Chapter 12
advertisement

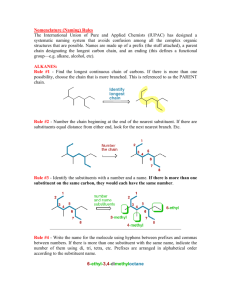
Saturated hydrocarbons Chapter 12 Coming up… • Ch-12: saturated hydrocarbons (alkanes, cycloalkanes) • Ch-13: Unsaturated hydrocarbons (alkenes, alkynes, aromatics) • Ch-14: Alcohols, phenols, ethers • Ch-15: Aldehydes and ketones • Ch-16: Carboxylic acids, esters, acid derivatives • Ch-17: Amines and amides • Ch-18: Carbohydrates • Ch-19: Lipids • Ch-20: Proteins Organic and inorganic compounds • Organic chemistry concerns the chemistry of carbon compounds contain C and H, but also maybe other p-block elements. • At the time of printing (Stoker, 5th edition), there were around 10 million organic compounds catalogued • Inorganic chemistry concerns the chemistry of the other 117 elements. Around 1.7 million of these are known Non-polar hydrocarbon tail ~1.7 million A polar, charged group ~10 million Organic and inorganic compounds • The reason why there are so many organic compounds is that carbon is very good at forming bonds with other carbon atoms. • Carbon atoms are commonly found in chain-like arrangements or C-rings (or both within the same molecule). • Carbon has four valence electrons. In organic compounds, it forms four covalent bonds to obtain an octet. C four single bonds C double bond, 2 single bonds C triple bond, single bond two double bonds Hydrocarbons and hydrocarbon derivatives • Hydrocarbons are compounds that contain only carbon and hydrogen in their formulas. • Two basic categories of hydrocarbon: – Saturated hydrocarbons: all carbon atoms are connected together with single bonds – Unsaturated hydrocarbons: involve one or more multiple (double, triple) C-C bonds • Hydrocarbon derivatives contain carbon and hydrogen, and one or more other elements (P, N, O, Cl, etc.) Hydrocarbons and hydrocarbon derivatives • Saturated hydrocarbons may be found in two possible formats: an acyclic, 6-C chain a cyclic 6-C structure carbon skeletal structures (these are not complete Lewis structures) Alkanes: acyclic saturated hydrocarbons • An alkane is a saturated hydrocarbon that is acyclic (does not possess ring-structure). • Because all C-C bonds are single bonds (and because the other bonds that carbon needs to get an octet are to H-toms), alkanes have the general formula CnH2n+2 (n = # of C-atoms) Examples of alkanes: CH4 C2H6 C3H8 Alkanes: acyclic saturated hydrocarbons • In an alkane, each carbon is tetrahedral (it has four bonds to other atoms. Rem: VSEPR) CH4 C2H6 C3H8 Alkanes: acyclic saturated hydrocarbons • Chemical formulas for alkanes are written as CnH2n+2; however, structural formulas give more information. – Chemical formula reveals the type and number of each element in the compound – Structural formulas show how each atom in the molecule is connected expanded structural formula condensed structural formula CH4 CH3-CH3 CH3-CH2-CH3 CH4 C2H6 C3H8 name methane ethane propane molecular formula Alkanes: acyclic saturated hydrocarbons • For longer carbon chains, an abbreviated, condensed structural formula is advantageous, as it shows most of the information of the expanded formula without taking up as much space CH3-CH2-CH2-CH2-CH2-CH2-CH2-CH3 CH3-(CH2)6-CH3 An 8-carbon chain (two CH3-groups linked by a 6-carbon chain (6-CH2- units) 6 –CH2- units between two CH3-groups Alkanes: acyclic saturated hydrocarbons • Sometimes, a simple skeletal structural formula can be used to convey hydrocarbon structure (these are usually not used) mean the same thing Alkane isomerism • The types of alkanes we’ve considered so far involve “straight chain” types, where the carbon atoms form a continuous series (i.e. no branches). • When alkanes having four or more carbons are considered, there is more than one structural formula that can be used to describe a given molecular formula. Alkane isomerism • The formula C4H10 can be represented by the following condensed structural formulas: butane (or n-butane) isobutane C4H10 These compounds possess the same chemical formula, but differ in the way the atoms are arranged (isomers) “isotopes” sounds like “isomers” – they don’t mean the same thing. Isotopes are nuclides of the same element that possess different numbers of neutrons (e.g. 12C and 13C). Alkane isomerism • The C4H10 shown on the left is called a continuous chain alkane (or an unbranched/”straight-chain” alkane). • The one on the right is called a “branched-chain” alkane butane (or n-butane) isobutane These are called “constitutional isomers” which differ in their atom-to-atom connectivity In every case, the two (or more) molecules that are being compared have the same molecular formula, but differ in their structures, somehow. Alkane isomerism • As the number of carbon atoms in the alkane grows, so do the number of possible isomers. C5H12 pentane isopentane neopentane Conformations of alkanes all single bonds • The carbon-carbon bonds in alkanes permit rotation of each carbon-group with respect to the others that are chemically bound to it. C4H10 Conformations of alkanes • Conformations are specific, 3-dimensional arrangements of atoms in organic molecules (at some instant) that result from rotation about C-C single bonds. • Several conformations of a six-carbon chain are shown using the skeletal structures below: All the same molecule: C6H14 Conformations of alkanes • Note that the following two skeletal structures describe two different alkanes: Alkane on the left is a 6-carbon, continuous chain structure. Alkane on the right is a branched structure (a 5-carbon, continuous chain that has a 1-carbon branch) Conformations of alkanes • Do the following pairs of condensed structural formulas describe the same alkane? a) b) c) IUPAC nomenclature for alkanes • The names that have been shown for the branched alkanes so far are common names (made as these compounds were identified). • As the number of organic compounds catalogued grew, a system for naming was developed by the International Union of Pure and Applied Chemistry (IUPAC). • The basic system used is one that employs a prefix-type name. IUPAC nomenclature for alkanes • Names for continuous chain alkanes (first ten) are shown below. The names use a prefix (e.g. meth-) to designate the number of carbon atoms in the chain. Alkanes have ”ane” at Memorize these prefixes the end of their name Prefix MethEthPropButPentHexHeptOctNonDec- (1) (2) (3) (4) (5) (6) (7) (8) (9) (10) IUPAC nomenclature for alkanes • Branched-chain alkanes can be described as continuous-chain alkanes with branches (substituents). • The IUPAC system of naming branched-chain alkanes describes the type and location of substituents before the name of the longest, continuous chain of carbon atoms in the alkane. Substituents are the “branches” in branched-chain alkanes. They are atoms (or groups of atoms for the kind we’ll look at first) that hang off the main carbon chain. IUPAC nomenclature for alkanes • Substituents in branched-chain alkanes are called alkyl groups. An alkyl group is the group of atoms that would be created by removing a hydrogen atom from an alkane. They are named according to the alkane from which they are derived. methane a "methyl" substituent To get the substituent name: take the alkane name and replace the “ane” part with “yl” propane a "propyl" substituent IUPAC nomenclature for alkanes • To name a branched alkane, follow these steps: 1) Identify the longest, continuous carbon chain in the structure. This will be the base of the branched alkane’s name. a 4-carbon, continuous chain So far, we know this compound is going to be called some kind of butane IUPAC nomenclature for alkanes 2) Number this chain in a way that gives the carbon(s) with the substituent the lowest possible, overall numbering. 1 2 3 4 The methyl substituent is thus located on C-2 (carbon-2) (something something butane) IUPAC nomenclature for alkanes • After locating the alkyl substituent by number, prefix the parent-chain alkane name (the longest, continuous carbon chain) with the number and the name of the substituent: 1 2 3 4 The “2” in this case is actually redundant, because if a methyl substituent were on any other carbon except carbon #2, the longest chain of carbon atoms would be 5, making the molecule a pentane. 2-Methylbutane Separate the number from the substituent name with a hyphen, and the last substituent name reads directly into the parent chain alkane name IUPAC nomenclature for alkanes • Another example 1) Find the longest, continuous chain of C-atoms 2) Number them in a way that gives all substituents the lowest total numbering 3) Prefix the name of the parent alkane with the number and name of the substituent IUPAC nomenclature for alkanes • One with multiple substituents: 2,3,4-Trimethylhexane In cases where multiple substituents of the same type are present, prefix the substituent name with di-, tri-, tetra-, etc. to indicate how many of them are present IUPAC nomenclature for alkanes • If more than one kind of substituent is present, the alphabetic order of the substituents take priority over the number of the substituent when numbering the parent chain Separate different substituents with hyphens 3-Ethyl-2-methylhexane (not 2-Methyl-3-ethylhexane) (come back to this structure later) 3-Ethyl-4,5-dipropyloctane The prefix part of the propyl substituents are not counted for alphabetical ordering IUPAC nomenclature for alkanes • IUPAC punctuation rules: 1. Separate numbers from letters with hyphens 2. Separate numbers from other numbers with commas 3. Don’t separate the last substituent name from the parent alkane chain 2 4-Ethyl-2,3-dimethyl-5-propylnonane 1 3 Line-angle structural formulas for alkanes • Line-angle structural formulas describe carboncarbon bonds with straight lines (each point in the diagram represents a carbon atom with four bonds to carbon(s) and hydrogen(s) around it) = = It is understood that each C-atom has four bonds; C-H bonds are there, but not shown Classification of carbon atoms • The carbon atoms in organic structures are classified as primary, secondary, tertiary, or quaternary, depending on the number of other carbon atoms bound to them. – – – – Primary (1o) C: bounds to one other C-atom Secondary (2o) C: bound to two other C-atoms Tertiary (3o) C: bound to three other C-atoms Quaternary (4o) C: bound to four other C-atoms 1o 4o 2o 3o Branched-chain alkyl groups • Sometimes, branched-chain substituents are encountered. These are named according to the parent alkane from which they are derived. Substituent derives from a 4-C alkane Substituent derives from a 3-C alkane (propane) and point of attachment is a secondary C of the substituent 4-Isopropyloctane could also call this 4-sec-Propyloctane (butane) and point of attachment is a tertiary C of the substituent 4-tert-Butyloctane Branched-chain alkyl groups …another point: for the purposes of capitalization (at the beginning of the name), tert- and sec-are not capitalized, but iso is 4-Isopropyloctane 4-tert-Butyloctane Branched-chain alkyl groups • Given a choice between unbranched substituents and branched substituents, should use unbranched ones for naming Preferable to call this 3-Ethyl-2-methylhexane also, not 3-Isopropylhexane Cycloalkanes • Cyclic alkanes (cycloalkanes) are alkane chains where the end carbons are linked together (need to kick off 2 H atoms from the formula of the corresponding straight-chain alkane to get the cycloalkane formula). • The general formula for a cycloalkane is CnH2n Cyclopropane Cyclooctane Cyclohexane Cyclobutane Cyclopentane Cycloheptane = C6H12 C6H12 Cyclononane Cycloalkanes IUPAC nomenclature for substituted cycloalkanes • Naming: If one substituent exists on a cycloalkane, no numbering is needed to denote its location Ethylcyclohexane IUPAC nomenclature for substituted cycloalkanes • If two substituents are present, the ring is numbered follows alphabetic priority. • If more than two substituents are present, the ring numbering is assigned in a way that gives the lowest overall substituent numbers (order they are reported in is still alphabetic) always ensure the lowest overall numbering is followed 1-Ethyl-2-methylcyclohexane 2-Ethyl-1-methyl-4-propylcyclohexane (not 1-Ethyl-2-methyl-5-propylcyclohexane or 1-Methyl-2-ethyl-4-propylcyclohexane) Isomerism in cycloalkanes • Constitutional isomers are possible for cycloalkanes having four or more carbons: C4H8 Cyclobutane C4H8 Methylcyclopropane These isomers differ in the way the carbon atoms are connected together (constitutional isomers) Isomerism in cycloalkanes • As before, as the number of carbons in the (cyclo)alkane grows, so do the number of constitutional isomers. C5H10 Cyclopentane C5H10 Methylcyclobutane C5H10 1,2-Dimethylcyclopropane C5H10 Ethylcyclopropane Isomerism in cycloalkanes • Another kind of isomerism we haven’t yet encountered, called stereoisomerism, involves molecules that have the same molecular formula, same atom-to-atom connectivity, but differ in the 3-dimensional arrangement of the atoms in space. • In cycoalkanes, there may exist the possibility of cis-, trans- isomers cis-1,2-Dimethylcyclopentane CH3-substituents are both above the C-C bond of cyclopentane trans-1,2-Dimethylcyclopentane One CH3-substituent is above the C-C bond of cyclopentane and the other one is below it Isomerism in cycloalkanes • There are two distinct molecules. One can’t be converted into the other without breaking bonds first. • Can have this form of isomerism for any cycloalkane that has more than one substituent. trans-1-Ethyl-2-methylcyclohexane cis-1-Ethyl-2-methylcyclohexane Isomerism in cycloalkanes • Substituents also don’t need to be on adjacent carbon atoms of the ring (but can’t be on the same carbon atom of the ring) trans-1-Ethyl-3-methylcyclohexane cis-1-Ethyl-3-methylcyclohexane Sources of alkanes and cycloalkanes • The crude petroleum that is obtained at drilling sites is a mixture of hydrocarbons (cyclic and acyclic) that is purified (refined) by taking advantage of the boiling point differences of the various components Sources of alkanes and cycloalkanes • Boiling point is observed to increase with increasing chain Cchain length (and ring size for cycloalkanes). • About a 30o increase per additional C (–CH2- unit) in the chain. Increasing London forces of attraction Physical properties of alkanes and cycloalkanes 1. 2. 3. Alkanes and cycloalkanes are waterinsoluble Alkanes and cycloalkanes have densities that are less than that of water (0.6 – 0.8 g/mL, as compared to ~1 g/mL for H2O) Boiling points of continuous chain alkanes and cycloalkanes increase with an increase in carbon-chain length or ring size Boiling point for alkanes having a given number of C-atoms: cycloalkane > “straight” chain > branched chain • • Cycloalkanes have higher boiling points than corresponding alkanes because they are more rigid Branched chain alkanes have lower boiling points because they are more compact and have less surface areas (less interaction with other, similar molecules) than straight-chain forms Why is something like this important in life science? • Cholesterol is used by the body for – – – – • building cell membranes making bile vitamin D steroid horomones cholesterol, (mainly a hydrocarbon) Cholesterol is transported through the bloodstream by lipoproteins. These shell structures have a water-unfriendly core and a water-friendly exterior Low-Density Lipoprotein • • LDL is a lipoprotein that has mostly cholesterol content. HDL has mostly protein content High-Density Lipoprotein LDL is used to transport cholesterol to sites in the body, through the bloodstream. http://www.answers.com/topic/lipoprotein Why is something like this important in life science? • If LDL transports cholesterol to a site (e.g. tissue) that doesn’t require cholesterol, the LDL remains in the bloodstream, eventually releasing its contents. • Cholesterol molecules aren’t watersoluble, so they aggregate, forming insoluble clumps that collect along the arterial walls (atherosclerosis). Main forces of attraction between cholesterol molecules are London forces, which increase with molecular weight. • This restricts blood flow and can eventually create a blockage (stroke/heart attack). Essentials of general, organic, and biochemistry. D. Guinn, R. Brewer, W.H. Freeman, NY, 2010. Why is something like this important in life science? • The condition (depending on the stage) can be corrected by: • diet/exercise • medication • angioplasty Essentials of general, organic, and biochemistry. D. Guinn, R. Brewer, W.H. Freeman, NY, 2010. Chemical properties of alkanes and cycloalkanes • Alkanes and cycloalkanes have low chemical reactivities. The C-C bonds and C-H bonds are non-polar, which do not encourage reactions with other species, and the bond strengths are fairly high (strong bonds) • Two reactions that they are susceptible to are combustion and halogenation Chemical properties of alkanes and cycloalkanes • In a combustion reaction, alkanes and cycloalkanes are reacted with O2 to form CO2 in an oxygen-rich environments (or CO or other C-products in less O2rich environments). • Some examples of alkane combustion reactions: CH4 + 2O2 CO2 + 2H2O + heat 2C6H14 + 19O2 12CO2 + 14H2O + heat Nomenclature and properties of halogenated alkanes • Halogenated alkanes (or haloalkanes) are hydrocarbons (or their derivates) that possess at least one halogen atoms • Naming rules – Halogens are treated just like other (alkyl) substituents when numbering and alphabetic naming are considered – Substituents are called fluoro-, chloro, bromo-, and iodofor the purposes of assigning names 2-Chloro-3-methylbutane 3-Bromo-1-chlorobutane 1-Ethyl-2-fluorocyclohexane Nomenclature and properties of halogenated alkanes • In terms of chemical reactivity, halogenated alkanes are more reactive than alkanes and cycloalkane analogues, because the C-X bond (X = halogen) makes the bond polar and thus susceptible to reactions that require initial dipole-dipole interactions.