Naming Acids
advertisement

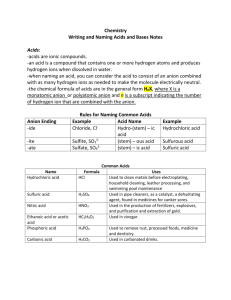
Acid Nomenclature How to identify an acid: The formula of an acid starts with “H” (for now) HCl hydrochloric acid How to identify an acid: The formula of an acid starts with “H” (for now) HNO3 nitric acid How to identify an acid: The formula of an acid starts with “H” (for now) H3PO4 phosphoric acid Naming Acids Acid names are derived from the anion that is bonded to hydrogen HCl HNO3 H3PO3 Naming Acids Binary Acids: • Contain “H” and one other element To Name: Hydro + second element name + ic + Acid Ex: HCl = Hydro chlorine – ine + ic Acid Hydrochloric Acid HF = Hydroflouric Acid Naming Acids (cont.) Ternary Acids: • Contain “H”, “O” and one more element To Name: 3rd element name + ic + Acid Ex: HNO3 = Nitrogen – ogen + ic + Acid Nitric Acid HClO3 = Chloric Acid Naming Acids (cont.) • If the Acid has one more Oxygen than the base acid, the naming goes as followed: To Name: Per + 3rd element + ic + Acid Ex: HNO4 = Per + Nitrogen – ogen + ic + Acid Pernitric Acid H2SO5 = Persulfuric Acid Naming Acids (cont.) • If the Acid has one less Oxygen than the base acid, the naming goes as followed: To Name: 3rd element + ous + Acid Ex: HNO2 = Nitrogen – ogen + ous + Acid Nitrous Acid H2SO3 = Sulfurous Acid Naming Acids (cont.) • If the Acid has two less Oxygens than the base acid, the naming goes as followed: To Name: Hypo +3rd element + ous + Acid Ex: HClO = Hypo +chlorine –ine + ous + Acid Hypochlorous Acid HNO = Hyponitrous Acid Rules for naming acids #1: Acids that do not contain oxygen HA Hydrogen/nonmetal Not in water In water 1. Hydrogen 2. name of anion Use prefix "hydro" -root- ic acid EXAMPLES: Formula not in water in water HCl hydrogen chloride hydrochloric acid H2S hydrogen sulfide hydrosulfuric acid HCN hydrogen cyanide hydrocyanic acid Rules for naming acids #2: Acids that contain oxygen HAOx (oxyacids) Hydrogen/polyatomic anion Not in water In water 1. Hydrogen 2. name of anion root name then check ending of anion - ite - ate The anion ending changes to -ous acid the anion ending changes to -ic acid Naming Acids If the anion name is… The acid formed from this anion is named… _____ide hydro_____ic acid hypo_____ite hypo_____ous acid _____ite _____ous acid _____ate _____ic acid per_____ate per_____ic acid For Example Formula If the anion name is… The acid formed from this anion is named… HCl chloride hydrochloric acid HClO hypochlorite hypochlorous acid HClO2 chlorite chlorous acid HClO3 chlorate chloric acid HClO4 perchlorate perchloric acid Examples 1. 2. 3. 4. 5. 6. 7. 8. 9. HCl HBr HI H2SO3 HNO2 H3PO3 HNO3 H2SO4 H3PO4 Examples 1. 2. 3. 4. 5. 6. 7. 8. 9. hydrochloric acid hydrobromic acid hydroiodic acid sulfurous acid nitrous acid phosphorous acid nitric acid sulfuric acid phosphoric acid Naming Bases • Name the metal first • Add Hydroxide Ex: NaOH = Sodium Hydroxide KOH = Potassium Hydroxide Practice Naming Bases Formula Name NaOH Sodium Hydroxide KOH Potassium Hydroxide Mg(OH)2 Magnesium Hydroxide Al(OH)2 Aluminum Hydroxide NH4OH Ammonium Hydroxide