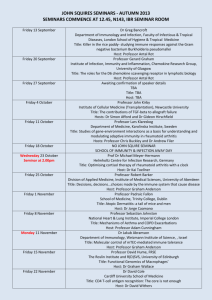
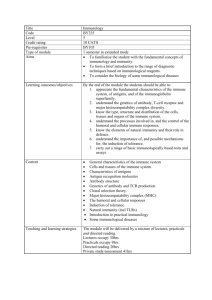
XXIX Meeting of the Swiss Immunology PhD students - BLV
advertisement

XXIX Meeting of the Swiss Immunology PhD students April 2 – April 4 2012 Schloss Wolfsberg Generously sponsored by: The Swiss Society for Allergology and Immunology, the Union Bank of Switzerland and the “Kontaktgruppe für Forschungsfragen” (Roche, Novartis, Merck Serono) WOLFSBERG MEETING April 2 – April 4, 2012 Bus transfer Monday, April 2, 2012 from Weinfelden railway station to Schloss Wolfsberg train departure Zurich at 12.07 h arrival Weinfelden at 12.58 h Wednesday, April 4, 2012 from Schloss Wolfsberg to Weinfelden railway station departing Schloss Wolfsberg at 13.15 h train departure to Zurich at 13.59 h CONFERENCE PROGRAM Sw iss Immunology PhD Meeting (2012) Monday, April 2 13:00-14:00 Check-in 14:00-14:05 Welcome Session 1 14:05-15:15 1° and 2° lymphoid organs, cell migration Chair: Christoph Müller 14:05-14:19 Angela Bosch - Uni Basel The role of continued Foxn1 expression in thymus organogenesis and maintenance 14:19-14:33 Chen-Ying Yang - Uni. de Lausanne What happens to the network of T zone fibroblastic reticular cells during lymph node swelling? 14:33-14:47 Qian Chai - Institute of Immunobiology Fibroblastic Reticular Cells coordinate structural dynamics of Lymph Nodes during viral infection 14:47-15:01 Romain BALLET - CMU The Junctional Adhesion Molecule C (JAM-C) regulates dendritic cell migration in inflammation. 15:01-15:15 Amiel Olivos - CHUV REGULATION OF DENDRITIC CELL MIGRATION BY MACROPHAGHE MIGRATION INHIBITOR FACTOR Session 2 15:15-15:45 Innate immunity, inflammation Chair: Christoph Müller 15:15-15:29 Jeremy Loyau - Novimmune Structure-function relationship between 15C1 and TLR4 15:29-15:43 Sebastian Heer - ETHZ Target gene(s) of the transcription factor Nrf2 mediate inflammasome activation. 15:45-16:00 Coffee break Session 3 16:00-16:50 Innate immunity, inflammation (2) Chair: Christoph Müller 16:00-16:12 Pascal Ziltener - ETH Zürich The role of innate immunity in control of Legionella pneumophila lung infection 16:12-16:24 deepa mohanan - USZ Inhibition of inflammasomes allows BCG to escape host immune responses by decreased phagosome maturation. 16:24-16:36 Patrycja Konieczna - SIAF Swiss Institute of Allergy and Asthma Research Plasmacytoid dendritic cells induce Foxp3 regulatory T cells in response to Bifidobacterium infantis 16:36-16:48 Md Mahbubul Morshed - University of Bern Thymic stromal lymphopoietin (TSLP) stimulates eosinophil extracellular trap release Session 4 16:50-17:40 Allergy Chair: Ton Rolink 16:50-17:02 Tess Brodie - Institute for Research in Biomedicine Distribution of allergen specific cells in diverse T cell subsets for seasonal and non seasonal allergens 17:02-17:14 Eva Gollwitzer - CHUV-UniL Influence of Respiratory Syncytial Virus infection on allergic airway responses depends on age 17:14-17:26 Helga Pawelski - ETH - Integrative Biology IL-21 promotes Th2 allergic airway responses by inhibition of Foxp3+ T regulatory cells 17:26-17:38 Kerstin Wanke - SIAF Regulation of Bronchial Epithelium Integrity and Tight Junctions by Regulatory T cells in allergic airway disease 17:45-18:30 UBS Presentation 18:45-20:00 Dinner Session 5 20:10-21:30 Allergy (2) Chair: Ton Rolink 20:10-20:23 Mattia Garbani - Swiss Institute for Allergy and Asthma Research Dendritic cells targeting and improved cell penetration in allergen-specific immunotherapy 20:23-20:36 Moira Prati - SIAF Targeting the Extracellular Membrane-Proximal Domain of IgE memory B cell 20:36-20:49 Susanne Hoesli - University of Bern Cytosolic vacuolization of eosinophils: Involvement of CD11b 20:49-21:02 Ursina Gurzeler - Institute of Pharmacology A System For Quantitative Production Of Murine Basophils Ex Vivo 21:02-21:15 patrick buschor - University Institute of Immunologie An Fc-engineered anti-IgE-DARPin fusion protein shows increased inhibition of ongoing allergic reactions 21:15-21:28 Stephan Lloyd Watkins - InselSpital-University of Bern Computational, Biochemical and Cellular Elucidation of Non-hapten Pharmaceutical Interactions (p-i) in Adverse Drug Hypersensitivity Tuesday, April 3 07:00-08:00 Breakfast Session 6 08:00-10:00 Adaptive immunity in infection Chair: Annette Oxenius 08:00-08:13 Josh Crouse - ETH Zurich Direct type I IFN signaling on CD8 T cells protects them from NK cell mediated killing 08:13-08:26 Sandra Kallert - University of Geneva Shaping of the immune response in persistent viral infections 08:26-08:39 Mai Matsushita - ETH-Molecular Health Science Glutathione Peroxidase 4 is pivotal for maintenance of peripheral CD8+T cell pool and anti-viral CD8+ T cell responses 08:39-08:52 Iwana Schmitz - Institute of Integrative Biology IL-21 restricts virus-driven Treg cell proliferation to prevent T cell exhaustion and viral persistence in chronic LCMV infection 08:52-09:05 Christoph Schneider - ETH Zürich IL-9 is a potent suppressor of antiviral T cell responses 09:05-09:18 Daniel Utzschneider - CHUV Recovery of functional T-cells from LCMV clone 13 infected mice 09:18-09:31 Gregor Bedenikovic - ETH Zürich THE ROLE OF “T HELP” DURING SECONDARY CD8+ T CELL RESPONSES IN ACUTE VIRAL INFECTIONS 09:31-09:44 Jovana Cupovic - Kantonal Hospital St. Gallen Tuning TCR avidity of virus-specific CD8+ T cells 09:44-09:57 Stéphanie Longet - Centre Hospitalier Universitaire Vaudois A murine genital-challenge model is a sensitive measure of protective antibodies against human papillomavirus infection 10:00-10:20 Coffee break Session 7 10:20-11:00 Adaptive immunity to infection (2) Chair: Annette Oxenius 10:20-10:34 Kerstin Weidner - Microbiology/ETH Antigen presenting cells involved in Th17 cell priming during Candidiasis 10:34-10:48 Sonia Liliana Bastidas Patino - ETH Zürich THE IMPACT OF HIV-1 REPLICATION ON THE ACTIVATION OF CD8+ T CELLS WITH SPECIFICITIES FOR DIFFERENT ANTIGENS 10:48-11:02 Kathrin Zimmermann - ETH Zürich Co-stimulatory effect of the HIV-1 envelope glycoprotein on CD4+ T cells during chronic HIV-1 infection Session 8 11:00-12:00 Selection and autoimmunity Chair: Maries van den Broek 11:00-11:14 Carlos Mayer - Universität Basel About the Role of Dicer-dependent miRNA in the Thymus 11:14-11:28 Anja Nusser - University of Basel The amount of self-antigen determines the effector function of murine T cells escaping negative selection 11:28-11:42 Sarah Enouz - CHUV Characterizing autoreactive CD8+ T cells that bypass negative selection. 11:42-11:56 Stanislav Pantelyushin - UZH Rorγt+ innate lymphocytes and γδ T cells initiate psoriasiform plaque formation in mice 12:00-13:00 Lunch 13:15-15:40 Excursion 15:40-16:00 Coffee break Session 9 16:00-18:30 Poster Chair: no chair 18:30-20:00 Dinner 20:00-23:20 After dinner sports and other activities Wednesday, April 4 07:00-08:00 Breakfast Session 10 08:00-09:30 T cell activation / polarization Chair: Walter Reith 08:00-08:13 Jean Enno Charton - Université de Lausanne Malt1 and Caspase-10: Proteases Essential for Lymphocyte Activation 08:13-08:26 Camilla Basso - Institute for research in Biomedicine Cervical lymph nodes and the pivotal role in the CCR6+ T helper cell priming. 08:26-08:39 simone becattini - institute for research in biomedicine Priming of polarized human T cells by microbes 08:39-08:52 Junhua Wang - Institute of parasitology Course of the Th-1, Th-2 and Th-17 Cytokines and of Associated Chemokines Involved in the Periparasitic Recruitment of Cells in Echinococcus multilocularis Infection 08:52-09:05 Peter Bretscher - ETH - Integrative Biology Oxidized phospholipid signaling in microbial infection and inflammatory disease 09:05-09:18 Natascha Wuillemin - University Hospital Bern Mechanisms of T cell stimulation by flucloxacillin in HLA-B*5701+ flucloxacillin-naïve individuals Natascha Wuillemin1, Jacqueline Adam1, Stefano Fontana2, Werner J. Pichler1, Daniel Yerly1 1 Clinic for Rheumatology and Clinical Immunology / Allergology, University Hospital of Bern, 3010 Bern, Switzerland 2 Regional Blood Transfusion Service of the Swiss Red Cross, 3008 Bern, Switzerland 09:18-09:31 Federico Mele - Institute for research in biomedicine Phenotype and function of pathogen-specific human CD4+ memory T cells Session 11 09:30-10:00 Immune regulation Chair: Walter Reith 09:30-09:43 Ilaria Mosconi - EPFL Role of Thymic stromal lymphopoietin (TSLP) signaling in intestinal immune homeostasis 09:43-09:56 Nina Suhartha - Pharmacology IFN-α modulates the expression of the gut-homing receptor α4β7 10:00-10:20 Coffee break Session 12 10:20-10:40 Immune regulation (2) Chair: Walter Reith 10:20-10:33 Franziska Zabel - University Hospital Zurich Primary and memory B cell responses to Qβ-VLP in mice Session 13 10:40-12:00 Tumor immunity Chair: Maries van den Broek 10:40-10:53 Ali Bransi - USZ A novel model for inducible autochthonous cancer to study local immune subversion 10:53-11:06 Sara Burkhard - Experimental Immunology Impact of innate lymphocytes on the tumor microenvironment 11:06-11:19 Sergio Haller - University of Lausanne Augmentation of antitumor immunity applying a semi-allogeneic dendritic cell line 11:19-11:32 Christian Hagedorn - University Hospital Zurich Tumour therapy by redirected Fibroblast Activation Protein-a specific CD8+ T cells 11:32-11:45 Cristina Riccadonna - Geneva University Hospitals and University of Geneva Modulation of astrocytoma-cytotoxic T lymphocyte interactions by chemotherapy 12:00-13:00 Lunch 13:15-13:15 Departure Last name Al Sayed First name Mohamad Institution DKF Department Tumor Immunology Address Murtenstrasse 35 Zip code 3010 City Bern Email mohamad.alsayed@dkf.unibe.ch BALLET Romain CMU Pathology Immunology Rue Michel Servet, 1 1211 Geneva ballet.r@gmail.com Basso Camilla Cellular Immunology Via Vincenzo Vela 6 6500 Bellinzona camilla.basso@irb.unisi.ch Bastidas Patino Sonia Liliana Institute for research in ETH Zürich sonia.bastidas@micro.biol.ethz.ch simone bellinzona simone.becattini@irb.unisi.ch Bedenikovic Gregor institute for research in ETH Zürich ETH Hönggerberg, HCI G 8093 405 Wolfgang-Pauli Str. via vincenzo vela 6 6500 Zurich becattini Institute of Microbiology, D-BIOL immune regulation lab Bosch Angela Uni Basel Botelho Natalia Bransi Ali Bretscher Peter Brodie Tess Burkhard Sara buschor patrick Chai Qian Charton Jean Enno Coutaz Manuel Crouse Josh Cupovic Jovana Enouz Sarah Fertig Marina Institute of Microbiology Wolfgang-Pauli-Strasse 10, HCI G405 Biomedicine Mattenstrasse 28 8093 Zürich gregor.bedenikovic@micro.biol.ethz.ch 4058 Basel angela.bosch@unibas.ch Ludwig Institute for Cancer USZ University of Lausanne Ch de Boveresses 155 1066 Epalinges natalia.botelho@unil.ch Oncology Wagistrasse 14 8952 Schlieren ali.bransi@hispeed.ch ETH - Integrative Biology Institute for Research in Experimental Immunology University Institute of Institute of Immunobiology Université de Lausanne University of Lausanne ETH Zurich d-UWIS Wagistrasse 27 8952 Schlieren peter.bretscher@env.ethz.ch Cellular Immunology Via Vincenzo Vela 6 6500 Bellinzona tess.brodie@irb.usi.ch Pathology Winterthurerstrasse 190 8057 Zürich sara.burkhard@neuroimm.uzh.ch DOLS Sahlihaus 2 3010 Bern buschor@students.unibe.ch Kantonsspital St.Gallen Institut für Immunbiologie Haus 09 Chemin des Boveresses 155 F512A Ch. des Boveresses 155 Wolfgang-Pauli str. 10 9007 St Galeen qian.chai@kssg.ch 1066 Epalinges jeanenno.charton@unil.ch 1066 Epalinges manuel.coutaz@unil.ch 8093 Zurich josh.crouse@micro.biol.ethz.ch St. Gallen jovana.cupovic Epalinges sarah.enouz@chuv.ch Fribourg marina.fertig@unifr.ch Departmement Biochimie Department Biochemistry Microbiology de of Kantonal Hospital Institut of Rorschacherstrasse 95 9007 St. Gallen Immunobiology IMMBIO CHUV IAL Chemin des 1066 Boveresses,155 University of Department of Medicine Chemin du Musée 5 2nd 1700 Fribourg / MED3 floor/ room 2.105 Garbani Mattia Gehrig Jasmine Gollwitzer Eva Govender Lerisa Gurzeler Ursina Hagedorn Christian Haller Sergio Hartley Mary-Anne Heer Sebastian Hoesli Susanne Kallert Sandra Konieczna Patrycja Kovtonyuk Larisa Longet Stéphanie Loyau Jeremy Mager Lukas Matsushita Mai Mayer Swiss Institute for Molecular Allergology Allergy and Ludwig Institute LUDWIG for Cancer CHUV-UniL Pneumologie Oberestr. 22 7270 Davos Platz mattia.garbani@siaf.uzh.ch 1066 Lausanne jasmine.gehrig@unil.ch 1011 Lausanne eva.gollwitzer@chuv.ch Transplantation Immunopathology faculty of medicine Ch. des Boveresses 155 CH-1066 Epalinges CHUV Laboratoire de Pneumologie BH19.105 Institut universitaire de pathologie - IUP Bureau Friedbühlstrasse 49 1011 Lausanne lerisag@hotmail.com 3010 Bern ursina.gurzeler@pki.unibe.ch Oncology Sternwartsrasse 14 8091 Zurich chagedor@student.ethz.ch Chemin de Boveresses 155 Chemin des Boveresses 155 ETHZ D-UWIS Molecular Biomedicine Wagistrasse 27 University of Bern Institute of Pharmacolocy Friedbühlstrasse 49 1066 Epalinges sergio.haller@unil.ch 1066 Epalinges mary-anne.hartley@unil.ch 8952 Schlieren sebastian.heer@env.ethz.ch 3010 Bern susanne.hoesli@pki.unibe.ch University of Geneva SIAF Swiss Institute of Allergy University Hospital Zurich Centre Hospitalier Universitaire Novimmune Pathology and Immunology Molecular Immunology 1211 Geneva sandra.kallert@unige.ch 7270 Davos patrycja.konieczna@siaf.uzh.ch Ramistrasse 100 G PATH 8091 37 Service of Immunology Vieux-Moulin 8 1018 and Allergy/Service of Antibody Engineering 14 chemin des Aulx 1228 Zurich larisa.kovtonyuk@usz.ch Lausanne stephanie.longet@unil.ch Plan Les Ouates jloyau@novimmune.com Experimetal Immunopathology D-BIOL Murtenstrasse 31 3010 3010 Bern Postfach 62 Wagistrasse 27 8952 Bern lukas.mager@pathology.unibe.ch Schlieren mai.matsushita@env.ethz.ch Carlos Insitute of Pathology ETH-Molecular Health Science Universität Basel Mattenstrasse 28 Basel carlos.mayer@unibas.ch Meister Fabian ETH Department Biomedicine Micro asdfasfs fabian.meister@micro.biol.ethz.ch Mele Federico Bellinzona federico.mele@irb.usi.ch mohanan deepa Institute for research in USZ safasdfasdfasdf sdfsdfsd 34234 sadfsdfsdf Via Vincenzo vela 6 6500 F14, Dermatologische Klinik Gloriastrasse 31 zurich deepa.mohanan@hotmail.com University of Lausanne Institute of Pharmacology University Hospital Zurich University of Lausanne UNIL Department Biochemistry Biochemistry of Centre Medicale Universitaire 1, rue Obere Strasse 22 Haematologie of Cellular Immunology Dermatology 4058 8091 Morshed Md Mahbubul University of Bern Institute of Pharmacology EPFL Global Health Institute Bern mahbubul.morshed@pki.unibe.ch Mosconi Ilaria Lausanne ilaria.mosconi@epfl.c Nueesch Ursina Nussbaum Kathrin Nusser Anja University Children's Hospital University of Zurich University of Basel Zürich ursina.nueesch@kispi.uzh.ch 8045 Zürich nussbaum@immunology.uzh.ch 4058 Basel anja.nusser@unibas.ch Olivos Amiel CHUV 1011 lAUSANNE amiel.olivos-ortiz@chuv.ch Oxenius Annette ETH Zurich Pantelyushin Stanislav Pawelski Helga Prati Moira Institute of Microbiology Wolfgang-Pauli-Str. 10 HCI G401 UZH Experimental Winterthurerstrasse 190 Immunology Y44J42 ETH - Integrative D-UWIS Wagistrasse 27 Biology SIAF Molecular Allergology Obere Strasse 22 8093 Zürich oxenius@micro.biol.ethz.ch 8057 Zurich pante@neuroimm.uzh.ch 8952 Schlieren helga.pawelski@env.ethz.ch 7270 Davos moira.prati@siaf.uzh.ch Presotto Danilo CHUV - UNIL Biopôle 3, Route de la 1066 Corniche 9B rue Gabrielle-Perret- 1211 Gentil 4 Wagistr.27 8952 Epalinges danilo.presotto@unil.ch Riccadonna Cristina Schmitz Iwana Schneider Christoph Geneva University Oncology Hospitals and Institute of D-UWIS Integrative Biology ETH Zürich D-UWIS geneva riccadonny@libero.it Schlieren-Zurich iwana.schmitz@env.ethz.ch Wagistrasse 27 8952 Schlieren christoph.schneider@env.ethz.ch Schuster Steffen Department of Chemin des Boveresses 1066 Biochemistry, WHO-IRTC 155 Medicine ch. du musée 5 1700 Epalinges steffen.schuster@unil.ch Fribourg thibaud.spinetti@unifr.ch Nina University of Lausanne University of Fribourg Pharmacology Spinetti Thibaud Suhartha Medicine 1700 Fribourg nina.suhartha@unifr.ch Thom Jenny ETH Zurich 8093 Zurich jenny.thom@micro.biol.ethz.ch Utzschneider Daniel CHUV 1066 Epalinges daniel.utzschneider@chuv.ch Wang Junhua 3001 Bern Wanke Kerstin Institute of parasitology SIAF Institute of Microbiology Wolfgang-Pauli-Strasse 10 SVRI Biopole 3 Ch. des Boveresses 155 University of Bern Laenggass-Strasse 122 Cellular Immunology 7270 Davos Platz wangjh28@163.com;bruno.gottstein@vetsuis se.unibe.ch k_wanke@gmx.de Immunology Friedbühlstrasse 49 EPFL SV GHI UPHARRIS 1015 SV 3820 (Bâtiment SV) Steinwiesstrasse 75 8032 Institute of Experimental Winterthurerstr. 190 Immunology Department of Mattenstrasse 28 Biomedicine MIN RUE DU BUGNON 46 Immunology 3010 ch. du Musée 5 Obere Strasse 22 Watkins Stephan Lloyd InselSpitalRIA-Immunology University of Bern Microbiology/ETH D-Biol BERN jasongrey333@yahoo.com Zürich kerstin.weidner@me.com Bern natascha.wuillemin@allergy.unibe.ch ZURICH chakradhar465@gmail.com Chen-Ying University Hospital Bern University Hospital, Zurich Uni. de Lausanne Inselspital Dept. RIA 3011 Sahlihaus 2 RM 43 HCI G435 Wolfgang- 8093 Pauli-Str. 10 Clinic for Rheumatology Sahli-Haus 1 3010 and Clinical Experimental Ramistrasse 100, G- 8091 Hematology PATH 37 Biochemistry Ch. des Boveresses 155 1066 Weidner Kerstin Wuillemin Natascha Yakkala Chakradhar Yang Epalinges irvineyang@gmail.com Yun James Inselspital Zabel Franziska Ziltener Pascal University Hospital Zurich ETH Zürich Department of Rheumatology, Clinical Dermatology Zimmermann Kathrin ETH Zürich Strandweg 9A 3400 Burgdorf james.yun@allergy.unibe.ch Gloriastrasse 31 8091 Zurich franziska.zabel@usz.ch 8093 Zurich pascal.ziltener@micro.biol.ethz.ch 8093 Zürich kathrin.zimmermann@micro.biol.ethz.ch Institute of Microbiology Wolfgang-Pauli-Str. 10 ETH Hongerberg HCI Institute of Microbiology Wolfgang-Pauli-Str. 10 Wolfsberg 2012 Particpating chairs Name Hans Hengartner Christoph Müller Annette Oxenius Walter Reith Antonius Rolink Maries van den Broek Institution University Hospital Zürich University of Berne ETHZ University of Geneva University of Basel University Hospital Zürich email hans.hengartner@usz.ch christoph.mueller@pathology.unibe.ch oxenius@micro.biol.ethz.ch Walter.Reith@medecine.unige.ch antonius.rolink@unibas.ch maries@van-den-broek.ch Presentation abstracts Session 1 1° and 2° lymphoid organs, cell migration The role of continued Foxn1 expression in thymus organogenesis and maintenance Angela Bosch1, Thomas Barthlott1, Saulius Zuklys1 and Georg A. Holländer1,2 1 Pediatric Immunology, Department of Biomedicine, University of Basel and Basel University Children’s Hospital (UKBB), Basel, 2Department of Paediatrics, University of Oxford, Oxford, United Kingdom Introduction: The transcription factor Foxn1 is essential for thymic epithelial cell (TEC) differentiation and its loss causes athymia. The goal of this study is to define the temporal and quantitative requirements of Foxn1 for the development, differentiation and maintenance of TEC and hence a regular thymic microenvironment. Methods: Two mouse models have been generated to test the role of Foxn1 expression for thymus organogenesis and maintenance. The first model allows to study the consequences of a loss of Foxn1 expression after thymus organogenesis has been initiated (embryonic day 12). In contrast, the second model expresses a hypomorphic Foxn1 allele that results in a reduction of Foxn1 expression enabling the analysis of a gene dosage effect. Results: The thymus of both mouse models was significantly reduced in size, displayed cysts and had lost its regular microarchitecture when compared to wild type mice. Blood-borne T-cell precursors homed - albeit at reduced frequencies - to the altered thymic microenvironment, but were functionally unimpaired. However, in situ they gave rise to only few double positive cells generating a reduced number of peripheral T-cells with partial functionality. Regulatory T cells were also generated although their suppressive potential was reduced. Conclusion: TEC differentiation and maintenance is dependant on continuous and sufficient Foxn1 expression. A short term presence or low amounts of Foxn1 do not support regular thymus development and function whilst homing of T-cell precursors to the postnatal thymus and minimal T-cell differentiation is observed in the absence of regular Foxn1 expression. What happens to the network of T zone fibroblastic reticular cells during lymph node swelling? Chen-Ying Yang1,2, Tobias K. Vogt1,2 and Sanjiv A. Luther1 1 Department of Biochemistry, University of Lausanne, Switzerland 2 contributed equally Abstract: (max. 250) Efficient primary T cell immune responses to lymph-born antigens are mainly initiated in the T cell-rich zone of lymph node (LN). Throughout the T zone, T zone fibroblastic reticular cells (TRCs) form a sponge-like network that serves as scaffold for adhesion of antigen-carrying dendritic cells (DCs) and for migration of T cells thereby facilitating the activation of the rare antigenspecific T cells. In addition TRCs produce CCL19, CCL21 and IL-7 to support the trafficking and homeostasis of T cells during homeostasis. TRC also produce the matrix scaffold responsible for organ stability. However, despite of their critical role in regulating the homeostasis and activation of T lymphocytes, the response of the TRC network to immune response and LN swelling in particular is poorly understood. To study this process we set up a LN swelling model in which the mice receive first low numbers of ovalbumin (OVA)-specific CD4 (OTII) and CD8 (OTI) T cells followed by subcutaneous immunization with OVA in Montanide adjuvant. Immunofluorescence microscopy demonstrated that the TRC network simply grows in size while preserving its original organization thereby allowing a greater number of T cells to localize there. Flow cytometric analysis further showed that the growth of the network is due to the increase in both the size and the number of TRC, which is mainly due to extensive proliferation. Therefore, the TRC network adapts rapidly to LN swelling while keeping its integrity. We are currently investigating the mechanism that triggers the rapid proliferation of TRC during immune response. Fibroblastic Reticular Cells coordinate structural dynamics of Lymph Nodes during viral infection Qian Chai1*, Lucas Onder1*, Elke Scandella1, Renzo Danuser 6, Tim Sparwasser2, Sanjiv Luther3, Volker Thiel1, Thomas Rülicke4, Thomas Hehlgans5, Jens V. Stein6 and Burkhard Ludewig1 1 Institute of Immunobiology, Kantonal Hospital St. Gallen, St. Gallen, Switzerland; 2Institute of Infection Immunology, TWINCORE, Hannover Medical School, Germany; 3EPFL, Lausanne, Switzerland; 4Institute of Laboratory Animal Science and Biomodels Austria, University of Veterinary Medicine Vienna, Vienna, Austria; 5 University of Regensburg, Regensburg, Germany; 6Theodor Kocher Institute, Bern, Switzerland Abstract The paracortex of lymph nodes (LNs) harbors a sponge-like scaffold of stromal cells known as fibroblastic reticular cells (FRCs). A major function of FRCs is to build and enwrap the conduit system of collagen fibers that directs interstitial fluids from the afferent lymph through the T-cell zone to high endothelial venules. Furthermore, FRCs regulate T-cell migration and survival by producing the chemokines CCL19 and the serine isoform of CCL21. To gain further knowledge on the biology of FRCs and possibly other stromal cell subsets, we have generated a bacterial artificial chromosome (BAC)-transgenic mouse model that utilizes the CCL19 promoter to direct the Cre-recombinase to LN stromal cells. Crossing of CCL19-Cre mice to the R26-EYFP reporter mice revealed that transgene expression in LNs was almost exclusively confined to podoplanin+CD31- cells. Likewise, transgene activity in spleens and Peyer’s patches of CCL19Cre mice was largely restricted to FRC-like cells, i.e. stromal cells within the T cell zone and the T-B border. Selective ablation of the lymphotoxin-beta receptor on CCL19-Cre-positive cells resulted in profound changes in the development and organization of secondary lymphoid organs. Taken together, stromal CCL19-Cre expression is well-suited (i) to characterize the development of LN FRCs in vivo, (ii) to molecularly dissect the contribution of stromal cells to lymphoid organogenesis, and (iii) to address the function of LN FRCs in the generation of innate and adaptive immune responses. The Junctional Adhesion Molecule C (JAM-C) regulates dendritic cell migration in inflammation. Authors: Romain Ballet, Mélanie Charmoy, Fabienne Tacchini-Cottier, and Beat Imhof During infections, dendritic cells (DCs) are recruited to sites of infection where they process antigens and start to migrate to the draining lymph node in order to trigger specific T cell responses. Here we showed that JAM-C, a tight junction protein expressed on both vascular and lymphatic endothelial cells regulates migration of DCs in inflammation. In vivo, using the Leishmania major experimental model of Leishmaniasis, we found that blocking JAM-C in endothelial cell-cell contacts enhances the recruitment of dendritic cell precursors from blood vessels to the site of infection. In addition, blocking JAM-C increases dendritic cell migration from the infected site to the draining lymph node. In order to decipher the mechanism, we will apply vascular specific JAM-C k.o. animals, perform transmigration assays and vital microscopy. Overall, blocking JAM-C could represent a therapy leading to an increased number of matured Antigen Presenting Cells in draining lymph node, potentially improving the protective T cell response against L. major. REGULATION OF DENDRITIC MIGRATION INHIBITOR FACTOR CELL MIGRATION BY MACROPHAGHE Amiel Olivos-Ortiz1, Thierry Roger1 and Thierry Calandra1. 1Division of Infectious Diseases, Centre Hospitalier Universitaire Vaudoise, Lausanne, Switzerland, 1011. Inflammation process involves a multistep cascade of interactions between leukocyte receptors and soluble mediators present in the inflammatory milieu. In order to be recruited to the inflammation site and to produce an effective immune response, leukocytes need to coordinate the action of multiple signaling molecules that bind to specific receptors expressed on the leukocyte membrane and the intracellular signaling pathways triggered as a result of these interactions, such as PI3K-Akt, MAPK and Rho GTPase pathways. Cytokines are soluble mediators and main players in the development of many inflammatory processes. The macrophage migration Inhibitor factor (MIF) is a pleiotropic proinflammatory cytokine that has been found to contribute to many inflammatory and autoimmune diseases. MIF effects include boosting of cytokine production, TLR-4 up-regulation, promotion of cell cycle progression and proliferation, augment of tumorigenesis and apoptosis inhibition. Evidence suggests that MIF also plays an important role during the migration of epithelial and tumoral cells, as well, during leukocyte recruitment in several pathological conditions. However, the mechanisms by which MIF is able to regulate all this processes are still unclear. In this study we show that in dendritic cells, MIF deficiency decreases the spontaneous migration and the chemokine-induced migration in response to CCL21 and CCL19. These effects depend on p38, PI3K and mTOR, but are independent of ERK 1/2 activation. Nevertheless, the molecular finetuning by which MIF is able to regulate the migration of dendritic cells remains to be determined. Session 2 Innate immunity, inflammation Structure-function relationship between 15C1 and TLR4 Loyan The mammalian Toll-like receptor (TLR) family has evolved to sense pathogens in the environment and protect the host against infection. TLR4 recognizes lipopolysaccharide (LPS) from Gram negative bacteria and induces a signaling cascade that, when exaggerated, has been associated with inflammatory disorders such as acute lung inflammation, septic shock and even arthritis. A monoclonal antibody directed against TLR4, the 15C1, was developed to block its activity. Interestingly, it was demonstrated that the antibody’s inhibitory effect is partially Fc-dependant and mediated by binding to Fc gamma receptors, in particular FcγRI/CD64 and FcγRIIA/CD32A. This mechanism of action has been described in the literature for other cell surface proteins yet is poorly understood. The main goals of my PhD project are to understand the structural and functional basis of 15C1’s inhibitory effect. The study of the structural basis of the 15C1-TLR4 interaction will be presented. Key words: TLR4, monoclonal antibody, phage display, structure-function relationship Target gene(s) of the transcription factor Nrf2 mediate inflammasome activation. Sebastian M. Heer, Esther Rosenwald, Abdijapar T. Shamshiev, Jan Kisielow & Manfred Kopf The transcription factor Nrf2 (nuclear factor, erythroid derived 2, like 2) is described to control expression of genes involved in the antioxidant responses during oxidative/electrophilic stress. We investigated the effects of Nrf2 in inflammatory responses using Nrf2-deficient (Nrf2-/-) mice. Stimulation with TLR ligands triggered enhanced production of inflammatory cytokines, such as IL-6 and IL-12 in macrophages and dendritic cells (DCs) derived from Nrf2-/- mice. In contrast, secretion of IL-1β, another pro-inflammatory cytokine, was reduced. Secretion of mature IL-1ß requires cleavage of intracellular proIL-1ß by multimeric protein complexes called inflammasomes. When Nrf2-deficient cells were treated with inflammasome inducers like ATP or urea crystals, they showed reduced inflammasome activation and IL-1β secretion. Interestingly, reactive oxygen species (ROS) have been thought to play a key role in inflammasome activation. Expectedly, we found that ROS breakdown was delayed in Nrf2-/- DCs. Yet, manipulation of ROS production using various ROS inhibitors or stimulators did not rescue IL-1β production by Nrf2-deficient DCs, indicating little if any role for altered ROS. Expression of various mutated Nrf2 proteins in Nrf2-deficient cells showed that transcriptional activity of Nrf2 is required for inflammasome activation. Thus one or several target genes of Nrf2, and not the Nrf2 protein itself, are needed for efficient inflammasome induction. However, retrovirus-driven expression of several Nrf2target genes including thioredoxin reductase or peroxiredoxin could not rescue inflammasome activation in Nrf2-deficient bone marrow DCs. Taken together, these findings demonstrate Nrf2-target gene(s) control of inflammasome activation independent of ROS levels. Keywords: Nrf2, inflammasome, reactive oxygen species The role of innate immunity in control of Legionella pneumophila lung infection Pascal Ziltener, Stefan Weber and Annette Oxenius Institute for Microbiology, ETH Zürich, Wolfgang-Pauli-Str. 10, 8093 Zürich, Switzerland. Though single cell amoeba are the environmental niche for Legionella pneumophila, the bacteria can also replicate in alveolar macrophages upon transmission to the lung via aerosols, and have the potential to cause a severe form of pneumonia known as Legionnaires’ Disease. The intracellular replication of L. pneumophila depends on the injection of multiple effector molecules into the host cell cytoplasm via a type IV secretion system (T4SS). These effectors promote phagocytosis, inhibit fusion of the phagosome with lysosomes and thus allow the bacteria to establish a replication permissive compartment. Alveolar macrophages are able to detect the activity of the T4SS with cytosolic pattern recognition receptors such as NLRC4, which in combination with MyD88-dependent signals leads to inflammatory secretion of IL-1β. This innate immune response is believed to be important for the control of primary L. pneumophila infection, characterized by IL-1β dependent induction of CXCL1 production by type I alveolar epithelial cells, resulting in the recruitment of neutrophils and subsequent clearance of bacteria. Though reduced L. pneumophila clearance has been shown in mice lacking various innate immune effector molecules, including NLRC4-/-, Caspase-1-/- and IFNγR-/mice, we show here that the bacterial load in the lung five days post intranasal infection is increased 10-fold more in mice lacking TNF production in macrophages, monocytes and neutrophils (mTNF mice), and in MyD88-/- mice. Furthermore, macrophages lacking TNF production are as permissive for intracellular L. pneumophila replication as NLRC4 deficient macrophages. Further investigation of the in vivo immune effector mechanisms mediated by TNF, inlcluding the source of TNF, the mode of action of TNF in restricting L. pneumophila growth and the cell type in which it restricts L. pneumophila growth, will enhance our understanding of how L. pneumophila infection is controlled by the innate immune system. Inhibition of inflammasomes allows BCG to escape host immune responses by decreased phagosome maturation. Mohanan Deepa1, Fettelschoss Antonia1 , Kündig Thomas M.1, Sander Peter2, Böttger Erik C.2, Johansen Pål1 1 Unit for Experimental Immunotherapy, Department of Dermatology, University Hospital of Zurich, Switzerland 2 Institute of Medical Microbiology, University of Zurich, Zurich, Switzerland Tuberculosis (TB) is a highly contagious and persistent infection caused by Mycobacterium tuberculosis and Mycobacterium bovis and has the highest mortality rate than any other infectious disease. .Although there is a vaccine (BCG), its efficacy is poor and there is a great need to improve it. A typical feature of M. tuberculosis infection is that the bacterium resists the fusion of the phagosomes it resides in with the lysosomes. Phagolysosomes are rich in machinery that help in efficient MHC class II antigen presentation and inhibition of the fusion allows the bacterium to dampen the immune response. In a previously reported study, a putative mycobacterial zinc metalloprotease, Zmp1, was shown to play an important role in disease pathogenesis by interfering with caspase-1 activation leading to decreased phagosome maturation. Hence this study’s aim is to dissect the role of inflammasomes in the increased immunogencity of this mutant. Plasmacytoid dendritic cells induce Foxp3 regulatory T cells in response to Bifidobacterium infantis Konieczna Patrycja, Ziegler Mario,Frei Remo, Ferstl Ruth, O’Mahony Liam Swiss Institute of Allergy and Asthma Research (SIAF), University of Zurich, Davos, Switzerland The commensal microbiota is required for optimal immune development and for ongoing intestinal homeostasis, which involves an inter-dependence between microbes and immunity. Bifidobacterium infantis is a commensal bacterium which has been previously shown to protect against inflammatory diseases in murine models e.g. colitis and respiratory allergy. The protective host immune response in murine models was demonstrated to include the induction of T regulatory cells. The aims of this study are: (1) To evaluate the plasmacytoid dendritic cell response to Bifidobacterium infantis; (2) To determine the effect of activated plasmacytoid dendritic cells on naive T cell priming. Firstly, plasmacytoid dendritic cells were stimulated with bacteria and cytokine secretion was measured by luminex. Moreover gene expression was quantified using qRT-PCR. Pattern recognition receptors were blocked using antibodies or oligonucleotides. Bacterial binding was visualized using multispectral flow cytometric imaging. Bifidobacterium infantis stimulated plasmacytoid dendritic cells expressed IL-10 and indoleamine 2, 3-dioxygenase (IDO), with low levels of IL-12p70 and IFN-alpha. The cytokine response was TLR-9, but not TLR-2, dependent. Bacterial activated plasmacytoid dendritic cells were cultured with autologous CD4+ T cells and Foxp3 expression was analysed by flow cytometry. Co-culture of naïve T cells with bacterial primed plasmacytoid dendritic cells resulted in enhanced expression of Foxp3 in CD4+CD25+ cells. Inhibition of IDO activity with 1-methyl-tryptophan blocked induction of T regulatory cells. These results demonstrate that Bifidobacterium infantis-stimulated plasmacytoid dendritic cells induce an immunoregulatory response, suggesting that this microbe may have therapeutic utility in patients with inflammatory disease. Thymic stromallymphopoietin (TSLP) stimulates eosinophil extracellular trap release Mahbubul Morshed 1• 2, Shida Yousefi1, Christina Merz-Stockle1, Hans-Uwe Simon 1, Dagmar Simon2 Institute of Pharmacology1 and Department of Dermatology, lnselspitaf, University of Bem Background: Eosinophils play an important role in host defense, immunoregulation and tissue remodelling. They may release mitochondrial DNA in association with toxic granule proteins forming eosinophil extracellular traps (EETs). EETs are potent in killing bacteria. They have been observed in infectious and non-infectious diseases. TSLP is a cytokine produced by epithelial cells at barrier surfaces. Elevated TSLP expression has been found in atopic diseases in which tissue eosinophilia is a characteristic finding. Aim: We sought to investigate TSLP receptor (TSLPR) expression, the effect of TSLP on eosinophils to release EETs and the extra- and involved intracellular mechanisms in vitro and in vivo. Methods: Human eosinophils were isolated from peripheral blood by negative magnetic selection method. For DNA release assay, cells were taken on cover slip, stimulated with TSLP at different concentrations, fixed and analyzed by confocal microscopy. TSLPR expression on eosinophils in vitro and in vivo was measured by immunofluorescent staining, Western blot and flow cytometry. Viability of TSLPstimulated eosinophils was measured by FACS and calcein-ethidium live cell staining. The role of ROS and adhesion was assessed by pharmacological inhibition of the NADPH oxidase and antibodies to integrins, respectively. Bacterial killing assay was analyzed in overnight cultured agar plate and by confocal microscopy. Results: TSLP stimulated eosinophils to release DNA in association with eosinophil cationic protein (ECP) in a time- and concentration-dependent manner. This DNA release was dependent on adhesion and ROS production by eosinophils. Expression of both, TSLPR and IL-7Ra, was observed on eosinophils. Eosinophil viability was not affected by TSLP stimulation. TSLP mediated EETs also shown bacterial killing property. Eosinophils bearing the TSLPR were also detected in lesional skin of AD, eosinophilic cellulitis and bullous pemphigoid together with TSLPR positive dendritic and mast cells and in association with TSLP expression by keratinocytes. Conclusion: These results demonstrate that TSLP may directly activate eosinophils to generate EETs consisting of mitochondrial DNA in association with granule proteins suggesting a possible role of eosinophils in early defense mechanism against environmental pathogens. Session 3 Allergy XXIV Meeting of the Swiss Immunology PhD students Wolfsberg, 2-4 April 2019 Distribution of allergen specific cells in diverse T cell subsets for seasonal and non seasonal allergens Tess Brodie1, Elena Brenna1, Ravi V. Kolla2, Alessandro Sette2, Antonio Lanzavecchia1, Federica Sallusto1 1 Institute for Research in Biomedicine, Via Vincenza Vela 6, 6500 Bellinzona, Switzerland; 2La Jolla Institute for Allergy and Immunology, 9420 Athena Circle, La Jolla, CA 92037, USA. Tess.brodie@irb.usi.ch Phenotypic and functional heterogeneity is the hallmark of effector and memory T cells. Upon antigenic stimulation, naïve CD4+ T cells make choices to become TH1, TH2, or TH17 cells, or even regulatory T cells. In addition to differences in cytokine repertoire, memory CD4+ T cells exhibit diversity in homing: central memory cells patrol lymphoid organs while effector memory cells act as sentinels in peripheral tissues such as the skin and the gut. To link the phenotype and function of the different memory T cell subsets with their antigen specificity, we have developed a high throughput cellular screening method and are applying this method, called T cell library, to dissect the T cell response to allergens. We will report data on the distribution and responsiveness of allergen-specific T cells in the different subsets in allergic and non-allergic individuals and how the response changes with season. Understanding the dynamics of allergenspecific T cells in naïve, effector and memory Th2 subsets can give insight to the mechanisms of pathology for allergic individuals and tolerance for non-allergic individuals. Influence of Respiratory Syncytial Virus infection on allergic airway responses depends on age Eva S. Gollwitzer1,2, Aurélien Trompette1, Koshika Yadava1,2, Anke K. Sichelstiel1,2, Robert Driscoll1, and Benjamin J. Marsland1,2 1 Service de Pneumologie, CHUV, Lausanne, Switzerland; 2Faculty of Biology and Medicine, University of Lausanne, Switzerland Respiratory Syncytial Virus (RSV) is linked to severe lower respiratory tract infections in infants and young children. It infects nearly all children at least once by the age of three and the age of the initial infection is thought to have a strong influence on the outcome of the disease. A correlation between severe RSV infections in infants and the development of asthma later in life has been established. Nevertheless, little is known about the mechanisms underlying this phenomenon. A predominant theory suggests that a TH2 response is mounted upon RSV infection in infancy as a default mechanism due to an immaturity in TH1 priming. This TH2 bias then influences the development of unrelated allergic responses. However, definitive evidence supporting or challenging this concept has yet to be found. In order to elucidate the mechanisms underlying the impact of RSV infection on subsequent allergic airway responses in vivo, neonatal as well as adult murine models have been established. Mice that were neonatally infected with RSV show increased eosinophilic infiltration into the airways, accompanied by increased mucus production and elevated proliferation of T cells following challenge with an unrelated allergen in adulthood. In contrast, adult RSV infection elicits a protective effect against subsequent allergic airway responses, as characterised by decreased airway eosinophilia, a decrease in allergen-specific T-cell proliferation as well as abrogated TH2 cytokine production. These findings illustrate, that the cellular responses and consequent sequelae following RSV infection differ depending on the age of the initial infection. IL-21 promotes Th2 allergic airway responses by inhibition of Foxp3+ regulatory T cells Helga Pawelski1, Sanchaita Sonar1 , Luigi Tortola1, Koshika Yadava1, Christoph Schneider1, Tim Sparwasser2 and Manfred Kopf1 1 2 Molecular Biomedicine, ETH Zurich, Switzerland Twincore, Institut für Infektionsimmunologie, Hannover, Germany. IL-21 plays an important role in the development of Th2-driven airway inflammation. Previous studies performed in IL-21 receptor deficient (IL-21R-/-) mice revealed a key role of IL-21 in TH2 cell driven inflammatory responses such as lung eosinophilia, airway hyperresponsiveness, blood basophils and type 2 granuloma in asthma and nematode infection. In this study we sought to identify IL-21 dependent mechanisms in the course of allergic asthma. IL21 has been previously shown to inhibit development of inducible T regulatory cells (iTregs) in vitro. We found a significantly increased population of Tregs in the lung of IL-21R-/- as compared to WT mice upon airway challenge with OVA. To further elucidate the role of Treg cells in asthma we utilized DEREG mice carrying a BAC transgene encoding for the diphtheria toxin receptor (DTR) under the Foxp3 promoter. Here specific depletion of Tregs by administration of diptheria toxin (DT) is possible. Depletion of Tregs led to an enhanced influx of Th2 cells into the lung and exaggerated features of asthma in both IL-21R+/+xDEREG and IL21R-/-xDEREG mice, indicating that Tregs are potent suppressors of asthma. To identify the time window when Tregs are imperative for the outcome of asthma, administration of DT was limited to either the sensitization or the challenge phase. Tregs were seen to be essential in both phases since depletion in either phase affected the outcome of asthma, suggesting an additive effect of Treg action. Taken together, these results suggest that IL-21R signaling negatively regulates Treg cell population and consequently the severity of OVA-induced lung inflammation. Regulation of Bronchial Epithelium Integrity and Tight Junctions by Regulatory T cells in allergic airway disease Kerstin Wanke, Ruth Ferstl, Zsolt I. Komlosi, Jeanette I. Kast, Maciej Chalubinski, Paulina Wawrzyniak, Michael Soyka, Stefan Söllner, Liam O’Mahony, Cezmi A. Akdis Swiss Institute of Allergy and Asthma Research (SIAF), University of Zurich, Davos, Switzerland, kerstin.wanke@siaf.uzh.ch Background: Bronchial epithelial cells represent a physical barrier that confers protection and plays a key role in asthma-associated lung inflammation. Tight junction (TJ) complexes are responsible for the integrity of the epithelial barrier. They form close cell-cell contacts and control cellular integrity, polarity and paracellular flow of molecules and cells. The interplay between epithelium integrity and resident T-cells has been unknown so far. Aim: To investigate the control of TJ and epithelium integrity by regulatory Tcells (Tregs). Methods: Pre-activated CD4+CD25+FoxP3+ Tregs were transferred systemically or intratracheally into BALB/c mice with OVA-induced allergic airway disease. Hallmarks of allergic airway disease such as differential cell count in BAL, cytokine production in lung cells and lung inflammation were assessed. Bronchial and TJ integrity was analyzed by total protein content in BAL supernatants, qPCR and confocal microscopy. Results: A wide range of TJ proteins was expressed in bronchial epithelial cells and had the tendency to be downregulated in lung inflammation. The adoptive transfer of Tregs decreased peribronchial inflammation, and prevented the down-regulation of TJ proteins. The expression of TJ proteins correlated strongly with the expression of the Treg cytokines IL-35 and TGFbeta, while they didn’t correlate with Th2 cytokines and negatively correlated with IFN-g. Further, intratracheal administration of Tregs seemed to be more effective than systemic administration in supressing hallmarks of allergic airway inflammation. This lead to a strong recovery in epithelial integrity including TJ expression. Conclusion: We provide here evidence for a key role of Tregs in controlling TJ and bronchial epithelium integrity. Dendritic cells targeting and improved cell penetration in allergen-specific immunotherapy Mattia Garbani, Claudio Rhyner, Reto Crameri Swiss Institute for Allergy and Asthma Research (SIAF), University of Zürich, Oberestr. 22, Davos, Switzerland Todays therapies for allergic diseases aim to induce a switch towards a T H 1/T reg dominated immune response by challenging the patient with increasing doses of allergen. This approach has two major disadvantages: the duration of the treatment and the anaphylactic side-effects. The Modular Antigen Translocation (MAT) Vaccine consist of a cell penetration peptide linked to a part of invariant-Chain (Li). In the cell Li targets the immunogenic protein directly to MHC-II, therefore i) less antigen is needed and ii) less injections are necessary. MAT vaccines are potentially internalized in every cell and do not specifically target antigen presenting cells. Moreover they show the undesired feature of migrating to the nucleus. The aim of the project is to create a new generation of vaccines where the TAT peptide is substituted or supplemented by newly discovered peptides, lacking nuclear localization sequence or able to specifically target dendritic cells. By confocal microscopy and flow cytometry it was shown that, when added exogenously and fused to a protein of interest, CPP512 triggers the internalization of the construct into HeLa cells without driving it towards the nucleus. Additionally, using Green Fluorescent Protein fusions, we were able to confirm the specific targeting of the DC-specific peptide pep3. Pep3 constructs were specifically binding to DCs, but not to other investigated cell types like CaCo-2 and THP1. In the next stage the efficacy of the modified versions of the MAT vaccine will be tested in vivo in mouse models of allergy. Targeting the Extracellular Membrane-Proximal Domain of IgE memory B cell Prati Moira, Rhyner Claudio, Crameri Reto Swiss Institute for Allergy and Asthma Research (SIAF), CH-7270 Davos, Switzerland E-mail: moira.prati@siaf.uzh.ch Background: In industrialized countries approximately 30% of the population is affected by IgE-mediated allergies, including asthma, atopic dermatitis or allergic rhinoconjunctivitis. Allergen-specific IgE is the key molecule in allergic diseases and it can be expressed either as secreted IgE (sIgE) or as membrane-bound form (mIgE), which contains two additional domains termed M1 and M2. The M1 region codes for an extracellular membrane-proximal domain (EMPD) and for the transmembrane domain, whereas the M2 region codes for the cytoplasmic domain expressed only in memory B cells as integral part of the B cell receptor (BCR). Aims: We think that specific targeting of mIgE on memory B cells is a useful strategy for prophylactic therapeutic interventions. With this study we aim therefore to demonstrate that anti-EMPD mAbs can be used to isolate IgE-switched memory B cells from blood of allergic patients. Results: One anti-murine and three anti-human hybridomas, were selected for further characterization and for production and purification of the corresponding monoclonal antibodies. The mAb specific for the human EMPD domain with higher specificity and affinity (KD ~ 10-10), was used to sort IgE+ memory B cell from allergic patients’ PBMC’s. Usually the frequency of IgE+ B cells is very low. Preliminary results have shown that about 0.3 % of the total B cell can be targeted with the monoclonal hAbC20. Conclusions: With this approach we want to generate clones producing human allergen-specific mAbs of the IgE isotype by immortalization in order to elucidate IgEbinding epitopes by co-crystallization of allergens and allergen-specific human Fab fragments. These initial studies can facilitate the discovery and development of new therapeutics, vaccines, and diagnostics, thus providing new insights into allergyrelated disorders. Cytosolic vacuolization of eosinophils: Involvement of CD11b Susanne Hoesli1 , Dagmar Simon 1,3, Beata Styp-Rekowska2 , Ruslan Hlushchek 2, Shida Yousefi 1, Hans-Uwe Simon 1 Institutes ofPharmacology 1 and Anatoml, University of Bern, and Clinic ofDermatology3 , Inselspital, University Hospital Bern Eosinophils are a subset of granulocytes involved in the pathogenesis of different diseases, including parasitic infections and allergies. An eosinophil infiltration has been observed in skin biopsies from patients suffering from a broad range of skin disorders of allergic, autoimmune, or neoplastic genesis. In samples derived from these patients, a fraction of eosinophils shows the distinct morphology of cytosolic vacuolization. Here, we performed in vitro experiments aiming to understand the molecular mechanisms and functional consequences of vacuole formation in eosinophils.Cytosolic vacuole formation was associated with activation and degranulation of eosinophils. Moreover, vacuolization was dependent on CD11b, an integrin expressed on eosinophils mainly upon activation and transmigration through endothelial cells. Firm adhesive processes, in which CDllb is involved, have been linked to a process called cytolysis, a form of degranulation leading to the loss of nuclear and cellular integrity and consequent cell death. We demonstrate that the progression of cytosolic vacuolization leads to eosinophil death characterized by loss of nuclear and cellular integrity. We thus suggest a morphological progression of cytosolic vacuolization to cytolysis. A System For Quantitative Production Of Murine Basophils Ex Vivo Ursina Gurzeler1, Tatiana Rabachini1, Clemens Dahinden2 and Thomas Kaufmann1 1 2 Institute of Pharmacology, University of Bern, Bern, Switzerland Institute of Immunology, University of Bern, Bern, Switzerland Basophils are the least common granulocytes in the circulation. They have recently been recognized as important immunomodulatory cells and play a major role as effector cells in allergic inflammation. However, functional and biochemical assays on basophils have long been hampered due to their extremely low number in the blood (<1% of peripheral blood leukocytes in the mouse). Consequently, very little is known about the regulation of apoptotic signalling pathway in basophils. In addition, basophils are short-lived cells with an expected half-life of approximately a few days. For these reasons, we established a system for quantitative production of murine basophils in vitro. We modified the method published by Wang et al. (Nat Meth 2006), where they described a method for the quantitative production of macrophages or neutrophils using conditional Hoxb8. Besides different cytokine cocktails, we use a 4hydroxytamoxifen-inducible lentiviral system for conditional Hoxb8 expression (Vince et al. Cell 2008). In order to generate Hoxb8-immortalised, basophilcommitted myeloid progenitor cell lines, we infect an enriched fraction of bone marrow-derived multipotent cells with a conditional Hoxb8 expression construct. This progenitor cell line is genetically stable (immortalised but not transformed), can be maintained in culture for a long time and progenitors can be differentiated into mature basophils in near unrestricted number upon shutdown of Hoxb8 expression Importantly, after differentiation, these cells exhibit many characteristics of mature murine basophils. They stop cycling, have multilobed nuclei, exhibit mouse basophil specific surface markers (c-kit -/FcεRIhi), express high-levels of basophil specific mast cell proteases 8 (Mcpt8) and MCP-11, degranulate in response to IgE crosslinking and start to produce IL4. To our knowledge, this is the first report presenting an in vitro model to produce large numbers of mouse basophils. This system is important not only to study the molecular mechanisms of basophil differentiation, but also because of the possibility of using genetically modified mice to further investigate basophil biology. In terms of apoptosis signalling, we have started to look into the role of BH3-only proteins (Bid, Bim) in death receptor-induced and mitochondrial apoptosis. Patrick Buschor1, Fabian Zellweger1, Alexander Eggel1, Michael Stumpp2, Beda M. Stadler1, Monique Vogel1 1 University Institute of Immunology, Inselspital, CH-3010 Bern, Switzerland Molecular Partners AG, Wagistrasse 14, 8952 Zürich-Schlieren, Switzerland 2 The high affinity receptor for human IgE (FcεRI) is expressed on the surface of mast cells and basophils. Allergen-induced cross-linking of FcεRI via receptor bound IgE stimulates the degranulation of pro- inflammatory mediators that induce the allergic symptoms. These effector cells also express the low affinity IgG receptor FcγRIIb that is known to inhibit allergeninduced cell degranulation when cross-linked with FcεRI. Considering the inhibitory function of FcγRIIb, we previously developed a bispecific molecule to efficiently aggregating FcεRI-bound IgE with FcγRIIb on the surface of basophils. The fusion protein is based on an IgE reactive DARPin (designed ankyrin repeat protein) that was linked to the Fc-part of a human IgG1 antibody for binding to FcγRIIb. This protein was not anaphylactogenic and inhibited allergen-induced basophil activation in whole blood assays. In order to increase the inhibitory effect the Fc part of the fusion protein was engineered by site directed mutagenesis. Through a single point mutation within the IgG1 CH2 domain (S267A) the binding affinity to FcγRIIb was improved 10-100 times, whereas the binding to IgE was not affected. Interestingly, inhibition of allergen-induced basophil activation in whole blood assays was more efficient with the altered construct. A long incubation time prior the allergen stimulation did not seem to be crucial for the inhibition. Furthermore the inhibitory effect was still present when the construct was applied after allergen exposure. The effect of the molecule to inhibit de novo synthesis of lipid mediators (e.g. Leukotriene) and cytokines is currently tested in purified human basophils. File: /home/jason/Desktop/Presentations/modwolfsberg-abstract.txt Page 1 of 1 Computational, Biochemical and Cellular Elucidation of Non-hapten Pharmaceutical Interactions (p-i) Adverse Drug Hypersensitivity in Watkins Our study began with computational screening of 10 T Cell Clones (TCC) isolated from a patient showing macular papular exanthema caused by sulfamethoxazol (SMX), using AutoDock and varied Docking software. All of these clones had been shown to produce some proliferative response to SMX stimulation prior to our initial computational analysis, which was dependent on the HLA DRB10 from the patient. All TCC T Cell Receptors (TCR) had been sequenced and models were generated from these. Initial DOCKing screens with SMX showed 2 hapten restricted clones, and 2 clones that gave binding sites for the compound at nonpeptide groove interacting sites. From these we chose 1 hapten restricted (clone 1) and one p-i clone (clone 2) to further characterize interaction mechanisms and kinetics. Our initial screening was expanded to 11 other SMX derivatives, showing a cross reactive profile for the p-i clone and a non-cross reactive profile for the hapten restricted clone. We were however able to demonstrate a competitive inhibition in Ca2++ assays to the clone 1, but not clone 2. From the original generated models, we began computational screening of peptides recognized by the respective TCR, and were able to generate a molecular dynamics simulation for clone 2 based on these results. Initial results from these runs give an estimated binding affinity and reaction mechanisms in the presence and absence of SMX. Together, these data indicate that Adverse Hypersensitivity reactions may not be caused solely by hapten restricted clones but may include a immix of other p-i interactions which yield the same outcomes. We also show that haptens may not be necessary for reactions to occur to specific drugs when a person has already been primed and generated a TCR against a hapten. The problem now remains to determine which of these reactions are the root cause of the adverse reactions, weather it is a combined effect of both interactions within an allergic individual, or whether it can be either type of molecular interaction. Session 4 Adaptive immunity in infection Direct type I IFN signaling on CD8 T cells protects them from NK cell mediated killing Josh Crouse and Annette Oxenius Institute of Microbiology, ETH Zurich josh.crouse@micro.biol.ethz.ch Direct type I IFN signaling on CD8 T cells is necessary for the proper expansion, differentiation and survival of responding CD8 T cells following certain viral infections such as during lymphocytic choriomeningitis virus (LCMV) infection. The reasons for the reduced survival of CD8 T cells lacking the type I IFN receptor (IFNAR-/-) following an acute LCMV infection remain unclear. Using an adoptive co-transfer system of WT and IFNAR-/- CD8 T cells followed by an acute LCMV-W E infection we have found that the ability to sense type I IFN by CD8 T cells protects them from natural killer (NK) cell mediated killing. In an NK cell sufficient environment IFNAR-/CD8 T cells exhibit reduced survival compared to WT CD8 T cells, but depletion of NK cells prior to LCMV-WE infection restores the expansion potential of IFNAR-/CD8 T cells up to day 4 post infection. This immunoregulatory role of NK cells is mediated in a perforin dependent manner and requires the activation of CD8 T cells. Therefore, following an infection associated with early and abundant levels of type I IFN, direct sensing of the proinflammatory cytokine by CD8 T cells protects them from NK cell mediated killing. This may serve as an immunoregulatory mechanism where improperly activated CD8 T cells are eliminated. XXIV Meeting of the Swiss Immunology PhD students Wolfsberg, 2-4 April 2012 Shaping of the immune response in persistent viral infections Sandra Kallert 1,2 and Daniel D. Pinschewer 1,2 1 2 Department of Pathology and Immunology, University of Geneva, 1 rue Michel Servet, 1211 Geneva 4, Switzerland World Health Organization Collaborating Center for Vaccine Immunology, University of Geneva, Switzerland Persistent infection of mice with lymphocytic choriomeningitis virus causes direct immunopathology and generalized immunosuppression. Mice acquiring LCMV infection at birth, however, develop a healthy life-long carrier state with impaired innate but intact adaptive immune responses. During this study we intend to analyze the molecular mechanisms by which chronic LCMV infection can shape the immune system. We are particularly interested in the influence of LCMV infection on dendritic cells (DCs) and stromal fibroblastic reticular cells (FRCs). DCs are known main players in anti-viral immunity whereas recent studies also emphasize the importance of FRCs in this context. Here we aim at analyzing how LCMV alters the gene expression profile of infected cells, resulting in modified immunocompetence and pathology. We can track infected cells using different viral reporter systems, which allow us to isolate infected and uninfected cells from the same animal. Whole genome cDNA expression analysis of infected and uninfected DCs and stromal FRCs will subsequently provide us with candidate host genes accounting for viral immunosuppression in chronic LCMV infection. Taken together this approach should give us new insights into how chronic viral infections can imprint host cells of both hematopoietic and non-hematopoietic origin leading to suppressed immune responses. Such knowledge may ultimately lead to new strategies and molecular targets to counteract viral persistence and virus-induced immunosuppression. Glutathione Peroxidase 4 is pivotal for maintenance of peripheral CD8+T cell pool and anti-viral CD8+ T cell responses Mai Matsushita1, Peter Bretscher1, Christoph Schneider1, Stefan Freigang1, Jan Kisielow1, Marcus Conrad2, Georg Bornkamm2, Manfred Kopf1 1 Molecular Biomedicine, Institute of Integrative Biology, ETH Zurich, Schlieren, Switzerland 2 Helmholtz Center Munich, Institute of Clinical Molecular Biology and Tumor Genetics, Germany The selenoenzyme glutathione peroxidase 4 (Gpx4) is regarded as a major scavenger of phospholipid hydroperoxides and thus represents a key component of the reactive oxygen species (ROS) scavenging network. In our studies, we study on the role of Gpx4 T cells, dendritic cells and macrophages using conditional knockouts, as the underlying molecular mechanisms still remain undefined. Focusing on T cells, our results from CD4cre-Gpx4lox/lox mice have revealed that despite normal thymic T cell development, CD8+ T cells failed to maintain a competitive pool size in the peripheral lymphoid organs resulting in homeostatic imbalance of naïve CD8+ T cells. Reconstitution analyses from mixed bone marrow chimera have further confirmed an intrinsic defect in CD8+ T cell survival in the periphery. CD4creGpx4lox/lox thymocytes have revealed the ability to expand properly yet failed to sustain survival upon transfer into RAG deficient mice. Furthermore, in vitro culture of both CD4+ and CD8+ T cells equally displayed rapid cell death, which was reverted upon addition of antioxidant Vitamin E. Under inflammatory conditions, Gpx4 plays a vital role in proper CD8+ T cell function and viral clearance as CD4cre-Gpx4lox/lox mice failed to mount efficient antiviral CD8+ T cell responses upon infection with LCMV and exhibited delayed viral clearance. However, the impaired virus-specific CD8+ T cell response was partially rescued upon diet supplementation with high concentrations of Vitamin E, thereby indicating the increased toxicity of accumulating phospholipid hydroperoxides as a mechanism. IL-21 restricts virus-driven Treg cell proliferation to prevent T cell exhaustion and viral persistence in chronic LCMV infection Iwana Schmitz1, Anja Fröhlich1, Helge Frebel2, Tim Sparwasser3, Annette Oxenius2, Stefan Freigang1,4, and Manfred Kopf1,4 1) Molecular Biomedicine, Institute of Molecular Health Sciences, Department of Biology, ETH Zurich, Switzerland 2) Institute of Microbiology, Department of Biology, ETH Zurich, Switzerland 3) Institute of Infection Immunology, TWINCORE/Centre for Experimental and Clinical Infection Research, Hannover, Germany 4) These authors contributed equally to this work. Foxp3+ regulatory T (Treg) cells are essential for the maintenance of immune homeostasis and tolerance; however, their impact on infectious diseases can be a double-edged sword and remains to be defined. During viral infections, Treg cells limit the immunopathology resulting from excessive inflammation, yet potentially inhibit effective antiviral T cell responses and promote virus persistence. We report here that LCMV Docile triggers a massive expansion of the Treg population that directly correlates with the size of the virus inoculum and its tendency to establish a chronic infection. This Treg cell proliferation was greatly enhanced in IL-21R-/- mice, suggesting that IL-21 restrains the virus-induced Treg cell expansion. Moreover, experimental augmentation of Treg cells drastically impaired the functionality of the antiviral T cell response and impeded virus clearance. As a consequence, mice became highly susceptible to chronic infection, even with a viral isolate that otherwise fails to establish persistence in immunocompetent hosts. These findings reveal virus-driven Treg cell proliferation as an immunoevasive strategy that facilitates T cell exhaustion and virus persistence. Furthermore, they suggest a dual role for IL-21 in chronic infections by providing survival signals to antiviral CD8+ T cells, but also by restricting the suppressive activity of infection-induced Treg cells. IL-9 is a potent suppressor of antiviral T cell responses Christoph Schneider 1, Jan Kisielow 1, Jean-Christophe Renauld 2, Manfred Kopf 1 1 2 Molecular Biomedicine, ETH Zürich Ludwig Institute for Cancer Research and Experimental Medicine Unit, Université Catholique de Louvain, Brussels, Belgium Interleukin-9 (IL-9) belongs to the common gamma-chain (γc) receptor cytokine family. Members of this family such as IL-2, IL-4, IL-7, IL-15 and IL21 are important regulators of T cell immunity. IL-9 has been primarily associated with Th2-type immune responses. More recently, IL-9 was proposed to enhance the function of regulatory T cells and facilitate the expansion of Th17 cells. However, the direct effects on T cell effector functions are not well understood. To investigate the role of IL-9 in the regulation of T cell responses, we studied IL-9 in the context of LCMV infection in mice. Using mice deficient in the IL-9 receptor, we could show that IL-9 signaling is a potent suppressor of LCMV-specific T cell responses. Upon infection with LCMV docile, IL-9R knockout mice show a more activated phenotype, produce more effector cytokines and eventually clear an otherwise persistent LCMV infection within 2 weeks. Moreover, IL-9 supplementation during infection results in the suppression of the antiviral CD8 T cell response and prolonged presence of the virus in the mice. These results provide evidence for a new function of IL-9 in regulating T cells by suppressing their effector function. 3 Key Words: IL-9, LCMV, T cell regulation Recovery of functional T-cells from LCMV clone 13 infected mice Daniel Utzschneider, Lucie Carrie, and Dietmar Zehn Swiss Vaccine Research Institue, IAL, Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland Persisting viral infections go along with the appearance of antigen-specific T-cells that show different degrees of functional impairments. These cells up-regulate inhibitory molecules such as PD-1, Lag-3, and Tim-3 and given their reduced ability to proliferate and produce cytokines in response to antigen stimulation they are often considered as exhausted T-cells. However, T-cell exhaustion progresses to different levels resulting in a very heterogeneous population of virus-specific T-cells. So far, it remained unclear to what extent exhausted T-cells might be able to regain functionality and if such a recovery potential could be associated with different levels of exhaustion. We address this question by transferring exhausted LCMV-specific CD8 T-cells into naïve mice and re-challenge with LCMV strains causing a chronic or an acute infection. We observed that the exhausted T-cell population robustly expands after a second antigen challenge. Moreover, we noticed that the extent of this re-expansion varies for different LCMV epitopes. By using modified LCMV Clone 13 strains, which differ in the affinity towards P14 transgenic CD8 T-cells, we were able to show that the functional avidity of a pMHC-TCR interaction impacts the level of T-cell exhaustion suggesting that differences in the strength of stimulation could be responsible for differences in the recovery potential. Thus, our data show that the fate of antigen-specific T-cells in a chronic infection is not finally terminated and that the population of exhausted T-cells comprises a substantial recovery potential. These results obtained by us broaden our basic knowledge of T-cell dynamics in persistent infections. THE ROLE OF “T HELP” DURING SECONDARY CD8+ T CELL RESPONSES IN ACUTE VIRAL INFECTIONS Gregor Bedenikovic, Melanie Wiesel, Annette Oxenius Swiss Federal Institute of Technology Zurich, Institute of Microbiology, gregor.bedenikovic@micro.biol.ethz.ch CD8 T cells represent an important part of the adaptive immune system as they are essential for the eradication of intracellular pathogens. They have the capacity to differentiate into long-lived memory cells, which allows them to resolve subsequent infect ions with the same pathogen. Upon re-exposure to their cognate antigen, these memory cells quickly expand to high numbers of effector T cells that control secondary infections faster and more efficient ly than CD8 T cells during the primary response. It was shown that CD4 T cells play a crucial role during primary CD8 T cell responses and also during the generation and recall responses of memory CD8 T cells. However, the timing and mechanisms by which these cells and other factors influence the mentioned pro cesses may differ between primary and secondary responses and they may even differ between different experimental models. Thus, the function of CD4 T cells and the role of "T help" during recall responses remains incompletely defined. Therefore, the goal of this project is to further examine the in vivo role of CD4 T cells and “T help” during secondary CD8 T cell responses, in the setting of acute viral infections. Special emphasis will be given to unravel the molecular mechanisms and timing of T help that support CD8 T cell recall responses (in comparison to the situation during primary CD8 T cell responses). In addition the contribution of APCs, different soluble mediators (such as IL-2, IL12, IL-15 or Type I IFN) and other cellular components (such as CD40/CD40L signalling, OX40 or PD-1) for CD8 recall responses will be defined in more detail. Tuning TCR avidity of virus-specific CD8+ T cells Three key-Words: Cytotoxic T cells, Mouse hepatitis virus, T cell receptor Jovana Cupovic1, Veronika Nindl1, Lilian Staerck 2, Wolfgang Uckert 2, Thomas Rülicke3 Volker Thiel1, Burkhard Ludewig1 1 Institute of Immunobiology, Kantonal Hospital St. Gallen, Switzerland Max Delbrück Center for Molecular Medicine Berlin, Campus Buch, Germany 3 Institute of Laboratory Animal Science, University of Veterinary Medicine, Vienna, Austria 2 Molecularly well-defined infection models can contribute to a better understanding of the basic mechanisms underlying interactions between the virus and the host immune system. The mouse hepatitis virus (MHV) is a cytopathic mouse pathogen able to establish infection in liver and CNS whereby CD8 + T cells are crucial for the control of acute MHV infection. To allow comprehensive studies of the role and behavior of MHV A59-specific CD8+ T cells, T cell receptor (TCR) retrogenic and transgenic mice were generated. Retrogenic TCR technology facilitated expression of variable numbers of TCRs on CD8+ T cells. According to TCR densities, retrogenic cells were classified as low, intermediate and high avidity T cells. Assessment of proliferative responses (judged by CFSE dilution) revealed faster onset of proliferation in high avidity retrogenic TCR T cells and a higher sensitivity of these cells to antigen. However, differences of TCR numbers had no influence on minimal dose of antigen needed for the initiation of proliferation. Using adoptive transfer of virus-specific CD8+ T cells to C57BL/6 mice revealed that both optimal avidity retrogenic and transgenic CD8+ T cells protected the recipients from virus-induced liver disease. However, during MHV CNS infection adoptively transferred TCR retrogenic and transgenic MHV-specific CD8+ T cells exacerbated virusinduced CNS disease. Altering the avidity of virus-specific CD8+ T cells will facilitate detailed analyses of CD8+ T cell activation thresholds and differentiation patterns during systemic and CNS virus infection. A murine genital-challenge model is a sensitive measure of protective antibodies against human papillomavirus infection Stéphanie Longet1, Martine Bobst1, John Schiller2, Patrice Jichlinski1, Denise NardelliHaefliger1 Department of Urology, CHUV and University of Lausanne, Lausanne, Switzerland1 ; National Cancer Institute, NIH, Bethesda, MD, USA2. The available virus-like particle (VLP)-based prophylactic vaccines against specific HPV types afford close to 100% protection against the type-associated lesions and disease. Based on papillomavirus animal models, it is likely that protection against genital lesions in humans is mediated by HPV type-restricted neutralizing antibodies that transudate or exudate at the sites of genital infection. However, a correlate of protection was not established in the clinical trials because few disease cases occurred, and true incident infection could not be reliably distinguished from the emergence or reactivation of prevalent infection. In addition, the current assays for measuring vaccine-induced antibodies, even the gold standard HPV pseudovirion (PsV) in vitro neutralization assay, may not be sensitive enough to measure the minimum level of antibodies needed for protection. Here, we characterize the recently developed model of genital challenge with HPV PsV and determine the minimal amounts of VLP-induced neutralizing antibodies that can afford protection from genital infection in vivo after transfer into recipient mice. Our data clearly demonstrate that, remarkably, the in vivo assay is substantially more sensitive than in vitro PsV neutralization and thus may be better suited for studies to establish correlates of protection. Antigen presenting cells involved in Th17 cell priming during Candidiasis Kerstin Weidner, André Gladiator and Salomé LeibundGut-Landmann Institute for Microbiology ETH Zürich, Wolfgang-Pauli-Strasse 10, HCI G435, 8093 Zürich, Switzerland kerstin.weidner@micro.biol.ethz.ch The fungus Candida albicans lives as part of the normal microflora in healthy individuals without triggering any harmful effects. However, it can cause severe disease in immunocompromised individuals. The increased prevalence of mycoses such as orophangyngeal candidiasis (OPC) in patients with profound T cell deficiencies provides evidence for CD4+ T cells playing a key role in protection from fungal diseases. C. albicans-specific CD4+ T cells are primed efficiently in response to oropharyngeal infection in mice and these T cells produce high levels of IL-17A and other Th17-type cytokines. However, it remains unclear how the differentiation of Th17 cells during OPC is controlled. Here we thus address the question which subset(s) of antigen presenting cells (APCs) mediate this process. Among others, inflammatory monocytes infiltrate the draining lymph nodes in high numbers during infection, suggesting that they mediate the priming of Candida-specific Th17 cells, either directly or in cooperation with lymph node-resident APCs. Inflammatory monocytes indeed present Candida-derived antigens in the draining lymph nodes of infected mice as they are able to activate Candida-specific T cell hybridoma ex vivo. The role of inflammatory monocytes for the activation of Candida-specific T cells during OPC in vivo is being confirmed. Together, this study shall illuminate and extend the prominent function of inflammatory monocytes as activators of the adaptive immune system that has recently been attributed to them in various tissues, including the lung, but not yet in the oral cavity, which is another prominent site of pathogen entry. THE IMPACT OF HIV-1 REPLICATION ON THE ACTIVATION OF CD8+ T CELLS WITH SPECIFICITIES FOR DIFFERENT ANTIGENS Sonia Bastidas, Huldrych Guenthard and Annette Oxenius Institute for Microbiology, Zürich, ETH Zürich, sonia.bastidas@micro.biol.ethz.ch Continuous loss of CD4+ T lymphocytes and chronic immune activation are hallmarks of chronic HIV-1 infection. It is suggested that apoptosis (activation induced cell death) of these abnormally activated cells is one of the main causes of T-cell decline. Interestingly, T cells with an activated phenotype are neither necessarily HIV-specific nor HIV-infected. Usually 1-2% of all circulating CD8+ T cells during chronic infection are HIV specific whereas the level of CD8+ T cells exhibiting activated phenotypes (as judged by CD38 and HLA-DR expression) is in the range of 60%, indicating that immune activation is not restricted to HIV-specific CD8+ T cells. Several hypotheses have been forwarded to explain the mechanism of CD8+ T cell activation. It is believed that most of these cells are activated by nonspecific mechanisms, including cross-reactivity and cytokine-driven activation, a phenomenon referred to as bystander activation. The mechanisms underlying this phenomenon are not well defined. Additionally, little to nothing is known about the antigen-specificity of bystander activated CD4+ and CD8+ T-cells. In this study we analyzed in a cohort of patients if cessation of antiretroviral therapy (ART) specifically leads to activation of CD8+ T cells with specificities for different antigens such as EBV, CMV, Adenovirus and Flu. Based on our data we propose that HIV-driven activation occurs for CD8+ T cells, and that they are getting activated in an antigen independent manner. These cells are likely to represent a large part of activated CD8+ T cells in untreated HIV infection and thereby significantly contribute to chronic immune activation. XXIV Meeting of the Swiss Immunology PhD students Wolfsberg, 2- 4 April 2012 Co-stimulatory effect of the HIV-1 envelope glycoprotein on CD4+ T cells during chronic HIV-1 infection Kathrin Zimmermann, Manuela Rehr, Anna Haas and Annette Oxenius kathrin.zimmermann@micro.biol.ethz.ch Institute of Microbiology, ETH Zürich Continuous loss of CD4+ T lymphocytes and systemic immune activation are the two major hallmarks of chronic HIV-1 infection. Chronic immune activation is associated with hyperactivation of T cells, reflected by the acquisition of an activated T cell phenotype and enhanced T cell turnover. Since the majority of activated CD4+ T cells is neither HIV-specific nor HIV-infected, it is currently believed that non-specific activation of CD4+ T cells occurs in the context of chronic HIV-1 infection. So far, mechanisms of non-specific T cell activation are poorly investigated and are currently attributed to pro-inflammatory cytokines and Toll-like receptor ligands which are both supposed to be elevated in the context of HIV-1 infection. In this study, we suggest a new mechanism of virus mediated CD4+ T cell costimulation during HIV-1 infection. Our in vitro experiments demonstrate that HIV-1 envelope glycoprotein gp120 (HIV-1 gp120) binding to the CD4 receptor of CD4+ T cells in temporal proximity of suboptimal T cell receptor (TCR) stimulation enhances their activation. We observe up-regulation of early T cell activation and exhaustion markers as well as increased levels of proliferation. We are currently investigating how HIV-1 gp120 ligation of the CD4 receptor affects proximal TCR signaling by reverse phase protein array technology. In order to validate the HIV-1 gp120 costimulation on a cellular level we aim to establish an assay in which HIV-1 gp120anti-gp120 immune complexes bound to dendritic cells might co-stimulate CD4+ T cells in the presence of suboptimal TCR stimulation. Session 5 Selection and autoimmunity About the Role of Dicer-dependent miRNA in the Thymus Carlos Mayer1, Saulius Zuklys1, Saule Zhanybekova1, Alberto PascualMontano2 and Georg Holländer1,3 1 Pediatric Immunology, Department of Biomedicine, University of Basel and University Children’s Hospital Basel (UKBB), Switzerland 2 Functional Bioinformatics Group, National Center for Biotechnology-CSIC, Universidad Autónoma de Madrid, Spain 3 Developmental Immunology, Department of Pediatrics, University of Oxford, United Kingdom The thymus stromal microenvironment is mainly comprised of two, namely cortical and medullary, subsets of thymic epithelial cells (TEC) that in aggregate provide immature T progenitor cells with the cell bound and soluble factors required for their commitment and maturation to fully functional T-cells. MicroRNA (miRNA) are small, non-coding nucleotides that have been implicated in many different aspects of biology, including morphogenesis and tissue maintenance. To investigate the role of miRNA in TEC development and function, we established a mouse model with a thymus epithelial targeted, conditional deletion of the miRNA-processing enzyme Dicer. Mice exclusively deficient for Dicer expression in TECs revealed severe defects in the composition and structure of their thymus microenvironment as demonstrated by an early and progressive loss of cortical and medullary thymus epithelial and significant changes in their phenotype. Consequently, these alterations compromised the commitment of T cell precursors to a T-cell fate and affected positive and negative thymocyte selection already at a stage when a physical loss of TECs was not yet detected. As a result, central tolerance became severely affected allowing for the generation of a repertoire of mature T-cells able to mediate overt autoimmunity. With age, the thymus of gene-targeted mice gained the structural features typical of secondary lymphoid tissues and supported in situ B cell development. In summary, our results demonstrate the importance of miRNA for TEC survival and function. The amount of self-antigen determines the effector function of murine T cells escaping negative selection Lee Kim Swee*,2,3, Anja Nusser*,2, Maurus Curti*,4, Matthias Kreuzaler*, Hannie Rolink*, Luigi Terracciano†, Fritz Melchers§, Jan Andersson* and Antonius Rolink* * Developmental and Molecular Immunology, Department of Biomedicine, University of Basel, Basel, Switzerland † Department of Pathology, University Hospital of Basel, Basel, Switzerland § Max Planck Institute for Infection Biology, Berlin, Germany 2 These authors contributed equally 3 Current address: Whitehead Institute, Cambridge, MA, USA 4 Current address: Viollier AG, Basel, Switzerland Autoimmune diseases develop when self-specific T cells that escaped negative selection initiate a harmful immune response against self. However, factors which influence the initiation and progression of an autoimmune response remain incompletely understood. By establishing a double transgenic mouse system in which dendritic cells express different amounts of a cellsurface neo-self antigen we demonstrate that antigen dose dramatically influences T cell tolerance mechanisms. Moderate antigen expression by dendritic cells favors the development of antigen-specific regulatory T cells and the establishment of a tolerogenic environment. In marked contrast, high dose of antigen expression by dendritic cells results in poor development of antigen-specific regulatory T cells and the early onset of anemia and splenomegaly and the late development of arthritis and high titers of IgG auto-antibodies. Disease is initiated by autoreactive T cells, which escape negative selection by expressing a second non-autoreactive T cell receptor. Transfer of antigen-specific regulatory T cells ameliorates the early onset signs of disease but does not prevent the development of long-term chronic pathologies. XXIV Meeting of the Swiss Immunology PhD students Wolfsberg, 2- 4 April 2012 Characterizing autoreactive CD8+ T cells that bypass negative selection Enouz S, Carrié L, Zehn D. Swiss Vaccine Research Institute, Centre Hospitalier Universitaire Vaudois, Service of Immunology and Allergy, Lausanne, Switzerland Central and peripheral tolerance mechanisms efficiently remove autoreactive T cells. However it has been shown that autoreactive CD8 + T cells that weakly react with selfantigen can bypass negative selection in the thymus and can be found in the periphery. Our aim is to precisely characterize these low avidity CD8+ T cells, study their selfdamaging potential, and their ability to circumvent central and peripheral tolerance. To answer these points we generated a new transgenic mouse model designated as OT-3 TCR transgenic mouse. Importantly, we can demonstrate that the OT-3 resemble the low avidity T cells found in the periphery of Rip-mOva mice and that the OT-3 can induce autoimmune diabetes. This indicates that we have generated a model that allows us to study the properties of auto-reactive T cells that bypass negative selection. OT-3 cells can also be recruited and activated when pathogens that encode crossreactive epitopes that are of lower affinity than the native ovalbumin antigen are introduced. These data define a threshold for activating T cells in the periphery that is below the threshold for negative selection. If these cells can be activated in the periphery upon low affinity epitopes highlights the existence of an affinity danger zone involved in the development of autoimmunity. XXIV Meeting of the Swiss Immunology PhD students Wolfsberg, 2- 4 April 2012 Rorγt+ innate lymphocytes and γδ T cells initiate psoriasiform plaque formation in mice S. Pantelyushin1*, S. Haak1*, B. Ingold2*, P. Kulig1 , Frank L. Heppner2, A. Navarini3, B. Becher1 1 – Institute of Experimental Immunology, University of Zürich, Switzerland 2 – Institute of Pathology, Charité University Hospital, Berlin, Germany 3 – Department of Dermatology,University Hospital Zürich, Switzerland 4 – Current address: Columbia University Medical Center, Dept. Microbiology & Immunology, 701 West 168th Street, New York, NY-10032, USA * - equal contribution Abstract: Psoriasis is a common, chronic, relapsing inflammatory skin disease characterized by erythematous scaly plaques. Histologically its manifestations are: keratinocyte hyperproliferation, elongated rete ridges and inflammatory infiltrates consisting of T cells, macrophages, dendritic cells and neutrophils. Despite the availability of new effective drugs to treat psoriasis, there are still many unknowns regarding disease pathogenesis. Recent studies have shown that Aldara cream (5% Imiquimod), used to treat benign skin abnormalities, triggers psoriasis-like disease in humans and mice. In both cases the IL-23/IL-17 pathway is implicated in the disease pathogenesis, with TH17 cells thought to be the main players. Using this model ofAldara psoriasiform plaque formation in mice, we could establish a predominant role of the TH17 signature cytokines, IL-17A, IL-17F and IL-22, in this process. Loss of either of these cytokines in gene-targeted mice strongly reduced disease severity. The main source of these cytokines, however, was not TH17 cells but a skin-invading population of γδ T cells and innate lymphocytes. Furthermore, our findings establish that RORγt+ innate lymphocytes are sufficient and necessary for psoriatic plaque formation in an experimental disease model that closely resembles early human psoriasis. Session 6 T cell activation / polarization Malt1 and Caspase-10: Proteases Essential for Lymphocyte Activation Jean Enno Charton, Fabien Rebeaud and Margot Thome Departement de Biochimie, Université de Lausanne, CH-1066 Epalinges, Switzerland. T cell activation via the T cell receptor (TCR) through antigen recognition is one of the key steps to initiate the adaptive immune response. The mechanisms of TCR-induced nuclear factor κB (NF-κB) and Activator Protein 1 (AP-1) are the subject of intense research since deregulated signaling in lymphocytes can lead to immunodeficiency, autoimmunity or lymphomas. In T lymphocytes, a complex composed of Carma1, Bcl-10 and Malt1 transfers signals from TCR proximal events to NF-κB activation. Gene translocations of the BCL10 or MALT1 genes or oncogenic mutations affecting CARMA1 result in constitutive NF-κB activation and are related to the development of certain forms of lymphomas. Here we report a cleavage of certain Caspase-10 isoforms by Malt1 in T and B cells after antigen receptor stimulation. Caspase-10 has so far only been shown to be involved in the induction of apoptosis and is closely related to Caspase-8. W e see a crucial role of the Caspase-10, but not Caspase-8, in T cell activation after T cell receptor stimulation in an IKK independent manner. Lack of Caspase-10 leads to abrogation of AP-1 activation, reduced NF-κB activation and a severe defect in IL-2 production, and thus drastically reduces T cell activation. W hether the cleavage is important for NF-κB or AP-1 activation and how Caspase-10 is involved in the signaling is the matter of ongoing research. Taken together, these results give new insights into lymphocyte activation and could lead to the development of new drugs modulating the immune response or inhibiting lymphoma formation. Cervical lymph nodes and the pivotal role in the CCR6+ T helper cell priming. Basso Camilla, Lanzavecchia Antonio and Sallusto Federica. Institute for Research in Biomedicine, Bellinzona, Switzerland. To perform their function, effector and memory T cells migrate to sites of antigen challenge. Previous studies have shown that CCR9 is expressed by T cells that migrate to the gut, while CCR10 is expressed on T cells that migrate to the skin. These receptors are induced in T cells by dendritic cells (DCs) that process vitamin A and D, which are selectively produced in the gut and skin, respectively, to their active metabolites for presentation with antigen to lymphocytes. Using the experimental autoimmune encephalomyelitis model, we have shown that pathogenic Th17 cells upregulate expression of CCR6 and use this receptor to cross the choroid plexus epithelium, which constitutively express the CCR6 ligand CCL20, to enter the CNS. Based on these data we hypothesized that CCR6 regulates constitutive migration of lymphocytes in the CNS and that this receptor may be induced in cervical lymph nodes (CLNs). Consistent with this hypothesis, we found that naive CD4+ T cells primed by antigens draining in the CLNs selectively upregulate CCR6 expression. In vitro experiments showed that CD4+ T cells cultured with cells isolated from CLNs also upregulate CCR6 expression. Migratory DCs seem to be responsible for CCR6 upregulation on T cells, since CCR6 was not induced in CLNs of CCR7 deficient animals which lack this DC subset. We are performing experiments to understand what are the signals required for induction of CCR6 expression in T cells primed in the CLNs and to define the cytokine profile of these CCR6+ CLN primed T cells. Priming of polarized human T cells by microbes Simone Becattini, Laurent Perez, Antonio Lanzavecchia, Federica Sallusto Institute for Research in Biomedicine, Via Vincenzo Vela 6, 6500 Bellinzona The innate immune system is endowed with invariant receptors that recognize pathogenassociated or danger-associated molecular patterns (PAMPS and DAMPS, respectively). These pattern-recognition receptors (PRRs) are highly expressed in antigen presenting cells (APCs) where they control the level of MHC and costimulatory molecules and the production of cytokines that contribute to T cell differentiation, such as IL-1β, IL-6, IL-12, and IL-23. Thus, it is thought that the combination of PRRs triggered by a given pathogen determines the class of CD4+ T cells elicited, i.e. Th1, Th2, or Th17. While most studies in humans have focused on the effect of single microbial components, much fewer dealt with the effect of whole microbes on APCs, especially on T cell priming. We therefore developed a combined in vitro/ex vivo approach to study the mechanisms of microbe-induced T cell priming and identify the PAMPS and the antigens associated with a given polarized T cell response using monocytes as APCs. Monocytes exposed to microbes prime in vitro naïve CD4+ T cells, leading to expansion of antigen-specific T cell populations with various phenotypes, despite the homogeneity of the culture conditions. The chemokine receptor pattern of in vitro primed T cells allows enrichment of specific T cell subsets, as it is the case for ex vivo analyzed memory T cells. Specific microbial components responsible for induction of a characteristic T cell response can be identified; in particular we found that C. albicans mannoprotein is a key component for induction of a Th17 response. Course of the Th-1, Th-2 and Th-17 Cytokines and of Associated C hemokines Involved in the Periparasitic Recruitment of Cells in Echinococcus multilocularis Infection Junhua Wang, Bruno Gottstein* Institute of Parasitology, University o f Bern, Bern, Switzer land Methodology/Principal Findings: Hepatic gene expression profiles were assessed using DNA microarray analysis, 1, 2, 3 and 6 months after injection of E. multilocularis metacestode into the liver of susceptible mice. Quantitative RT-PCR was used to describe the changes in the expression of 10 selected cytokine and chemokine genes from 2 days to 12 mo nths. Microarray analyses showed that CCL8, CCL12 and CCL17 were up -regulated 30fo ld, 6-fo ld and 3- fold at 1 month, respectively. CXC L-9 was up-regulated 3- fold at 6 months. Among genes o f cytokine receptors, only those for IL-1 (IL-R1 like) and IL-7 (at 1 month, 2.92 and 2.25 fold respectively), and IL-13 (IL-13 Rα1) and IL-17 (IL-17 R) were upregulated (2.39 at 3 months and 2.90 at 6 months respectively). Real time RT-PCR data showed that a Th1 immune response was predominant in the early stage, but shifted to Th2 in the middle/late stage. IL-17 mRN A increased from ~2.41-fold at day 2 to 6.58-fold at month 6 with a maximum expressio n at month 6. In the periparasitic infiltrate, positive immunostainings for IL17 were observed fro m day 8 to mo nth 12, and highly correlated with CD4 (r=0.864), C D8 (r=0.684) and CD4/8 ratio (r=0.524). Conclusions: Our data confirm that Th1, then later Th1/Th2 profiles characterize the local immune response during E. multilocularis infection at the early and middle/late stage, respectively; in addition, the marked expression of IL-17 and chemokines suggest that they may p lay an important role in cell homing to inflammatory reaction sites around the parasite vesicles. Peter Bretscher, Julian Egger, Erick M. Carreira, Manfred Kopf, and Stefan Freigang Oxidized phospholipid signaling in microbial infection and inflammatory disease Phospholipids are major constituents of cellular membranes that ensure structural integrity and provide a barrier to maintain the membrane potential. However, accumulating evidence also documents their important role in cellular signaling processes. In particular, oxidatively modified phospholipids (OxPL) that are generated from unsaturated phospholipids by reactive oxygen species exert a multitude of biological effects. OxPL are present at sites of inflammation in vivo and have been implicated in acute and chronic inflammatory conditions, such as microbial infections, acute lung injury, atherosclerosis and rheumatoid arthritis. However, the biologically active lipid species as well as the pathways that mediate their signaling still remain elusive. Here we have characterized the complex mixture of OxPL species generated during the non-enzymatic oxidation of the common phospholipid 1-palmitoyl2-arachidonyl-sn-glycero-3-phosphocholine (PAPC) and identified the isoprostane containing phospholipids 1-palmitoyl-2-(5,6)-epoxyisoprostane E2-sn-glycero-3-phosphocholine (PEIPC) and 1-palmitoyl-2-[(5R,6R,7E,12S)5,6-epoxyisoprostane A2]-sn-glycero-3-phosphocholine (PECPC) as the main components mediating the biological activities of OxPAPC. Using synthetic compounds we show that PEICP and PECPC modulate the production of proinflammatory cytokines by dendritic cells and macrophages. Furthermore, dendritic cells treated with these OxPL become licensed to polarize naïve CD4+ T cells towards Th2 differentiation. These effects directly correlated with the ability of OxPL to activate nuclear factor erythroid 2-related factor 2 (Nrf2), the transcriptional master regulator of the antioxidant response. Moreover, we demonstrate that structurally related endogenous lipids, e.g. the prostaglandin J2 (15d-PGJ2), also signal via this OxPL/Nrf2 axis, suggesting that OxPL induce their biological effects by mimicking the activity of endogenous signaling lipids. 24th Meeting of the Swiss Immunology PhD students, Wolfsberg, April 2 - April 4, 2012 Mechanisms of T cell stimulation by flucloxacillin in HLA-B*5701+ flucloxacillin-naïve individuals Natascha Wuillemin1, Jacqueline Adam1, Stefano Fontana2, Werner J. Pichler1, Daniel Yerly1 1 Clinic for Rheumatology and Clinical Immunology / Allergology, University Hospital of Bern, 3010 Bern, Switzerland 2 Regional Blood Transfusion Service of the Swiss Red Cross, 3008 Bern, Switzerland Purpose: Genetic studies identified HLA-B*5701 as risk factor for flucloxacillin (FLUX) induced liver injury. To better understand this side effect, we established an in vitro model for FLUX induced T cell activation. Methodology: Peripheral blood mononuclear cells from FLUX-naïve, HLA-B*5701+ healthy donors were cultured by iterative in vitro restimulations with FLUX. FLUX-specific T cells (FLUX-Tc) were characterized by flow cytometry (IFNγ, CD107a) and cytotoxicity assays. Results: FLUX-Tc could be generated in 7/7 healthy HLA-B*5701+ donors and were mainly CD8+. They react by IFNγ secretion and CD107a upregulation upon drug stimulation and exert cytotoxicity against autologous antigen presenting cells (APC). In most cases, FLUXpulsed APC were not able to activate FLUX-Tc. In fact, the addition of FLUX in solution was sufficient to fully activate FLUX-Tc even in absence of autologous APC. Moreover proteasome inhibition by lactacystin and bortezomib did not affect the Tc reactivity. Nevertheless, in a minority of FLUX-Tc the reactivity upon FLUX stimulation was reduced when APC were not pulsed with flucloxacillin in advance or when APC were not present at all. Conclusions: The observations that FLUX in solution can activate FLUX-Tc do not support the classical hapten theory, explaining the immunogenicity of drug through covalent binding to macromolecules. In contrast, FLUX-Tc reactivity is best explained by a direct interaction of FLUX with the restricting HLA-B*5701 molecule. Different ways of drug presentation may lead to distinct clinical manifestations of FLUX allergy and will further be investigated. Phenotype and function of pathogen-specific human CD4+ memory T cells Federico Mele1, Christina E. Zielinski1, Dominik Aschebrenner1, Silvia Monticelli1, Antonio Lanzavecchia1,2 and Federica Sallusto1 1 Institute for Research in Biomedicine, Bellinzona, and 2ETH Zurich, Switzerland Using high throughput cellular screening methods we are performing an analysis of the functional properties of human memory T cells specific for different pathogens. To do this we established libraries of human memory T cell subsets isolated from blood samples of immune donors according to the expression of CD45RO and chemokine receptors. The libraries were interrogated for reactivity against a wide panel of antigens from viruses, bacteria and fungi. We found that i) virus-specific T cells are present mainly in CXCR3+CCR6+ and CXCR3+CCR6– Th1 subsets; ii) T cells specific for bacteria are mainly present in the CXCR3+CCR6+ Th1 and CCR6+CCR4+ Th17 subsets; iii) T cells specific for Mycobacterium tuberculosis are almost exclusively present in the CXCR3+CCR6+ Th1 subset; iv) T cells specific for Candida albicans are present in the CCR6+CCR4+ Th17 subset and in some donors, are also present in the CCR4+ Th2 subset. C. albicans-specific Th17 clones were isolated and analyzed for their capacity to produce IL-17 at different time points following restimulation. We found that while resting clones produced high amounts of IL-17, day 5-activated clones strongly downregulated IL-17 production and then gradually regained the capacity to produce IL-17 as the cells reverted to the resting state. The analysis of transcription factors showed that on day 2 and 5 following restimulation, Th17 clones downregulated RORC mRNA expression, concomitant with downregulation of IL17 mRNA. Overexpression of RORt significantly restored IL-17 production in activated Th17 clones, and restimulation in the presence of a STAT5 inhibitor rescued RORC mRNA expression and IL-17 production in a proportion of clones. These data suggest that decreased RORt expression and increased pSTAT5 levels, contribute to the transient downregulation of IL-17 production in activated human memory Th17 cells. Taken together our findings reveal a novel type of self-regulatory mechanism and suppressive function of IL-2. Session 7 Immune regulation Role of Thymic stromal lymphopoietin (TSLP) signaling in intestinal immune homeostasis Ilaria Mosconi1, Markus B. Geuking2, Joanna C. Massacand1, Kathy D. McCoy2, Nicola L. Harris1 1) Laboratory of Intestinal Immunology, School of Life Sciences, EPF Lausanne, Switzerland. 2) Mucosal Immunology Lab, Maurice Müller Laboratories, University Clinic for Visceral Surgery and Medicine, University of Bern, Switzerland TSLP is a cytokine constitutively expressed by intestinal epithelial cells (IECs) which has been hypothesized to regulate intestinal inflammation and homeostasis. To dissect its role in modulating intestinal immunity we utilized TSLP receptor (TSLPR) deficient mice. We observed significant changes in immune cell populations of both the lymphoid tissues and intestinal lamina propria (LP) of TSLPR-/- mice: we found a higher proportion of interferon-γ (IFNγ) producing-CD8+ T cells and of IFNγ and interleukin (IL)-17 producing-CD4+ T cells. To determine whether dysregulated cytokine production was occurring as a result of pathogen infection or was in response to commensal intestinal bacteria we repeated our experiments in germ-free mice colonized with a minimal, and entirely commensal, bacterial community called the altered Schaedler flora (ASF). Following recolonization of wildtype mice a mutualistic TLR-dependent Treg response occurs in wild type mice, leading to a successful immune adaptation and establishment of host-microbial mutualism (Geuking et al, 2011). However, in presence of a defective Treg cell compartment, colonization causes an immune deviation to Th1 and Th17 cell responses. Consistent with our earlier experiments TSLPR-/- mice re-colonized with ASF bacteria exhibited an increased percentage of CD4+ T cells producing IFNg or IL-17. They also exhibited a lower percentage of Helios- Foxp3+ Tregs, indicating a defect in the expansion of inducible Tregs. Taken together, these data indicate that the mechanism by which TSLP impacts on colonic inflammation may involve a role for this cytokine in the activation or maintenance of Tregs in response to the commensal intestinal bacteria. IFN-a modulates the expression of the gut-homing receptor a 417 1,2 2 2 2 2 Nina Suhartha , Simon Heidegger , Nadja Sandholzer , Nicolas Stephan , David Anz , Stefan 2 Endres , Carole Bourquin 1 1 Chair of Pharmacology, Department Medicine, University of Fribourg, Switzerland 2 Department of Medicine, Division of Clinical Pharmacology, Ludwig-Maximilians-Unversity of Munich, Germany a417 is an integrin that is expressed at low levels on naïve T cells. Through interaction with its ligand MAdCAM-1, a4 17 enables naïve T cells to enter the gutassociated lymphoid organs (GALT) such as mesenteric lymph nodes and Peyer’s patches. Upon T cell activation in the GALT, a417 expression on T cells is upregulated allowing the T cells to migrate into the gut. Previous in vitro finding in our lab showed that Toll-like receptor (TLR) agonists decrease a417 expression on T cells. Similarly, I found that when poly I:C, a TLR3 ligand, is injected into mice, a417 expression on CD8+ T cells is reduced in all secondary lymphoid organs. This reduction was abolished when poly I:C was injected into IFNAR-/- knockout mice which lack the type I interferon receptor. When splenocytes are incubated with recombinant mouse IFN-a, a417 expression on T cells is significantly reduced. Likewise, when mice are treated with IFN-a, a417 expression on T cells in secondary lymphoid organs is downregulated. When IFN-a-treated splenocytes are transferred into mice, these cells show clearly an impaired migration into the GALT in comparison to the untreated splenocytes. Furthermore, splenocytes infected with the Sendai, EMCV or VSV viruses, show a decrease of a417 expression on the T cells. In conclusion, this project has revealed that IFN-a decreases a417 expression on naïve CD8+ T cells and blocks their migration to GALT. This may be a mechanism to prevent activated non-cognate T cells from initiating an overwhelming immune response in the gut. Primary and memory B cell responses to Qβ-VLP in mice Franziska Zabel, Alexander Link, Juliana Bessa, Philippe Saudan, Martin F. Bachmann Department of Dermatology, Zurich, University Hospital Zurich Long-lived humoral memory is induced by the interaction of B cells with their cognate antigen in the presence of T cell help through both cell bound (e.g. CD40L) and secreted molecules (e.g. IL-4, IFNγ or IL-21) which promote isotype-switching, B-cell proliferation and differentiation. In contrast to the well understood differentiation of naïve B cells into memory and/or plasma cells, relatively little is known about the fate of memory B cells upon exposure to antigen. To address this question, we used virus-like particles (VLPs) derived from the bacteriophage Qβ, which are highly immunogenic due to their structure. To trace memory B cells, congenic Ly5.1+ C57BL/6 mice were immunized with Qβ, splenic memory B cell isolated and transferred intravenously into naïve Ly5.2+ C57BL/6. 24 hours later recipient mice were challenged with Qβ. The transferred memory B cells homed to the spleen but did not proliferate as efficiently as naïve B cells after immunization not did they contribute effectively to the germinal centre reaction. In contrast, they differentiated more quickly into plasma cells and secreted antibodies at a higher level than plasma cells derived from naïve B cells. Thus, memory B cells are likely the main source of antibodies during secondary B cell responses. Session 8 Tumor immunity Ali Bransi, Alexander Knuth, Maries van den Broek University Hospital Zurich, Wagistrasse 14, 8952 Schlieren, Switzerland Ali.Bransi@usz.ch The tumour environment delivers suppressive signals to the immune system, which impair the effector function of tumour-infiltrating lymphocytes. The identity of these signals, their targets and their mode of action, however, are largely unresolved. Using mouse models of inducible cancer in a tissue specific fashion, we aim to identify mechanisms that locally suppress the function of tumour-specific T cells. We are establishing an inducible Cre/LoxP-based model system to express the immunologically well-characterized oncogene SV40 large T antigen with the luciferase in a tissue specific fashion. While waiting for the birth of the new transgenic mice, pilot studies were realized using the already available TRAMP mice. These mice develop progressive forms of prostate cancer with an early onset of the disease (12 weeks). The carcinogenesis is driven by the SV40 large T antigen (SV40LT) under the androgen-regulated rat probasin promoter to limit expression to the prostate. We observed rapid local and peripheral immune subversion of adoptively transferred tumour-specific CD8 T cells in tumour-bearing animals. Homing and proliferation were not affected, but effector functions of these cells were greatly reduced at all sites compared to controls. This immunosuppression was also more severe inside the tumour than in the periphery. In addition, we observed a striking difference in tolerance of CD8 T cells towards different epitopes of SV40LT. This project will give new knowledge about molecules and pathways that are crucial in deviating tumour-specific immunity. Impact of innate lymphocytes on the tumor microenvironment Sara Burkhard, Maya Eisenring, Burkhard Becher University of Zurich, Institute of Experimental Immunology The potent tumoricidal activity of interleukin 12 (IL-12) has been described in numerous studies. Previously our group has identified the mechanism by which IL-12 mediates tumor suppression in the subcutaneous layer. It was found that the IL-12-mediated repression of tumor growth within the skin surprisingly functions completely independent of T lymphocytes or NK cells. In contrast, by systematically analyzing the IL-12 mediated anti-tumor immunity in a series of mouse mutants, they discovered that the IL-12 responsive tumor-invading cell type is an innate, lineage negative leukocyte expressing the transcription factor RORγT. This was demonstrated with a subcutaneously inoculated melanoma cell line and attributes a novel function to this cell type in tumor immunology. The presence of these innate lymphoid cells (ILC) leads to the upregulation of the adhesion molecules ICAM-1 and VCAM-1 in the tumor vasculature and increased leukocyte invasion. To unravel the mechanism of action, the endothelial cell line MS-1 was cocultured with YFP+ RORγT+ ILCs, obtained from RORc-Cre x Rosa26-stop-eYFP mice deficient in Rag1. The in vitro adhesion molecule upregulation by the IL-12 administered ILCs was found to be dependent on IFNγ. Furthermore timelapse imaging of tumor cell lines and tumor explants will be performed. B16 melanoma cells stably transfected with tdTomatoPEST will be cocultured with YFP+ RORγT+ ILCs, which enables us to visualize tumor as well as effector cells. Augmentation of antitumor immunity applying a semi-allogeneic dendritic cell line Sergio Haller and Hans Acha-Orbea: Department of Biochemistry, University of Lausanne, Switzerland Dendritic cells (DCs) are master regulators of adaptive immune responses and tolerance. They efficiently capture, process and present to T cells, non- self and self-antigens such as pathogens and cancerous cells respectively. Although various approaches using DCs as therapeutic cancer vaccines have been established in murine models, they have met only limited success in clinical trials. Present publications are predominantly restricted monocyte or bone marrow-derived DCs which are limited by cell number, homogeneity and phenotype of the applied cells. A stable murine DC line has been established in our group previously. They were derived from CD11c promoter driven large T oncogene expressing transgenic mice. This allows the preparation of large batches of homogenous CD8α+ DCs and its application as a more standardized vaccine in terms of phenotype and maturation. This will give us a better insight of the fate and function of injected DCs within a tumor bearing host. We first increased the immunogenicity of the DC line using two different approaches: The cell line was modified in order to express MHC-I H-2Kd. We then demonstrated that in vitro these DCs are able to present tumor antigens to CD8+ lymphocytes also on BALB/c background and induce a Th1 response. Preliminary in vivo experiments indicate delayed tumor growth upon the injection of semi-allogeneic H-2Kd+ DCs when compared to autologous or full-allogeneic vaccination. We further transduced the DC lines to secrete the pro-inflammatory cytokines IL-2, IL-12 or IL-15. The combination of vaccination and local delivery of pro-inflammatory cytokines should induce a more effective anti-tumor immunity. Tumour therapy by redirected Fibroblast Activation Protein- specific CD8+ T cells Christian Hagedorn1, Alex Soltermann2, Maries van den Broek1, Chrisoph Renner1, and Ulf Petrausch3 1 Department of Oncology, University Hospital Zurich, 8091 Zurich, Switzerland Institute for Surgical Pathology, University Hospital Zurich, 8091 Zurich, Switzerland 3 Departent of Immunology, University Hospital Zurich, 8091 Zurich, Switzerland 2 Through introduction of a chimeric antigen receptor (CAR), T cells can be redirected to recognize tumour-associated antigens (TAAs). CARs consist of an antibody-derived domain (scFv) specific for a particular TAA plus intracellular signaling moieties of CD3 and CD28. Engagement of the CAR with the TAA will thus result in bona fide T cell activation. We aim to redirect T cells against Fibroblast Activation Protein-FAP- FAP- is a surface protein expressed by tumour stroma of the majority of human carcinomas as well as by sarcomas. In addition, in particular malignancies, tumour cells themselves express FAP-. In healthy adult tissue FAP-is only expressed by pancreatic islet cells, which makes targeting FAP- an attractive therapy for the treatment of cancer. Using a phage library, we selected two FAP- specific scFvs and generated CARs with thoseIn contrast with the well-known and clinically tested human FAP- specific F19 antibody, both newly selected scFvs (ESC11 and ESC14) recognized both human and murine FAP-. We expressed F19-, ESC11- and ESC14-CARs in human primary CD8+ T cells by retroviral transduction. Redirected human T cells against FAP-specifically lysed FAP-+ tumour cells in vitro. In addition, redirected FAP- specific human CD8+ T cells produced IFN- in vitro in a FAP- specific fashion. Next, we will test the therapeutic efficacy of FAP- specific redirected T cells using xenografted FAP-+ human tumours in NOD.SCID.c-/- mice. In addition, we will generate murine CARs with ESC11 and ESC14 and test their therapeutic potential in syngeneic mouse models in which the tumour stroma is FAP-+. XXIV Meeting of the Swiss Immunology PhD students Wolfsberg, 2- 4 April 2012 Modulation of astrocytoma-cytotoxic T lymphocyte interactions by chemotherapy Cristina Riccadonna and Paul R. Walker Geneva University Hospitals and Geneva University, Centre of Oncology Introduction Malignant astrocytoma is an aggressive brain tumour with poor prognosis, despite treatment (surgical resection, radiotherapy and chemotherapy). It is infiltrated by immune cells, including cytotoxic T lymphocytes (CTLs). The majority of astrocytoma cells are differentiated, whereas a small fraction (defined as astrocytoma initiating stem-like cells), is de-differentiated, reported to be chemoresistant, and is likely to be the cause of tumour recurrence. Here, we explore the potential of immunotherapy for astrocytoma. Aims 1) Can chemotherapy impact on astrocytoma-CTL interactions? 2) Can death pathways induced in astrocytoma by immune and chemotherapy mediators be synergistically combined? Astrocytoma model and preliminary results GL261 murine astrocytoma grown as adherent cultures are differentiated cells, whereas GL261 neurospheres are astrocytoma initiating stem-like cells. Neurospheres showed enhanced resistance to two different chemotherapeutic drugs: temozolomide, an alkylating agent, and paclitaxel, which stabilises microtubules and interferes with mitosis. Both adherent cells and neurospheres express MHCI, essential for recognition by CTL, and cytotoxicity tests confirmed sensitivity to CTL-mediated lysis. Perspectives CTL-tumour cell adhesion as well as subsequent death pathway events will dictate sensitivity to immune-mediated killing, and will be characterised in the presence of chemotherapy treatment. Granzyme uptake by tumour cells, proposed to be modulated by some drugs, will be explored for astrocytoma using confocal microscopy. Synergistic immune and chemotherapy approaches will ultimately be validated using stringent in vivo models. Poster Abstracts The Role of ICOS/ICOSL in anti-tumour immune response. Natalia Botelho, Rachel Perret, Alena Donda, Pedro Romero Ludwig Center for Cancer Research of the University of Lausanne ICOS is a costimulatory molecule expressed on T cells upon cell activation. Its ligand, ICOSL, belongs to the B7 family and is constitutively expressed on APCs as well as nonhematopoietic tissues, such as endothelial cells, brain, liver, lung and others. ICOS ligation by ICOSL provides costimulatory signals that promote T cell proliferation, differentiation and increase in cytokines secretion. There is conflicting evidence in the literature as to whether ICOS/ICOSL ligation in a cancer setting is beneficial or detrimental to antitumour immune responses. Some murine studies show that ICOSL- tumours get less rejected by the immune system than ICOSL+ tumours, while others have shown that ICOSL+ melanoma promote Treg survival and expansion, thus dampening antitumour immunity. We hypothesize that the functional outcome of ICOS ligation on T cells is context dependent. The stimulatory or regulatory outcome of the ICOS-ICOSL connection would depend on whether the ICOSL signal is provided by immature DCs (or nonprofessional APCs), leading to a regulatory outcome, or by mature DCs, or in the presence of a strong inflammatory environment, leading to a Th1/Tc1 polarity. The Sanroque mouse line is a strain that carries a mutation in the roquin gene, which leads to overexpression of ICOS, even in naïve T cells. Unexpectedly, upon human gp100 lentivirus immunisation, specific CD8+ T cell expansion in the circulating lymphocyte compartment was markedly reduced in Sanroque mice as compared to that measured in the wild type mice. We do not know whether this is due to the ICOS overexpression in T cells or to other cell extrinsic mechanism in these mice. A possible mechanism could result from ICOS induced downregulation of ICOSL in dendritic cells that would lead to a reduced ability to prime a CD8 T cell response. Alternatively, the role of ICOS overexpressing Tregs in Sanroque mice will be evaluated, as they might limit T cell priming. Wolfsberg Meeting 2012 Manuel Coutaz1, Floriane Auderset1, Stéphanie Corgnac2, Pedro Romero2, Fabienne Tacchini-Cottier 1 1 Department of Biochemistry, WHO Immunology Research and Training Center, University of Lausanne, Epalinges, Switzerland 2 Ludwig Institute for Cancer Research Ltd, Lausanne Branch, University of Lausanne, Epalinges, Switzerland. TH17 cells have been described as an effector CD4+ T helper subset on the basis of their secretion of specific cytokines (IL-17A, IL-17F) and their dependence of specific transcription factors (RORγt, RORα). Notch signalling is a highly evolutionary conserved pathway regulating cell lineage and cell differentiation fates in many organs and tissues. Notch receptors regulate cell differentiation during development but their role in the differentiation of CD4+ T helper subsets remains unclear. Of the four Notch receptors, only Notch1 (N1) and Notch2 (N2) are expressed on activated CD4+ T cells. In contrast to its documented role in TH1 and TH2 cell differentiation, only a few studies have demonstrated that Notch signalling might be involved TH17 cell differentiation. To investigate this, initial experiments were performed in attempt to define the role of Notch in TH17 differentiation in vitro. Naive CD4 T cells isolated from mice carrying a T cell-specific gene ablation of Notch receptors (N1N2ΔCD4Cre) were differentiated in TH17 condition. The absence of Notch receptors specifically decreased IL-17A secretion and mRNA expression but not the mRNA expression of RORγt. We then investigate the role of Notch signalling in Th17 differentiation in vivo, using the experimental subcutaneous melanoma. An increased percentage of IL-17A+ in CD4+ T cells was measured in the TDLNs in N1N2ΔCD4Cre mice. These results suggest that Notch signalling pathway may have an impact on IL-17A cytokine release correlating with delayed tumor growth observed in B16 N1N2ΔCD4Cre mice. Marina Fertig, Carole Bourquin Chair of Pharmacology, Department of Medicine, Faculty of Science, University of Fribourg Immunotherapy of gastric cancer: enhancing T cell recruitment into tumors Gastric cancer is one of the most common types of cancer in the world. So far, there is no effective therapeutic treatment for stomach cancer which is characterized by its high mortality rate. One promising approach to treat gastric tumors is immunotherapy. Previous studies have shown that intratumoral T cell infiltration positively correlates with the survival of patients with gastric cancer. Thus, T cell recruitment into the tumor is one of the important goals of cancer immunotherapy. However, the immunosuppressive tumor microenvironment can impair the efficiency of T cell infiltration. The gastric tumor microenvironment and its role in affecting lymphocyte infiltration are the main interests of this project. To examine the efficacy of the immunotherapy, we are using the CEA424-SV40 T antigen mouse model. These mice develop spontaneous gastric tumors in the pyloric region of the stomach at an early age and this model reflects the development of gastric cancer in humans. Previous in vivo findings in our lab revealed that autochthonous gastric tumors do not respond to T cell-based therapy, in contrast to subcutaneous tumors that are induced by injection of a stomach tumor-derived cell line. In the present project, we are comparing both autochthonous and subcutaneous tumors in the same mice in order to investigate the factors that control the T cell recruitment. The results of this study may provide an important contribution to the development of additional therapeutic strategies to target gastric tumors. Strength and limitations of regulatory T cells for immunotherapy in transplantation Lerisa Govender1 and Dr Dela Golshayan (MD PhD) 2 1 University of Lausanne, PhD Cancer and Immunology Program, Transplantation Immunopathology Laboratory,CHUV 2 Dpt of Medicine, Nephrology and Transplantation Centre, Transplantation Immunopathology Laboratory, CHUV In many experimental models, CD4+CD25+Foxp3+ regulatory T cells (nTreg) have been identified as key players in promoting peripheral transplantation (Tx) tolerance. We are focusing on therapies based on antigen-specific nTreg that control effector T cells (Teff) and prevent allograft rejection. The use of nTreg in immunotherapeutic protocols for solid organ Tx is however limited by their overall low numbers (2-10% of CD4+ T cells). In this study, we aimed to expand Treg directly in vivo and determine their suppressive function, efficacy and stability in promoting donor-specific tolerance in a stringent murine Tx model. Our data suggest that IL-2-based therapies lead to a significant increase of Treg in vivo. The expanded Treg suppressed Teff proliferation and allowed prolonged graft survival of major MHC-mismatched skin grafts in wild-type non-lymphopenic recipients. The expanded Treg alone were however not sufficient to induce tolerance in stringent experimental conditions. Rapamycin reduced the frequency of Teff but did not impede expansion of Treg. Pro-inflammatory stimuli hindered the expansion of Treg and resulted in an increase in the frequency of CD4+IFN-γ+ and CD4+IL17+ T cells. We propose that IL-2-based treatments would be an efficient method for expanding functional Treg in vivo, thereby favorably shifting the pool of alloreactive T cells towards regulation in response to an allograft. However, we also highlight some potential limitations of Treg expansion such as concomitant inflammatory events. TITLE: Infectious metastasis of mucocutaneous leishmaniasis: An IL-17-mediated response to leishmania RNA virus PRESENTED BY: Mary-Anne Hartley AFFLIATION: University of Lausanne, Department of Biochemistry, Laboratory of Professor Nicolas Fasel (Co-supervision: Dr. Catherine Ronet) Leishmania guyanensis (L.g) parasites have been plaguing humankind for centuries, provoking a deleterious inflammatory response, destroying host tissue and forming the ulcerating lesions, which typify most forms of the disease. About 15% of patients infected with a certain subtype of these parasites develop secondary lesions in the mouth and nose, where parasites metastasise to mucocutaneous tissues. We have recently linked disease severity in these infections to a virus infecting this parasite subtype: Leishmania RNA virus (LRV). Using L.g. clones, which are either naturally infected by LRV (V+) or depleted in it (V-), we set out to characterise the immune phenotype in a murine model of infection. Here, we found that V+ parasites potently induced the production of IL-17A as compared to its V- equivalent, thus insinuating that this proinflammatory cytokine plays a destructive role in the devolution and severity of metastatic leishmaniasis. Indeed, IL-17A-/- mice infected with V+ parasites had a significantly reduced disease severity compared to wild type and in vitro studies revealed that IL-17A directly increased the parasitism of macrophages while simultaneously decreasing their ability to clear the infection. Although IL17 has a promiscuous role in immunity, stimulating both the innate and adaptive immune systems (as well as a plethora of non-immune cells), a major shared goal of the IL-17 pathways is its self-propagation, where downstream effects converge to create a hyper-inflammatory feedback loop. This study will hopefully open a door to the development of a simple immunotherapeutic solution to a devastating and neglected disease. Intrinsic and extrinsic change in HSC behaviour upon aging Larisa Kovtonyuk, Ramin Radpour, Markus G. Manz and Hitoshi Takizawa Division of Hematology, University Hospital Zürich, Rämistrasse 100, CH-8091 Zürich, Larisa.kovtonyuk@usz.ch Hematopoiesis is an active ongoing process in bone marrow (BM) in which life-­ long self-­­ renewing hematopoietic stem cells (HSCs) replenish short-­ lived mature blood cells each day. Aging has an impact on homeostasis and regeneration of many tissues and their respective stem cells. Upon aging, HSCs also change their function including BM­ homing ability, differentiation and self renewal capacity. Our group demonstrated with in vivo single HSC divisional tracking that at any given time HSC consists of actively cycling and dormant pools with fluctuating hematopoietic contribution. HSCs with high proliferative history during physiological aging or serial transplantation tend to be dormant in a permissive environment. We hypothesized that cell-­ intrinsic drive toward dormancy is imprinted on HSCs through extensive divisions. The project aims to answer the questions on how the extrinsic and intrinsic factors determine HSC behaviour upon hematopoietic stress and aging, and what their relative contribution and molecular signature are on HSC cell fate decision. Preliminary data have demonstrated that aged HSCs increased dormancy when transplanted into non-­ irradiated young animals, suggesting cell-­ intrinsic regulation of dormancy. Fast-­ cycling aged HSCs showed balanced lineage repopulation as do both fast-­­ cycling and dormant young HSCs, while aged HSCs contributed to more myelopoiesis than lymphopoiesis, indicating the link between HSC lineage choice and cell cycle activity. Similar biology as in aged HSCs was observed in HSCs with extensive divisions during serial transplantation. It was shown, that both young and aged HSCs proliferated faster in an aged than in a young environment. In addition, hematopoietic stress as bleeding and infection, drives dormant HSCs into division, suggesting the extrinsic drive of HSC division. To dissect aging-­ associated intrinsic and extrinsic factors, we performed antibody based protein arrays with young versus aged BM cell lysates followed by bioinformatical analysis. The result revealed that expression levels of some secreted factors and cell surface receptors were altered in aged BM. Our findings suggest that extensive proliferative history imprints on HSCs intrinsic programs for dormancy and myeloid-­­biased differentiation, and that the dormant HSC can be activated possibly via age-­­and hematopoietic stress-­ related protein(s). Understanding of the molecular signature of intrinsic and extrinsic factors will provide new knowledge of regulatory mechanisms of stem cell-­­sustained organ systems, and open new avenues to control normal HSC cell cycle and function and to potentially allow specific eradication of pre-­­malignant stem cells in aged individuals. Title: Triaka, a novel mouse model for intestinal disease. Authors: Lukas Mager1, Philippe Krebs1 1 Division of Experimental Pathology, Institute of Pathology, University of Bern, Bern, Switzerland Abstract: Inflammatory Bowel Disease (IBD) is of multifactorial origin, encompassing genetic predispositions, intestinal microbiota and immunopathologic responses to them. The intestinal epithelial barrier plays a vital role in gut homeostasis and its integrity is altered in intestinal disorders such as IBD and colorectal cancer (CRC). Gut epithelial cells express genes like occludin and claudin but also immuno-modulatory molecules, among them IL-10, TGF-b and IL-8, depicting their importance for controlling the intestinal flora via interaction with the immune system. Here, we investigated a novel mouse model called Triaka that is characterized by a mutation in a conserved region of the Epithelial Splicing Regulator Protein 1 (Esrp1) gene. Esrp1 is highly expressed in the gut epithelium and Esrp1Triaka mice were found to be more susceptible to the dextran sodium sulfate (DSS) model of acute colitis. Histological sections of colon of DSS treated Esrp1Triaka mice showed massive loss of intestinal epithelial architecture, exacerbated inflammation, leucocyte infiltration, which are all features that can be found in IBD patients. Therefore, we hypothesize that the mutation leads to an alteration in the alternative splicing pattern of Esrp1 target genes, such as CD44, FGFR2, that are critical for the homeostatic properties of the gut epithelium. We are currently in the process of identifying these target genes that may become prognostic markers of IBD susceptibility. Also, since there is an undeniable relationship between IBD and CRC, the role of the Triaka mutation for carcinogenesis will be studied in a model of CRC. Clinical and histological features of flaky skin (fsn) mice Ursina Nüesch, Reinhard Seger, Jana Pachlopnik Schmid Division of Immunology/Haematology/Bone marrow transplantation, University Children’s Hospital Zürich, Steinwiesstrasse 75, 8032 Zürich Mice homozygous for the flaky skin (fsn) mutation in the gene encoding tetratricopeptide repeat protein 7 (TTC7) show a complex phenotype including inflammation and autoimmunity. The aim of our study was to perform a clinical and histopathological examination of the fsn/fsn mice mandatory for our future immunological investigations. We observed decreased body weight in fsn/fsn mice compared to their littermates. At the age of 8 weeks, spleens, axillary lymph nodes and livers were significantly increased in size in the fsn/fsn mice, while the thymus was smaller in fsn/fsn mice compared to the one in littermates. Histological analysis showed a disturbed splenic and lymph node architecture. At the age of 8 weeks, we could not find any deposits or inflammatory infiltrates in the kidneys of fsn/fsn mice. However, in 6 of 9 fsn/fsn mice we observed diarrhea. Inflammatory infiltrates and apoptotic cells were detected in the intestine. Our observations of lymphadenopathy and splenomegaly are consistent with previous publications. In contrast, we did not see any histopathological changes in the kidneys but this might be due to the younger age at examination in our study (8 vs 12 weeks). Interestingly, we observe an intestinal pathology that has not been described before. In order to better characterize the infiltrating cell subpopulations, we intend to perform flow cytometric analyses and immunohistochemistry. key words: flaky skin mouse, TTC7, inflammation 1 Kathrin Nussbaum Group of Prof. Dr. Burkhard Becher University of Zurich Institute for Experimental Immunology Winterthurerstrasse 190 CH-8057 Zürich Abstract Molecular mechanisms of innate lymphoid cells on IL12-mediated tumor suppression in the skin Malignant melanoma is a particularly aggressive tumor due to its metastasizing propensity and is the cause of most skin cancer-related deaths. Interleukin-12 (IL12) has consistently been demonstrated to possess potent tumor suppressing activity in murine melanoma models, however systemic administration in melanoma patients led to limited responses and severe adverse effects. Our lab showed that local instead of systemic IL12 delivery likewise results in strong anti-tumor suppressive activity mediated by innate lymphoid cells (ILCs). Although the molecular underpinnings of this phenomenon remain elusive we hypothesize that IL12 initiates tumor suppression through ILCs by changing the tumor microenvironment to be more conducive to subsequent immune invasion. Our overall goal is to determine how the immune system can be utilized to recognize and eliminate cancer cells. Therefore, we specifically aims to (1) systematically elucidate the impact of local IL12 treatment and combinatorial therapies using co-inhibiting immunosuppressive molecules in prophylactic and therapeutic settings in distinct murine melanoma models and (2) to determine the molecular mechanism underlying the IL12 mediated tumor suppression by identifying and characterizing molecular targets of ILCs. A better understanding of the role of IL12 and ILC function in tumors suppression and may lead to new immunotherapeutic applications taking advantages of the specific ability of cytokine-mediated changes to the tumor microenvironment. 1 PD-1 and SHP-1 expression finely tunes TCR-affinity-mediated cell activation and function in human tumor reactive T lymphocytes Danilo Presotto1, Michael Hebeisen1, Lukas Baitsch2, Petra Baumgaertner2, Olivier Michielin2,3, Daniel E. Speiser2 and Nathalie Rufer1,2 1 Department of Research, Lausanne University Hospital Center and University of Lausanne, Lausanne, Switzerland 2 Ludwig Center for Cancer Research, University of Lausanne, Lausanne, Switzerland 3 Multidisciplinary Oncology Center (CePO), University Hospital Center, Lausanne, Switzerland Adaptive immune responses rely on TCR-dependent recognition of antigenic peptides displayed by self-MHC molecules, and the strength of this interaction is a major correlate of disease protection. Because of central tolerance, T cells bearing TCRs against (self)-tumor antigens have lower affinity/avidity than pathogen-specific ones and elicite weaker immune responses. One strategy to overcome this limitation is to increase TCR affinity for tumor Ags. We generated a panel of optimized TCR variants having gradual increases in affinity for the melanoma cancer-germline Ag NY-ESO-1. We found that optimal mRNA expression and T cell function were achieved for CD8+ T cells expressing TCR with upper physiological affinities (KD 1-5 mM). Unexpectedly, a drastic functional decline occurred in T cells with TCRs of supraphysiological affinities (< 1 mM). Biochemical characterization of the TCR activation pathway in these cells showed reduced phosphorylation levels of both ZAP-70 and ERK 1/2 compared to the WT, with a concurrent increase in the expression levels of PD-1 and its associated phosphatase SHP-1. Importantly, PD-1 and SHP-1 blockade could partially restore T cell function in T cells of highest affinity. Collectively, these results indicate that SHP-1 and PD-1 act very proximally in the TCR pathway and could serve as negative regulators to control cell activation, signaling and function in T cells bearing self TCRs of supra-physiological affinities. Such studies are essential to identify putative targets to increase T cell function, allowing further promoting of therapeutic immune interventions like adoptive T cell therapy. XXIV Meeting of the Swiss Immunology PhD students Wolfsberg, 2- 4 April 2012 _ Abstract CD27-CD70 costimulatory axis Mohamad Al Sayeda and Adrian Ochsenbeina, b a. University of Bern; Department of Clinical Research, Tumor immunology Mohamad.Alsayed@dkf.unibe.ch b. University Hospital Bern; Institute for Medical Oncology Adrian.Ochsenbein@insel.ch T cell activation and survival are critical processes for an efficient immune response against various infections and cancer. Besides T cell receptor stimulation, another signal through a co-stimulatory molecule is required for efficient T cell activation. The co-stimulatory molecule CD27, a member of the TNF receptor superfamily, is expressed on subsets of T, B and NK cells. CD27 promotes expansion, survival and memory formation of T cells. To date, two isoforms of CD27 have been identified, but the differences in expression and function of these two isoforms in different immune cells have not been addressed. In this study, we identified a third isoform of CD27 which we characterized on the RNA level. We also studied the expression of the three isoforms in different immune cell populations. Further characterization of CD27 isoforms, at the protein level, is required. On the other hand, CD70, the unique ligand of CD27, which is expressed on activated T cells and antigen-presenting cells, has been shown to have a reverse signaling activity on B cells. Our preliminary results shows a possible CD70 reverse signalling activity on T cells. Taken all together, CD27-CD70 co-stimulatory axis seems to have both negative and positive regulatory role during immune response, and more investigation about CD27 isoforms and their ligand may reveal the underlying mechanisms of such regulatory effects. 1 Controlling the recruitment of myeloid-derived suppressor cells into the tumor microenvironment. Thibaud Spinetti, Carole Bourquin Chair of Pharmacology, Department of Medicine, University of Fribourg, Switzerland Myeloid-derived suppressor cells (MDSC) are a heterogeneous population of immature myeloid cells with the capacity to inhibit immunological responses. During cancer progression, MDSC are recruited to the tumor sites and secondary lymphoid organs. This recruitment leads to the suppression of the anti-tumor function of T and natural killer cells. Factors released by tumor cells or their microenvironment such as GM-CSF or IL1b lead to the migration of MDSC into the tumor. Previous studies in the Bourquin group show that treatment by different TLR ligands in tumor-bearing mice leads to a change in the phenotype and their capacity to suppress the T cell response. The aim of this project is to identify effects of these different TLR ligands on the migration patterns of MDSC. Investigation into the regulation of chemokines and their receptors as well as homing molecules such as selectins or integrins after TLR challenge are ongoing. For a global understanding of the mechanisms involved, in vitro and in vivo experiments are planned and analysis by different methods like flow cytometry or RT-PCR are used to identify the modulation of protein expression. In conclusion, this project should give new information about the impact of innate immune stimulation on MDSC migration in tumor-bearing mice. Considering the suppressive activity of MDSC in anti-tumor immunity, identification of new strategies to control trafficking of these cells may improve immune responses against cancer. XXIV Meeting of the Swiss Immunology PhD students Wolfsberg, 2- 4 April 2012 Immune Control of MCMV in the Salivary Gland Jenny Thom, Annette Oxenius ETH Zürich - Institute of Microbiology Wolfgang-Pauli-Strasse 10 CH-8093 Zürich Jenny.thom@micro.biol.ethz.ch Cytomegaloviruses (CMVs) are ubiquitous β-herpesviruses that establish lifelong infection and latency. Prolonged secretion of virus in saliva represents a major route of CMV transmission with acinar glandular epithelial cells propagating virus for months after virus replication is controlled in all other organs. While CD8 T cells are sufficient to control viral replication in visceral organs, they fail to do so in the salivary glands where virus is eventually controlled by IFNg producing CD4 T cells. Using murine CMV (MCMV) our group showed that despite MCMV-specific CD8 T cells being recruited to the salivary glands, their activation is prevented as MCMV encoded immune evasion genes impair MHC I expression on infected acinar glandular epithelial cells. Moreover, our data indicate that CD4 T cells in the salivary glands are activated by non-infected antigen presenting cells (APCs) which present internalized antigen via MHC II but are unable to cross-present on MHC I, thus failing to activate CD8 T cells. Our aim is to further analyze the mechanisms leading to CD4 T cell mediated control of lytic viral replication in the salivary glands. To this end, we will characterize salivary gland resident APCs and compare their phenotypes and functions with APCs derived from lymphoid tissue and from other secretory organs. Furthermore, we aim to visualize MCMV spread, T cell migration, antigen recognition, and finally control of lytic viral replication via intravital microscopy of the salivary glands to understand the dynamic processes involved in creating the unique resistance towards CD8 T cell mediated viral clearance. Flt3-mediated regulation of dendritic cell homeostasis Chakradhar Yakkala, Dior Kingston, Hitoshi Takizawa, Yasuyuki Saito, Tim Sparwasser, Markus G Manz Division of Hematology, University Hospital Zurich, Rämistrasse 100, CH-8091 Zurich Chakradhar.Yakkala@usz.ch Dendritic cells (DC) are the antigen presenting cells that act as pedagogues for the generation of an immune response. Because of their short life span, they need to be continuously replenished by the upstream hematopoietic stem and progenitor cells. FMS like tyrosine kinase 3 (Flt3) is a class III membrane bound tyrosine kinase receptor that is expressed on early hematopoietic progenitors, DC precursors and DCs, and has been shown to instruct the differentiation of hematopoietic progenitors towards DC lineage. However, the regeneration kinetics of DC lineage cells via their respective progenitors and functional dynamics of DC precursors in steady state and inflammation remain unclear. To address these questions, our group has generated a reporter-depleter mouse model, in which human diphtheria toxin receptor (DTR) fused with green fluorescent protein (GFP) is expressed under the control of murine Flt3 promoter and thus, allows to transiently deplete Flt3 expressing cells upon diphtheria toxin (DT) administration. The flow cytometric analysis showed that GFP signal is well correlated with Flt3 expression in various hematopoietic compartments, ranging from early progenitors to mature cells in non-hematopoietic and hematopoietic tissues. The DT dose was successfully titrated to ensure a complete depletion of GFP+ cells and no obvious toxicity in vivo. Given the importance of DC in the context of infection, cancer and autoimmunity, acquiring the knowledge of DC homeostasis under physiological and inflammatory conditions will be highly valuable for the development of new drug and design of therapeutic strategy. Deciphering Allopurinol Hypersensitivity – Characterisation of risk factors for allopurinol hypersensitivity in vitro. Authors: JamesYun, Daniel Yerly, Werner Pichler Affiliation: Division of Allergology, Clinic for Rheumatology and Clinical Immunology/Allergy, University of Bern, Inselspital, Bern, Switzerland Background: Allopurinol hypersensitivity is a major cause of severe cutaneous adverse reactions (SCAR) and it is strongly association with HLA-B*58:01 allele. We took advantage of this association to investigate the immune mechanism of allopurinol-induced SCAR. Method: Allopurinol-specific T cell lines (TCL) were generated from PBMC isolated from allopurinol allergic individuals, allopurinol-naïve HLA-B*58:01 positive donors and allopurinol-naïve HLA-B*58:01 negative donors. Autologous Epstein-Barr virustransformed lymphoblastoid B cell lines (EBV-BLCL) were used in some experiments as antigen presenting cells (APC). The specificity of TCL was assessed by FACS analysis. Results: In allopurinol allergic patients (2), allopurinol-specific TCL could be generated within 4 weeks but this was not possible in all allopurinol-naïve individuals (8). However, in allopurinol-naïve HLA-B*58:01 positive individuals (5), allopurinol-specific TCL could be generated after 4 weeks with multiple restimulations. In allopurinol-naïve HLAB*58:01 negative individuals (3), allopurinol-specific TCL could not be generated at all. However, when EBV-BLCL were used as APC for TCL induction in these HLAB*58:01 negative individuals (3), allopurinol-specific TCL could also be generated. There were no obvious phenotypic differences between these groups. Conclusion: Our in vitro data suggest that HLA-B*58:01 molecule is likely involved in the pathogenesis of allopurinol hypersensitivity as it facilitates reactivity to allopurinol in allopurinol naïve individuals. However, this allele is not an absolute requirement for the development of allopurinol hypersensitivity as the in-vitro induction occurred when EBV-BLCL were used as APC. This implies that in the presence of a co-factor, such as viral infection, the threshold for the development of allopurinol hypersensitivity may be lowered.