Using a Beaker, Graduated Cylinder, and Digital Balance
advertisement

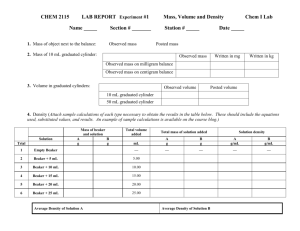
Name: ______________________________ Date: _______ Period: _____ Using a Beaker, Graduated Cylinder, and Digital Balance Background: All matter has mass and volume. Mass is a measure of the amount of matter an object has. Its measure is usually given in grams (g) or kilograms (kg). Volume is the amount of space an object occupies. There are numerous units for volume including liters (l), meters cubed (m3), and gallons (gal). Mass and volume are physical properties of matter and may vary with different objects. For example, it is possible for two pieces of metal to be made out of the same material yet for one piece to be bigger than the other. If the first piece of metal is twice as large as the second, then you would expect that this piece is also twice as heavy (or have twice the mass) as the first. If both pieces of metal are made of the same material the ratio of the mass and volume will be the same. We can use both volume and mass to calculate the density of object, or use density as a conversion facture and either volume to calculate either mass or volume. Using the following equation: D = M/V Which can be changed to either M = D * V In this lab you will use a graduate cylinder and beaker to measure 50mL 5 times. Each time you will record how much water you have added, and record the mass of the water. Once you complete all of the measurements you will use the density of water to determine the actual mass of water in the beaker. You will then compare you measured and calculated results to determine which is more accurate, the beaker or the graduated cylinder, and determine how precise you were in the lab. Materials: ∙ 1 50mL Graduated cylinder ∙ 1 50mL Beaker ∙ 1 400mL Beaker ∙ Water ∙ Balance Procedure: 1. Put your 400mL beaker onto one of the balances and record the mass of the empty beaker; make sure to note which beaker you used, you will need to use the same balance again. 2. Measure 50mL of H2O from the faucet in your empty 50mL graduated cylinder and poor in into the empty beaker. 3. Record you volume in your data chart, and record the mass using the same balance as previous. 4. Repeat 4 more times. 5. After completing the task poor the H2O down the drain and dry the beaker. 6. You will then complete the same task using a 50mL beaker. 7. Once you are finished clean and dry your glassware and table, put your stools on top of you bench and work on your lab report. Data: Table 1: Experiment 1 Beaker Measured Volume Measured Mass Calculated Mass Measured Mass Calculated Mass 1 2 3 4 5 Total Mass of Empty Beaker Table 2: Experiment 2 Graduated Cylinder Measured Volume 1 2 3 4 5 Total Mass of Empty Beaker Questions: 1. Why do we measure liquids using the meniscus? 2. How does accuracy and precision differ? 3. Is the graduated cylinder or beaker more accurate? How do you know? Why? 4. Where you precise? How do you know? 5. If you were not precise, what could you do differently next time?