TOPIC: 2
advertisement

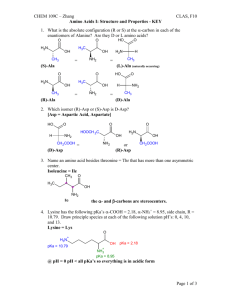
TOPIC: 2. Acids / Bases SUBTOPIC: 21. Structure and Acidity 1. Which acid has the smallest pKa value? A. HC≡CH B. NH3 C. H2O D. CH2=CH2 ____________________________________________________________________________________________________________ ________________________________________________________________________ 2. Rank the following compounds in order of acidity (1 = most acidic). CH3CH2OH CH3CH2NH2 CH3C CH CH3CH=CH2 ________________________________________________________________________ ________________________________________________________________________ 3. Which is the most basic compound? Which is the least basic? A. NaOH B. NaOOCCH3 C. NaOCH3 D. NaNH2 ________________________________________________________________________ ________________________________________________________________________ 4. Which is the most acidic compound? Which is the least acidic? O A. O O O O OEt OEt EtO O B. C. D. O ________________________________________________________________________ ________________________________________________________________________ 5. Guanidine (structure shown below) is one of the stronger neutral nitrogen bases. Its protonated form has a pKa of 13.6. (a) Write the structure of a neutral carbon acid that guanidine would deprotonate. NH H2N NH2 (b) Two different products of protonation are possible for guanidine. Write each and using resonance structures, show which protonation product is more stable. ________________________________________________________________________ ________________________________________________________________________ 6. Which is the most basic aniline and which is the least basic aniline? NH2 NH2 A. NH2 NH2 C. B. NO2 D. Cl OCH3 ________________________________________________________________________ ________________________________________________________________________ 7. Which is the most acidic and which is the least acidic phenol? OH OH A. NO2 B. C. CH2CH3 OH OH D. NO2 NHCH3 ________________________________________________________________________ ________________________________________________________________________ 8. Which is the most basic compound and which is the least basic compound? For extra credit, write the approximate pKa of the conjugate acid under each base. CH3Li LiOH LiOOCH LiNHCH3 ____________________________________________________________________________________________________________ ________________________________________________________________________ 9. Which is the most acidic compound and which is the least acidic compound? For extra credit, write the approximate pKa under each acid. OH O OH CH2OH NH3+ ________________________________________________________________________ ________________________________________________________________________ 10. Which is the strongest carbon acid and which is the weakest carbon acid? For extra credit, write the approximate pKa under each acid. O O O O O O O O ________________________________________________________________________ ________________________________________________________________________ 11. Rank in order of basicity ( 1 = most basic ). O O NH NH NH NH HN NH O ________________________________________________________________________ ________________________________________________________________________ 12. Which is the weakest acid? O A. HBr B. CH3CH2OH C. O N + OH O D. CH3 C OH ________________________________________________________________________ ________________________________________________________________________ 13. Which is the strongest base that can exist in liquid NH3? A. ─OH B. ─NH2 C. ─CH3 D. NH3 ____________________________________________________________________________________________________________ ________________________________________________________________________ 14. Which is the strongest base? A. CH3CH2─ B. NH2─ C. CH3O─ D. HC≡C─ ________________________________________________________________________ ________________________________________________________________________ 15. Which amine would you expect to be more basic aniline or cyclohexylamine? Briefly explain why? NH2 NH2 ________________________________________________________________________ ________________________________________________________________________ 16. Which is more acidic phenol or p-nitrophenol? Explain using resonance structures. OH OH NO2 ________________________________________________________________________ ________________________________________________________________________ 17. Which is the strongest acid? O A. HC CH C. H2O B. NH3 D. CH3 C OH ________________________________________________________________________ ________________________________________________________________________ 18. Which is the strongest base? A. NaOH B. KSH C. CH3Li D. NaNH2 ____________________________________________________________________________________________________________ ________________________________________________________________________ 19. Which is the strongest acid? A. CH3CH2OCH2CH3 B. CH3CH2OH C. CH3CF2OH D. CH2CH2NH2 ________________________________________________________________________ ________________________________________________________________________ 20. Which compound is not a Lewis acid? A. BH3 B. CH3SSCH3 C. + C(CH3)3 D. AlCl3 ________________________________________________________________________ ________________________________________________________________________ 21. Which is the strongest base? A. K O CH3 B. Li N C. CH3CH2MgBr D. NaOH CH3 CH3 ________________________________________________________________________ ________________________________________________________________________ 22. Which is the strongest carbon acid and which is the weakest carbon acid? O O O A. O O C. B. EtO O O O D. O OEt ________________________________________________________________________ ________________________________________________________________________ 23. Which is the most basic compound.and which is the least basic compound? O NH2 NH2 NH2 NH2 C. B. A. D. Br ________________________________________________________________________ ________________________________________________________________________ 24. Which is the most acidic phenol and which is the least acidic phenol? OH OH OH OH NO2 A. NO2 C. B. NO2 NO2 NO2 NO2 D. ________________________________________________________________________ ________________________________________________________________________ 25. Which is the strongest acid? O A. CH3COH O B. CH2COH NH3 OH NH3+ C. D. + ________________________________________________________________________ ________________________________________________________________________ 26. Which acid has the highest pKa? O OH C. CH3O Br O OH OH B. A. O O OH D. CH3 ________________________________________________________________________ ________________________________________________________________________ 27. Which acid has the lowest pKa? O OH A. D. O N 2 C. CH O 3 Br OH OH OH B. O O O ________________________________________________________________________ ________________________________________________________________________ 28. Draw the most important resonance structure of p-aminophenol that explains why paminophenol is a stronger base than aniline. ________________________________________________________________________ ________________________________________________________________________ 29. Which is the strongest base that can exist in acetic acid? O A. OH B. O C. H2O D. NH3 ________________________________________________________________________ ________________________________________________________________________ 30. Rank the following groups in accordance of the Cahn-Ingold-Prelog priority rules. O CH3C CH3CH2O HC C ________________________________________________________________________ ________________________________________________________________________ 31. Which is the strongest acid? A. CH3CH3 B. CH2=CH2 C. HC CH D. NH3 ________________________________________________________________________ ________________________________________________________________________ 32. Acetic acid (pKa = 4.8) and ethanol (pKa = 16) have very different acid strengths. The major species present when acetic acid and ethanol are added to water and the pH is adjusted to 7.0 are (identify two): A. CH3CO2H B. CH3CO2 C. CH3CH2OH D. CH3CH2O ________________________________________________________________________ ________________________________________________________________________ 33. Acetic acid is a stronger acid than ethanol. Explain using resonance structures. ________________________________________________________________________ ________________________________________________________________________ 34. Which is the strongest acid? A. CH3CH3 B. CH2=CH2 C. HC CH D. NH3 ________________________________________________________________________ ________________________________________________________________________