Acids and Bases

Section A2.4
Unit A
Science 10
Objective Checklist
At the end of this lesson, will be able to:
Identify and classify acids and bases based on their properties
Name acids and bases
Acids and Bases
Important
Control digestion in our bodies (stomach acid and activation and deactivation of enzymes)
Found in foods and used in industry
Acid or a Base?
Taste:
Acids- sour (ex. Lemons- citric acid)
Bases- bitter (ex. Soap)
Touch:
Bases- slippery to the touch and corrosive to the skin
What does corrosive mean?
Acids- not slippery and corrosive to the skin
Reaction with Metal:
Acids- react; forms H2 gas (bubble formation)
Bases- don’t react
Using pH
What is pH used for?
What does the scale look like?
What substance is considered neutral?
Where do the following substances place on the scale?
Acid rain, normal rain, lemon juice, stomach juices
Baking soda, human blood, oven cleaner, window cleaner (ammonia)
Conductivity
Both acids and bases have or form ions in solution
In solution, these ions can conduct electricity, so they are electrolytes
Can you think of any everyday items that contain an acid or a base (alkali) to conduct electricity?
Indicators
To determine pH and whether something is an acid or base, we use indicators
Indicators change color depending on pH
Litmus paper is created through extracting the chemical from lichens
Acid: blue litmus paper turns red (BRA)
Base: red litmus paper turns blue (RBB)
Neutral: paper stays same color
ACI D
RE D
B ASE
B LUE
Demonstration
What color should the acid (lemon juice, citric acid) turn the blue litmus paper? Red litmus paper?
What color should the base (bleach) turn the red litmus paper? Blue litmus paper?
How do we test whether the unknown is an acid or base? If blue litmus stays blue, can we conclude that it is a base? Why or why not?
Recognizing Acids
In acids, the formula often has a “H” (hydrogen ion – may be more than one) on the left side of the formula
(ex. HCl)
Can also appear on the right side as “-COOH” (organic acid group) (ex. CH3COOH)
They are bonded to an anion (one or more nonmetals)
To identify acids from formulas, look for “H” on the left or “-COOH” on the right
Recognizing Acids
Which of the following are acids?
HBr(aq) NaCl(aq) H2SO4(aq) HCOOH(l)
NaOH(aq) H2O(l) HNO3(aq) C
6
H
5
COOH(l)
K2SO4(aq)
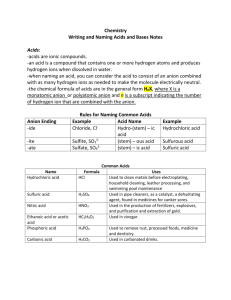
Naming Acids
IUPAC method: name as an aqueous ionic compound
What would HCl be using these rules?
Another method:
1) When containing hydrogen and non-metallic element
(anion name ends in –ide):
Use prefix “hydro” and suffix “ic”
Ex. HCl becomes hydrochloric acid
What would HF be using this rule?
Naming Acids cont…
2) When acid contains oxygen (ex. H2SO4
(aq)
), name based on the anion
What is the anion in H2SO4
(aq)
?
If anion ends in “ate”, name of acid ends in “ic”
Ex. H2SO4
(aq) contains sulfate anion (SO4 2) and is then named sulfuric acid
What is the acid name of H3PO4
(aq) approach naming it?
? How do we
Naming Acids cont…
3) if anion ends in “ite”, name of acid ends in “ous”
H
2
SO
3 (aq) contains the anion sulfite (SO named sulfurous acid
3
2), so the acid is
What is the name of the acid HNO
2(aq)
?
Practice
Acid Formula
HNO3
(aq)
H2CrO4
(aq)
HI
(aq)
HBr
(aq)
HClO
(aq)
Name nitric acid chromic acid hydroiodic acid hydrobromic acid hypochlorous acid
For your reference
Back of periodic table sheet
Recognizing Bases
Often, bases are identified by presence of “OH ”
(hydroxide ion) with a metal ion or the ammonium ion (NH
4
+ )
It is more complicated than this, but for now:
Any compound with high solubility and an OH on right is a base
Name base as would name an ionic compound
What would NaOH be named using rules we already know?
Practice
Base
KOH(s)
NH4OH(aq)
Ca(OH)2
Mg(OH)2(s)
Al(OH)3(s)
Name potassium hydroxide ammonium hydroxide calcium hydroxide magnesium hydroxide aluminium hydroxide
Neutralization
Reaction between acid and a base which produces water and a salt
The acidic and basic properties disappear
Why would this be important? Give an example
Ex. A bee’s sting contains methanoic acid which dissolves nerve endings in the skin (lots of pain!).
Sting creams containing ammonia limit the pain as the ammonia neutralizes the acid
Neutralization Demo
What pH is water?
Is vinegar an acid or a base?
What color do we observe initially?
If I add alka-seltzer (a base) predict the color change
What do we observe as I add the base?
Explain why this occurred