Chapter 10 (Hill/Petrucci/McCreary/Perry Bonding Theory and
advertisement

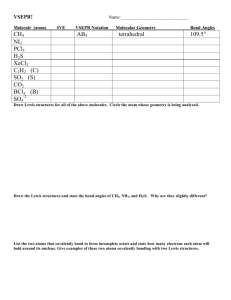
Chapter 10 (Hill/Petrucci/McCreary/Perry Bonding Theory and Molecular Structure This chapter deals with two additional approaches chemists use to describe chemical bonding: valence-shell electron pair repulsion (VSEPR) theory and hybrid orbitals (valence bonding). Lewis dot structures give us limited information about the types of bonds between atoms in molecules, but tell us nothing about the 3-D shapes of the molecules. VSEPR and valence bond structures give us the “molecular geometry” (shape of the mo lecules). “Precise molecular geometry can be determined only by experiment but the shapes of many molecules and polyatomic ions can be predicted fairly well …” (Hill, p. 388) “As the name implies, the valence-shell electron pair repulsion method is based on the idea that pairs of valence electrons in bonded atoms repel one another” … and “push electron pairs as far from one another as possible”… (Hill, p. 388) Molecular Geometries (see Figure 10.1, p. 388, Hill) The molecular geometry for a molecule is its 3-D structure as represented by various kinds of 3D models …These 3-D models for O2, CO2 and H2O are called “space- filling” models … as much as possible, they approximate the actual shape of the molecules and of their electron clouds… The Water Molecule (right margin, p. 389, Hill) The central oxygen atom has two lone pairs and two bonded pairs of electrons Central atom = A Bonded pairs = X Lone pairs = E VSEPR notation: AX2 E2 Lewis Dot Structures for Methane, CH4 The central C atom is bonded by single bonds (-) to 4 individual H atoms The VSEPR Method VSEPR: an approach used to determine/describe the 3-D shape of molecules by arranging the pairs of valence electrons around a “central atom” so that they are as far apart from each other as possible Consider a molecule like CH4 … each H-atom is bonded directly to the central C-atom …Question: what shape is the best way to arrange four “groups” (here, four H-atoms) around a central atom (C-atom) so they are as far apart from each other as possible? not a square !! So, then what shape if not a square?? A tetrahedron … H-atoms located at the corners or apices of a tetrahedron…a three-sided pyramid … H–C–H bond angles are all 109.50 in this structure. VSEPR Geometries group: an atom or a group of atoms, attached directly to a central atom in a chemical structure by one, two, or three electron pairs (:) one pair (:) = a single covalent bond two pairs (::) = a double covalent bond three pairs (:::) = a triple covalent bond Begin by visualizing the surface of a sphere, with a central atom at the center of the sphere What’s the best way to arrange two groups around a central atom? a straight line .. linear geometry. A Linear Molecule: BeCl2 VSEPR notation: AX2 Another Linear Molecule: CO2 Again, VSEPR notatio n: AX2 What’s the best way to arrange three groups around a central atom? Trigonal planar (or planar triangular)… trigonal planar geometry has bond angles of 1200 . If all “X” atoms are the same, the bond lengths should also be the same. A Trigonal Planar Molecule: BF3 VSEPR notation: AX3 Another Trigonal Planar Molecule: SO3 VSEPR notation: AX3 Trigonal Planar Electron Group Geometry: AX2 E AX2 E …the presence of the lone pair means that the molecular geometry is angular or bent What’s the best way to arrange four groups around a central atom? As seen earlier, at the corners of a tetrahedron! tetrahedral geometry. VSEPR notation: AX4 Tetrahedral geometry has bond angles of 109.50 . If all “X” atoms are the same, the bond lengths from X to the central atom should also be the same. Tetrahedral Electron Group Geometry: AX3 E AX3 E …the presence of the lone pair means that the molecular geometry is trigonal pyramidal Tetrahedral Electron Group Geometry: AX2 E2 AX2 E2 …the presence of the two lone pairs means that the molecular geometry is angular What’s the best way to arrange five groups around a central atom? an equilateral triangle with a group above and a group below the triangular (“equatorial”)plane! AX5 trigonal bipyramidal geometry has bond angles of 900 ,1800 and 1200 . “equatorial positions” are in the triangular plane “axial positions” are above or below the triangular plane Trigonal Bipyramidal Electron Group Geometry: AX4 E AX4 E …the presence of the lone pair means that the molecular geometry is “seesaw” Note that the lone pair goes into the equatorial plane, not into one of the axial positions! Trigonal Bipyramidal Electron Group Geometry: AX3 E2 AX3 E2 …the presence of the two lone pairs means that the molecular geometry is “T-shaped” Note that the lone pairs again both go into the equatorial plane, not into the axial positions! Trigonal Bipyramidal Electron Group Geometry: AX2 E3 AX2 E3 …the presence of the three lone pairs means that the molecular geometry is linear Note that now all three lone pairs again go into the equatorial plane, not the into axial positions! What’s the best way to arrange six groups around a central atom? a square with a group above and a group below the square, “equatorial” plane! AX6 Octahedral geometry has bond angles of 900 and1800 . “equatorial positions” are in the square; “axial positions” are above or below the square Octahedral Electron Group Geometry: AX5 E AX5 E …the presence of the lone pair means that the molecular geometry is “square pyramidal” Note that now the lone pair goes into an axial position, not into the equatorial plane! Octahedral Electron Group Geometry: AX4 E2 AX4 E2 …the presence of the two lone pairs means that the molecular geometry is “square planar”… Note that here the lone pairs again both go into the axial positions, not into the equatorial plane! What Is This Molecular Geometry? (for :NH3 ) Answer: trigonal pyramidal (AX3 E) What Is This Molecular Geometry? (for H-O-H)) Answer: angular (AX2 E2 ) Summary of structures for linear and trigonal plana r geometries Octahedral (“square pyramid”) (AX5 E) … e.g. BrI5 Octahedral (“square planar”) (AX4 E2 )… e.g. XeF4 Octahedral (AX6 ) … e.g. SF6 Octahedral (“linear”) (AX2 E4 ) … e.g. XeF2 Trigonal bipyramid (“T-shaped”) (AX3 E2 ) Trigonal bipyramid (“seesaw”) (AX4 E) Trigonal bipyramid (AX5 ) Summary of VSEPR Principles Repulsive forces increase as electron pairs are placed closer together. Lone-pair electrons “spread out” more than bonding- pair electrons, e.g. bond angles in H2 O and NH3 are smaller than 109.50 because the lone pairs take up more room and “squeeze” the H atoms closer together. Molecular Polarity and Dipole Moments dipole moment: a measure of the unequal charge distribution in a molecule µ = δ x d = debyes (D) When µ = 0, a molecule is perfectly nonpolar (NP); otherwise it is polar (P) Example. CH4 Example. HCl µ = 0.00 D µ = 1.07 D nonpolar (NP) polar (P) Representation of Dipole Vectors for Linear CO2 (see text figure, top of p. 400) The dipole “vectors” are equal in magnitude but exactly opposite in direction so they cancel each other and the net molecular dipole moment is 0. Representation of Dipole Vectors for Angular H2 O (see text figure, middle of p. 400) The dipole “vectors” are equal in magnitude but not opposite in direction so they don’t cancel each other and the net molecular dipole moment is not 0. Polar or Nonpolar? (see and review Example 10.4, pp. 400-401) Lewis Structures for NOF and NO2 F (see and review Example 10.5, pp. 401-402) NF3 and Ammonia (NH3 ) Same geometry … why different dipole moments? N-F ∆Χ = 4.0 – 3.0 = 1.0 and N-H ∆Χ = 3.0 – 2.1 = 0.9 VALENCE BOND (VB) THEORY A third approach to molecular geometry … valence bond theory: a covalent bond involves the overlap of atomic orbitals on two different atoms, each atom contributing one electron to the bond itself … region of overlap is the covalent bond and has a high electron density… In the valence bond approach, only valence electrons participate in the formation of a covalent bond … Valence Bond Explanation for Bonding in H2 (see Figure 10.10, p.403) Overlap of two 1s orbitals – one from each H atom participating in the bond! Valence Bond Explanation for Bonding in H2 S (see Figure 10.11, p.403) - the three 3p orbitals are mutually “orthogonal” - S-H Bonds result from overlap of 1s orbitals on the H atoms with half- filled 3p orbitals on the S atom (predicted bond angle is 900 ) Explanation of sp3 Hybridization for a C Atom (see Figure 10.12, p.405) Note: shape of sp3 hybrid orbital Another way to Explain sp3 Hybridization for Carbon s + 3 p " 4 sp3 hybrid orbitals sp3 Hybridization in N and O (see discussion, pp. 405-406) Hybridization and Bonding in CH4 (see Figure 10.13, p.406) Hybridization and Bonding in NH3 (see Figure 10.14, p.406) Hybridization for Boron in BF3 (see Figure 10.15, p.407) Need 3 energetically equivalent orbitals for B-F bonds B atom hybridization: s + 2 p " 3 sp2 hybrid orbitals Explanation of sp2 Hybridization for B (see text figure, bottom of p. 406) Shape of sp2 Hybrid Orbital (see text figure, bottom left, p. 406) Hybridization for Be, e.g. in BeH2 (see Figure 10.16, p.407) Need 2 energetically equivalent orbitals for B-H bonds Be atom hybridization: s + 1 p " 2 sp hybrid orbitals Explanation of sp Hybridization for Be (see text figures, middle of p. 407) Shape of sp Hybrid Orbital (see text figure, middle right, p. 407) Explanation of sp3 d Hybridization for P in PCl5 (see text figure, upper middle, p. 408) Shape of sp3 d Hybrid Orbital: (see Figure 10.17, p.408) Opportunity for up to 5 bonds, one with each “lobe” Explanation of sp3d2 Hybridization for S in SF6 (see text figure, lower middle, p. 408) Shape of sp3 d2 Hybrid Orbital: (see Figure 10.18, p.408) Opportunity for up to 6 bonds, one with each “lobe” IF5 (see and review Example 10.6, pp. 409-410 … see Figure 10.19, p.410) Hybridization and Bonding for IF5 Central I atom has 6 energetically equivalent sp3d2 hybrid orbitals available for bonding … 5 with F atoms and one for a lone pair on the I atom. Multiple Covalent Bonds and Hybrid Orbitals Lewis dot structure for ethylene, C2 H4 (see text figures, upper and middle, p. 411) Lewis Structures and VSEPR Interpretation for C2 H4 (see Figure 10.20, p.411) Review caption for Figure 10.20! Summary of VB Bonding in C2 H4 (see Figure 10.21, p.412) Note text description of types of orbital overlap in bonds in C2 H4 Sigma Bonds and Pi Bonds sigma (σ) bond: a bond in which electron density is concentrated in the region (along the internuclear axis) between the two nuclei in a chemical bond – can form by end-to-end overlap of unhybridized p- orbitals or overlap of lobes of adjacent hybrid orbitals on two different atoms pi (π ) bond: a bond oriented along the internuclear axis in which the overlap results from sideto-side overlap of unhybridized p- or d-orbitals single bond = 1 σ bond double bond = 1 σ bond + 1 π bond triple bond = 1 σ bond + 2 π bonds Sigma Bonds and Pi Bonds Sigma bond from end-to-end overlap of two p-orbitals Sigma bond from end-to-end overlap of s orbital with p-orbital Pi bond from side -to-side overlap of two p-orbitals Overlap of sp2 Hybrid Orbitals to Form a s Bond Two sp2 hybrid orbitals s bond from sp2 hybrid overlap with another sp2 hybrid Hybridization Scheme for C2 H2 (see text figures, lower middle, p. 413) Summary of VB Bonding in C2 H2 (see Figure 10.22, p.413) Note text description of types of orbital overlap in bonds in C2 H2