Activated Partial Thromboplastin Time
advertisement

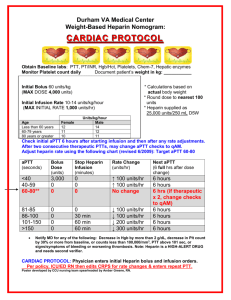
Activated Partial Thromboplastin Time Activated Partial Thromboplastin Time 1 Roger S. Riley, M.D., Ph.D. April, 2005 Feature Synonyms aPTT, APTT. Test Description The aPTT is functional determination of the intrinsic pathway of coagulation (factors XII, XI, IX, VIII, V, II, I, prekallikrein, high molecular weight kininogen). This pathway is intitated by the interaction of Factor XII with a negatively charged surface. A cascade mechanism results in fibrin production and clot formation. The aPTT is utilized to detect congenital and qcquired abnormalities of the intrinsic coagulation pathway and to monitor patients receiving heparin. Patient Preparation No specific patient preparation is required. However, since lipemia may interfere with photo-electric measurements of clot formation, specimens should not be obtained after a meal. In patients receiving intermittent heparin injections, peripheral blood for aPTT analysis should be obtained one hour before the next dose of heparin is scheduled. The specimen should not be drawn from an arm with a heparinized catheter or heparin lock. Specimen Citrated, platelet-poor plasma is used for the aPTT. Specimen Collection and Preparation Specimen requirements for coagulation assays are described in NCCLS H21-A3 (Collection, Transport, and Processing of Blood Specimens for Coagulation Assays; Approved Guideline - Third Edition, December, 1998). These requirements are summarized below. Citrated, platelet-poor plasma is prepared from peripheral venous blood collected by clean, nontraumatic venipuncture directly into a plastic or siliconized glass tube containing 109 nM (3.2%) trisodium citrate at a ratio of 9:1. With a blood collection set, the tube for coagulation analysis automatically fills to the correct volume. The tube should be immediately inverted at least four time after filling. A needle with a gauge of 22 to 19 should be utilized in adult patients, while a 21 to 23 gauge needle is suitable in pediatric patients. A traumatic venipuncture can activate coagulation factors, leading to a shortened aPTT. In patients receiving heparin, extreme care must be taken to avoid release of platelet factor 4 (PF4), which is a potent heparin inhibitor. Syringe draws - Blood obtained from a syringe draw is not preferred for aPTT analysis due to safety issues and the increased chance of specimen hemolysis or clotting. If a syringe must be used for aPTT specimen collection, a small volume syringe (< 20 mL) is recommended. The double syringe technique is recommended, with the second tube used for coagulation alalysis. The blood must be transferred from the syringe to a plastic or siliconized glass tube tube containing the proper amount of anticoagulant within one minute after collection. Indwelling catheters - Blood dilution and contamination with heparin are risks when blood specimens collected from an indwelling catheter are utilized for the aPTT. If such a specimen must be used, the line should first be flushed with saline, and the first 5 mL or six dead space volumes of the catheter drawn and discarded. Care must also be taken free the catheter and blood collection system from air leaks. Activated Partial Thromboplastin Time Specimen Collection and Preparation (Cont’D) 2 Multiple specimens - If multiple blood specimens are obtained, the aPTT should be performed on the second or third tube. If the blood is being obtained only for coagulation testing, the first tube should be drawn and discarded, and the second tube submitted to the laboratory. Specimen preparation - Platelet-poor plasma (platelet count < 10 x 109/L) is prepared from the whole blood specimen by centrifuging the capped specimen tube at an appropriate speed for an appropriate time. Centrifugation at 1500 g for 15 minutes at room temperature is recommended for this purpose. A centrifuge with a swing-out bucket rotor should be used to avoid remixing of platelets and plasma during plasma removal. Specimen storage Unheparinized specimens - Specimens for aPTT analysis from unheparinized patients should be maintained centrifuged or uncentrifuged with plasma remaining on top of the cells in an unopened tube maintained at 2-4oC or 1824oC and tested within four hours of specimen collection. Heparinized specimens - Specimens suspected of containing heparin should be centrifuged within one hour of collection and the plasma analyzed within four hours of collection. Plasma should be separated and removed within one hour if the specimen is being transported to a remote location for analysis or otherwise agitated. Frozen plasma - Plasma should be separated from specimens which cannot be analyzed within four hours and frozen at -20oC for up to two weeks or at -70oC for up to six months in a frost-free freezer. Frozen plasma specimens should be rapidly thawed at 37oC, gently mixed, and analyzed immediately or stored at 4oC for a maximum of two hours prior to analysis. Causes for Specimen Rejection - The aPTT cannot be performed on speciments that are clotted, visibly hemolyzed, collected into the wrong anticoagulant, collected into an improper quantity of anticoagulant, not labeled, or improperly labeled. Test Methodology The aPTT is usually performed by automated testing in the batch or stat mode. In the aPTT an aliquot of undiluted, platelet-poor plasma is incubated at 37oC with a particulate factor XII activator (i.e., silica, celite, kaolin, micronized silica, ellagic acid, etc.). A reagent containing phospholipid (partial thromboplastin) is added, followed by CaCl2. The time required for clot formation is measured by one of a variety of techniques (photo-optical, electromechanical, etc.). The aPTT result is reported as the time required for clot formation after the addition of CaCl2. Many different phosophlipid reagents animal and plant origin, such as cephalin, have been used as partial thromboplastins, and a variety of activating substances are in use. The sensitivity of the assay to factor deficiencies, inhibitors, and heparin varies with the reagents used in the assay. Normal Values and Critical Limits 24 - 37 sec (Jordan, C.D. et al., Normal reference laboratory values. N. Engl. J. Med. 327:718-724, 1992). Statistically, the aPTT is slightly lengthened in young individuals and slightly shortened in older populations. Premature infants have prolonged aPTT values which return to normal by 6 months of age. However, age-specific normal ranges are not utilized in patient care at this time. Interferences Lipemia and hyperbilirubinemia interfere with the detection of clot formation by photo-optical methods. The results of the aPTT may be affected by a wide variety of factors, including the manner of blood coagulation, the type of container, the type of anticoagulant, specimen transport and storage conditions, incubation time and temperature, assay reagents, and the method of end point detection. Activated Partial Thromboplastin Time Clinical Utilization 3 The PT and aPTT are the fundamental assays of the coagulation system. The principal clinical uses of the aPTT include: (1) the detection of hereditary or acquired deficiencies or defects of the intrinsic and common pathway coagulation factors (factors XII, XI, IX, VIII, prekallikrein, high molecular weight kininogen), (2) monitoring heparin anticoagulant therapy, (3) the detection of coagulation inhibitors (i.e., lupus anticoagulant), and (4) to monitor coagulation factor replacement therapy in patients with hemophilia. The aPTT is increased above the upper limit of normal with hereditary or acquired intrinsic factor deficiencies < 40% (factor VIII:C, Factor IX, Factor XI, Factor XII, vWf), lupus anticoagulants, or specific inhibitors of the intrinsic coagulation factors. Other causes of an elevated aPTT include liver disease, disseminated intravascular coagulation (DIC), heparin or anticoagulant therapy, or improper specimen collection (i.e., traumatic phlebetomy or hemolyzed specimen). References Asaf T, Reuveni H, Yermiahu T, et al: The need for routine preoperative coagulation screening tests (prothrombin time PT/partial thromboplastin time PTT) for healthy children undergoing elective tonsillectomy and/or adenoidectomy. Int J Pediatr Otorhinolaryngol 61:217-222, 2001 Bajaj SP, Joist JH: New insights into how blood clots: implications for the use of APTT and PT as coagulation screening tests and in monitoring of anticoagulant therapy. Semin Thromb Hemost 25:407-418, 1999 Bamberg R, Cottle JN, Williams JC: Effect of drawing a discard tube on PT and APTT results in healthy adults. Clin Lab Sci 16:16-19, 2003 Chen CC, You JY, Ho CH: The aPTT assay as a monitor of heparin anticoagulation efficacy in clinical settings. Adv Ther 20:231-236, 2003 Dahlback B: The multiple faces of the partial thromboplastin time APTT. J Thromb Haemost 2:2256-2257, 2004 Hirsh J, Bates S: The multiple faces of the partial thromboplastin time APTT. J Thromb Haemost 2:2254-2256, 2004 rationale of measuring APTT in risk assessment. Haematologica 86:328, 2001 tee adequate coagulation factor levels. Anesthesiology 94:542; author reply 543, 2001 Olson JD: Addressing clinical etiologies of a prolonged aPTT. CAP Today 13:28, 30, 32 passim, 1999 Tetrault G: Establishing reference ranges for PT and aPTT times. Am J Clin Pathol 113:741-742, 2000 Palkuti HS: Selecting an APTT reagent. MLO Med Lab Obs 32:14-16, 2000 van den Besselaar AM, Sturk A, Reijnierse GL: Monitoring of unfractionated heparin with the activated partial thromboplastin time: determination of therapeutic ranges. Thromb Res 107:235-240, 2002 Poller L: The multiple faces of the partial thromboplastin time APTT. J Thromb Haemost 2:2258-2259, 2004 Shetty S, Ghosh K, Mohanty D: Comparison of four commercially available activated partial thromboplastin time reagents using a semi-automated coagulometer. Blood Coagul Fibrinolysis 14:493-497, 2003 Siegel JE, Bernard DW, Swami VK, et al: Monitoring heparin therapy: APTT results from partial- vs full-draw tubes. Am J Clin Pathol 110:184-187, 1998 Smythe MA, Koerber JM, Westley SJ, et al: Use of the activated partial thromboplastin time for heparin monitoring. Am J Clin Pathol 115:148-155, 2001 Liepman CI, Koerber JM, Mattson JC, et al: Comparing methods of establishing the aPTT therapeutic range of heparin. Ann Pharmacother 37:794-798, 2003 ten Boekel E, de Kieviet W, Bartels PC: Subjects with a shortened activated partial thromboplastin time show increased in-hospital mortality associated with elevated D-dimer, C-reactive protein and glucose levels. Scand J Clin Lab Invest 63:441-448, 2003 Lippi G, Franchini M, Brazzarola P, et al: Preoperative screening: the Teruya J, Oropeza M, Ramsey G: A normal aPTT does not guaran- White GC, 2nd: The partial thromboplastin time: defining an era in coagulation. J Thromb Haemost 1:2267-2270, 2003 Wojtkowski TA, Rutledge JC, Matthews DC: The clinical impact of increased sensitivity PT and APTT coagulation assays. Am J Clin Pathol 112:225-232, 1999