18 for liquid g°C 2.05 C p (water) Specific heat of water Metal Specific Heat J g°C Density (g/cm3) Melting Point (°C) Aluminum 0.897 2.702 660 Copper 0.385 8.92 1083 Gold 0.129 19.31 1064 Iron 0.449 7.86 1535 Lead 0.129 Magnesium 1.023 Mercury 0.140 Nickel 0.444 8.90 1455 Titanium 0.523 4.5 1660 Zinc 0.388 7.14 420 NCDPI Reference Tables for Chemistry (October 2006 form A-v1) 11.3437 1.74 13.5939 328 649 —39 Page 1 Organic Substances Name Density Melting Point (°C) Ethanol (CH3CH2OH) 0.7893 g/cm 3 −119 Glucose (C6H12O6) 1.54 g/cm 3 Hexane (C6H14) 0.6603 g/cm 3 Methane (CH4) 0.716 g/L Methanol (CH3OH) 0.7914 g/cm 3 Sucrose (C12H22O11) 1.27 g/cm 3 86 Boiling Point (°C) 79 Decompose −95 69 −182 −164 −94 65 86 Decompose Inorganic Substances Name *Density @ STP Melting Point (°C) Boiling Point (°C) Chlorine 3.21 g/L −101 −35 Hydrogen 0.0899 g/L −259 −253 Hydrogen chloride 1.640 g/L −115 −85 Hydrogen sulfide 1.54 g/L −85 −61 Nitrogen 1.25 g/L −210 −196 Nitrogen monoxide 1.34 g/L −164 −152 Oxygen 1.43 g/L −218 −183 Sodium carbonate 2.532 g/cm 3 851 Decomposes Sodium chloride 2.165 g/cm 3 801 1413 Sulfur dioxide 2.92 g/L −73 −10 *Water (at 4°C) 1.00 g/cm 3 0 100 NCDPI Reference Tables for Chemistry (October 2006 form A-v1) Page 2 Formulas D= m V D = density K = °C + 273 m = mass PV PV 1 1 = 2 2 T1 T2 V = volume Pt = P1 + P2 + P3 + ... K = Kelvin M1V1 = M2V2 P = pressure PV = nRT R = gas constant M = moles of solute liters of solution T = temperature q = mCpΔT M = molarity q = mHv n = number of moles q = mH f q = quantity of heat energy pH + pOH = 14 Cp = specific heat pH = − log[H +] Hv = heat of vaporization pOH = − log[OH −] H f = heat of fusion K w = [H +][OH −] = 1 × 10−14 K w = equilibrium constant for the ionization of water [H +] = 10 −pH [OH −] = 10−pOH NCDPI Reference Tables for Chemistry (October 2006 form A-v1) Page 3 PERIODIC TABLE 1 IA 1 H 1.008 2 IIA 3 Li 4 Be Lithium Beryllium 6.941 9.012 11 Na 12 Mg Hydrogen Sodium Magnesium 22.99 24.31 3 IIIB 4 IVB 5 VB 6 VIB 7 VIIB 8 VIIIB 9 VIIIB 19 K 20 Ca 21 Sc 22 Ti 23 V 24 Cr 25 Mn 26 Fe 27 Co Potassium Calcium Scandium Titanium Vanadium Chromium Manganese Iron Cobalt 39.10 40.08 44.96 47.88 50.94 51.99 54.94 55.85 58.93 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh Rubidium Strontium Yttrium Zirconium Niobium Molybdenum Technetium Ruthenium Rhodium 85.47 87.62 88.91 91.22 92.91 95.94 (98) 101.07 102.91 55 Cs 56 Ba 57 La 72 Hf 73 Ta 74 W 75 Re 76 Os 77 Ir Cesium Barium Lanthanum Hafnium Tantalum Tungsten Rhenium Osmium Iridium 132.91 137.38 138.91 178.49 180.95 183.84 186.21 190.23 192.22 87 Fr 88 Ra 89 Ac 104 Rf 105 Db 106 Sg 107 Bh 108 Hs 109 Mt Francium Radium Actinium Rutherfordium Dubnium Seaborgium Bohrium Hassium Meitnerium (223) (226) (227) (261) (262) (263) (264) (269) (268) 58 Ce 59 Pr 60 Nd 61 Pm 62 Sm 63 Eu 64 Gd Cerium Praseodymium Neodymium Promethium Samarium Europium Gadolinium 140.12 140.91 144.24 (145) 150.36 151.96 157.25 90 Th 91 Pa 92 U 93 Np 94 Pu 95 Am 96 Cm Thorium Protactinium Uranium Neptunium Plutonium Americium Curium 232.04 231.04 238.04 (237) (244) (243) (247) NCDPI Reference Tables for Chemistry (October 2006 form A-v1) Page 4 OF THE ELEMENTS 18 VIIIA 2 He 13 IIIA 14 IVA 15 VA 16 VIA 17 VIIA 4.003 5 B 6 C 7 N 8 O 9 F 10 Ne Boron Carbon Nitrogen Oxygen Fluorine Neon 10.81 12.01 14.01 16.00 19.00 20.18 13 Al 14 Si 15 P 16 S 17 Cl 18 Ar Aluminum Silicon Phosphorus Sulfur Chlorine Argon 26.98 28.09 30.97 32.07 35.45 39.95 Helium 10 VIIIB 11 IB 12 IIB 28 Ni 29 Cu 30 Zn 31 Ga 32 Ge 33 As 34 Se 35 Br 36 Kr Nickel Copper Zinc Gallium Germanium Arsenic Selenium Bromine Krypton 58.69 63.55 65.39 69.72 72.61 74.92 78.96 79.90 83.80 46 Pd 47 Ag 48 Cd 49 In 50 Sn 51 Sb 52 Te 53 I 54 Xe Palladium Silver Cadmium Indium Tin Antimony Tellurium Iodine Xenon 106.42 107.87 112.41 114.82 118.71 121.76 127.60 126.90 131.29 78 Pt 79 Au 80 Hg 81 Tl 82 Pb 83 Bi 84 Po 85 At 86 Rn Platinum Gold Mercury Thallium Lead Bismuth Polonium Astatine Radon 195.08 196.97 200.59 204.38 207.2 208.98 (209) (210) (222) 110 Ds 111 Rg 112 Uub Darmstadtium Roentgenium Ununbium (271) (272) (277) 65 Tb 66 Dy 67 Ho 68 Er 69 Tm 70 Yb 71 Lu Terbium Dysprosium Holmium Erbium Thulium Ytterbium Lutetium 158.93 162.50 164.93 167.26 168.93 173.04 174.97 97 Bk 98 Cf 99 Es 100 Fm 101 Md 102 No 103 Lr Berkelium Californium Einsteinium Fermium Mendelevium Nobelium Lawrencium (247) (251) (252) (257) (258) (254) (262) NCDPI Reference Tables for Chemistry (October 2006 form A-v1) Page 5 SOLUBILITY RULES Soluble: • All Nitrates, Acetates, Ammonium, and Group 1 (IA) salts • All Chlorides, Bromides, and Iodides, except Silver, Lead, and Mercury(I) • All Fluorides except Group 2 (IIA), Lead(II), and Iron(III) • All Sulfates except Calcium, Strontium, Barium, Mercury, Lead(II), and Silver Insoluble (0.10 M or greater): • All Carbonates and Phosphates except Group 1 (IA) and Ammonium • All Hydroxides except Group 1 (IA), Strontium, Barium, and Ammonium • All Sulfides except Group 1 (IA), 2 (IIA), and Ammonium • All Oxides except Group 1 (IA) Guidelines for Predicting the Products of Selected Types of Chemical Reaction Key: M = Metal NM = Nonmetal 1. SYNTHESIS: a. Formation of binary compound: A + B → AB b. Metal oxide-water reactions: MO + H2O → base c. Nonmetal oxide-water reactions: (NM)O + H2O → acid 2. DECOMPOSITION: a. Binary compounds: AB → A + B b. Metallic carbonates: MCO3 → MO + CO2 c. Metallic hydrogen carbonates: MHCO3 → MO+ H2O(l) + CO2(g) d. Metallic hydroxides: MOH → MO + H2O e. Metallic chlorates: MClO3 → MCl + O2 f. Oxyacids decompose to nonmetal oxides and water: acid → (NM)O + H2O 3. SINGLE REPLACEMENT: a. Metal-metal replacement: A + BC → AC + B b. Active metal replaces H from water: M + H2O → MOH + H2 c. Active metal replaces H from acid: M + HX → MX + H2 d. Halide-Halide replacement: D + BC → BD + C 4. DOUBLE REPLACEMENT: AB + CD → AD + CB a. Formation of a precipitate from solution b. Acid-Base neutralization reaction 5. COMBUSTION REACTION Hydrocarbon + oxygen → carbon dioxide + water NCDPI Reference Tables for Chemistry (October 2006 form A-v1) Page 6 ACTIVITY SERIES of Halogens: Polyatomic Ions NH F2 Cl2 Br2 I2 + 4 BrO Ammonium − 3 − CN C2H3O2− Bromate Cyanide (CH3COO−) Acetate ClO4− Perchlorate ACTIVITY SERIES of Metals ClO3− Chlorate Li Rb K Ba Sr Ca Na Mg Al Mn Zn Cr Fe Cd Co Ni Sn Pb [ H2 ] Sb Bi Cu Hg Ag Pt Au ClO2 − Chlorite ClO− IO3 − Hypochlorite Replace hydrogen from cold water Replace hydrogen from steam MnO NO − 3 Permanganate Nitrate NO2− Nitrite OH − Hydroxide HCO3− Hydrogen carbonate HSO4− SCN − CO32 − Hydrogen sulfate Thiocyanate Carbonate Cr2O72 − Dichromate CrO Replace hydrogen from acids Iodate − 4 2− 4 Chromate SO 2− 4 Sulfate SO 2− 3 Sulfite 3− 4 Phosphate PO React with oxygen to form oxides NCDPI Reference Tables for Chemistry (October 2006 form A-v1) Page 7 Bohr Model for Hydrogen Atom (measurement in nanometers) n=6 n=5 n=4 n=3 n=2 n=1 UV = Ultraviolet IR = Infrared 1282 1875 656 434 122 97 486 1094 103 410 Visible IR UV Electromagnetic Spectrum (measurement in meters) 104 103 102 101 100 10 —1 10 —2 10 —3 10 —4 10 —5 10 —6 10 —7 10 —8 10 —9 10 —10 10 —11 10 —12 10 —13 X rays Microwaves Infrared Ultraviolet Gamma rays Radio waves Shorter Wavelength Longer Wavelength Visible Light Red Orange 7.0×10—7 6.5×10—7 Yellow Green 5.9×10—7 5.7×10—7 NCDPI Reference Tables for Chemistry (October 2006 form A-v1) Stock No. 10569 Blue 4.9×10—7 Violet 4.2×10—7 4.0×10—7 Page 8