Lewis Structures: A Chemistry Guide
advertisement
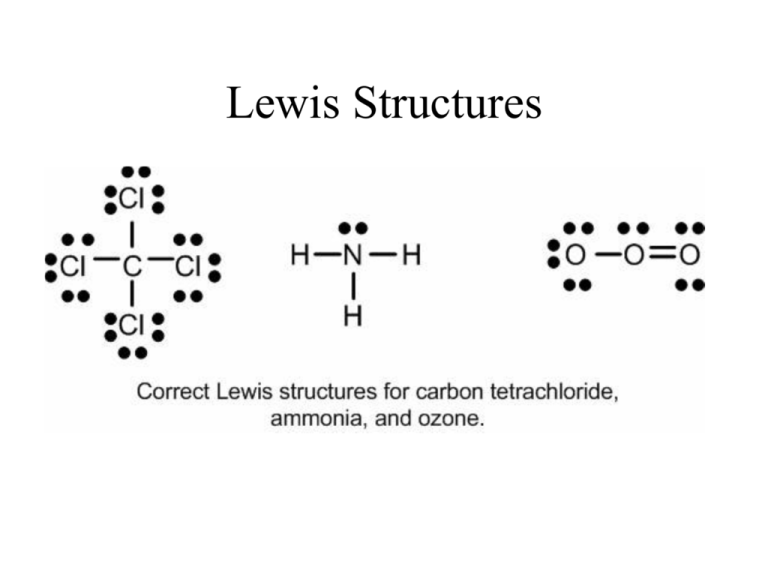
Lewis Structures Valence electrons for Elements Recall that the valence electrons for the elements can be determined based on the elements position on the periodic table. Lewis Dot Symbol Valence electrons and number of bonds Number of bonds elements prefers depending on the number of valence electrons. In general - → Fa m i l y H alo g e n s F , B r, C l , I C alco g e n s O, S N i t ro g e n N, P C a rb o n C, Si X # C o v a l e n t B o n d s* 1 bond O → → N → 3 bond often C → 4 bond often 2 bond often always The above chart is a guide on the number of bonds formed by these atoms. Lewis Structure, Octet Rule Guidelines When compounds are formed they tend to follow the Octet Rule. Octet Rule: Atoms will share electrons (e-) until it is surrounded by eight valence electrons. 4 unpaired 3unpaired 2unpaired 1unpaired up = unpaired e- 4 bonds 3 bonds 2 bonds 1 bond O=C=O N≡ N O=O F-F Atomic Connectivity The atomic arrangement for a molecule is usually given. CH2ClF Cl H C H HNO3 O F N O H CH3COOH O H H C H H2SO4 O C O H O S O H O H O In general when there is a single central atom in the molecule, CH2ClF, SeCl2, O3 (CO2, NH3, PO43-), the central atom is the first atom in the chemical formula. Except when the first atom in the chemical formula is Hydrogen (H) or fluorine (F). In which case the central atom is the second atom in the chemical formula. Find the central atom for the following: 1) H2O 2) PCl3 3) SO3 4) CO32- 5) BeH2 6) IO3- Setting up Bond Table Bond table requires the chemical formula, and determining the number of electrons around each atom. A) Chemical Formula I.e., HNO3 B) Oe - Octet Electrons. This is always either 8 or 2 (for H) C) Tve - Total Valence Electron. ChemFormula HNO3 H N O Octet eOe Tot Val eTve 1x2=2 1x1=1 1x8=8 1x5=5 3 x 8 = 24 3 x 6 = 18 34 24 Note this recipe works only if the chemical specie obeys the octet rule. For chemical specie which violates the octet rule, this method must be modified. Calculating the Number of Bonds and the Remaining electrons After setting up the bond table, calculate the number of bonds in the chemical specie and the number of electrons. The remaining electrons are place around the atoms in the chemical specie such that each atom obeys the octet rule HNO3 Oe Bond Table # of Bonds 34 Remaining e- Tve - 24 # Bonding e= 10 10/2 = 5 Tve(24) - electrons in Bond (10) = 14 Putting it Together 1) Chemical formula: HNO3 3) Number of bonds 2) Atomic sequence: =5 O N O O H 4) Remaining electrons = 14 5) Lewis Structure with 5 bonds: O N O H O 6) Complete Lewis Structure with 14 remaining electrons O N O H O Lewis Dot Structure of CO2 by Bonds Table A. Calculate Octet electrons (Oe-) and Total Valence electrons to determine number of bonds CO2 1C 2O Chg Oe 1•(8)= 8 2•(8)=16 B. Calculate the number of bonds in compound structure. # bonds = TVe 1•(4) = 4 2•(6)=12 24 (Oe - TVe) 2 = (24- 16) = 2 8 = 4 bonds 2 C. Calculate the remaining electrons to add to structure to complete Lewis dot structure. 16 Remaining e- = TVe - e- used in bonding. = 16 - 8 = 8 e Remaining Writing Lewis Structure: Use information from the above calculations. Total of 16e- in CO2, of which 8 electrons are used to form 4 bonds and 8 remaining electrons are used to complete Lewis structure. - O C O O C O 1,2. Write atom connectivity 3,4,5. Draw the four bonds in the structure. for CO2. O C O 6. Place the remaining 8 electrons in the structure to complete the Lewis Structure Lewis Dot Structure of ClO4A. Calculate (Oe-) and (TVe) ClO4- Oe 1 Cl 1•(8)= 8 4O 4•(8)= 32 Chg 40 B. Number of Bonds. # bonds = TVe 1•(7) = 7 4•(6)= 24 1 32 (40- 32) = 8 = 4 bonds 2 2 C. Remaining electrons. Remaining e- = 32 - 8 = 24 e-Remaining Writing Lewis Structure: 1,2. Write atom connectivity for ClO4-. O O Cl O O 3.4.5. Draw the four bonds in the structure. O O O Cl O 6. Place the remaining 24 electrons in the structure such that each atom has an octet to complete the Lewis Structure O O Cl O O Summary Lewis Structure Determination: • Molecular Formula • Atomic Sequence (H and F are terminal) • Determine the # of bonds Oe- and TVe# of Bonds = (Oe - TVe-) / 2 • Determine remaining electrons Re = (TVe-) - (# e- in bonding) • Make sure all atoms satisfy octet rule (Except H which is satisfied with 2 electrons) Rules for molecules with an overall charge: 1. When figuring out the number of electrons available (ie. counting the valence electrons), add or subtract electrons as indicated by the charge. 2. Create the Lewis structure the same as always 3. Put [square] brackets around the structure 4. Write the charge as a superscript Try it: CO3-2 C: 4 e- , O: 6 x 3 e- , (-2): e- = total 24 e- O O C O Try it: CO3-2 C: 4 e- , O: 6 x 3 e- , (-2): e- = total 24 e- O O C O Try it: CO3-2 C: 4 e- , O: 6 x 3 e- , (-2): e- = total 24 e- O O C O Try it: CO3-2 All 24 e- have been used, buy C does NOT have a complete octet….. Need to make a double bond….Does it matter which O it makes the double bond with? O O C O Try it: CO3-2 The bond can go here but it could also go between the C and either of the other Os O O C O Finished Product J -2 C C C Resonance Structures • The Lewis structure for CO3 shows two different types of bonds, single and double. • Double bonds also have a shorter bond length, the distance between the nuclei of the two atoms in the bond, than single bonds do. Thus, if the above Lewis structure for carbonate were correct, the carbonate ion would have one bond that is shorter and stronger than the other two. • This is not the case. All three of the bonds in the carbonate ion are the same strength and the same length. • Interestingly, the behaviour of the bonds suggests they are longer than double bonds and shorter than single bonds. • They are also stronger than single bonds but not as strong as double bonds. Resonance Structures (2) • So the carbonate ion would actually 3 bonds, with the bond lengths between the characteristic single and double bond lengths • The important points to remember about resonance forms are: àThe molecule is not rapidly oscillating between different discrete forms àThere is only one form of the each molecule, we need to draw two (or more) Lewis structures because a single structure is insufficient to describe the real structure Drawing them: • Expressing resonance when drawing Lewis structures is done by drawing each of the possible resonance forms and placing double-headed arrows between them Do you remember polyatomic ions? Formula Name Formula Name NH4+/NH3 Ammonium/Ammonia CrO4-2 Chromate C2H3O2- Acetate Cr2O7-2 Dichromate CH3COO- Acetate MnO4- Permanganate CN- Cyanide MnO4-2 Manganate CO3-2 Carbonate NO2- Nitrite HCO3- Bicarbonate NO3- Nitrate C2O4-2 Oxalate OH- Hydroxide ClO- Hypochlorite PO4-3 Phosphate ClO2- Chlorite SCN- Thiocyanate ClO3- Chlorate SO3-2 Sulfite ClO4- Perchlorate SO4-2 Sulfate S2O3-2 Thiosulfate HSO4- Hydrogen sulfate BrO- Hypobromite IO3- Iodate AsO2-3 Arsenite SeO4-2 Selenate BrO3- Bromate HSO3- Hydrogen sulfite Example Lewis Structures: Examples a) CH2ClF Cl H C H F b) SO2 O S O c) SO42- O O S O O d) H3PO4 O H O P O H O H Co-ordinate Covalent Bond • A covalent bond is formed by two atoms sharing a pair of electrons. The atoms are held together because the electron pair is attracted by both of the nuclei. • In a simple covalent bond, each atom supplies one electron to the bond - but that doesn't have to be the case. • A co-ordinate bond is a covalent bond (a shared pair of electrons) in which both electrons come from the same atom. Example: NH4+ Carbonate ion: -2 C C C Try these: 1. 2. 3. 4. 5. O3 NO3-1 HCO2-1 CN-1 SO32- 6. BrO-1 7. PO438. IO3-1 9. CHO2 -1 10. NO2+1