Basic Concepts in Fluidization and Industrial applications
advertisement

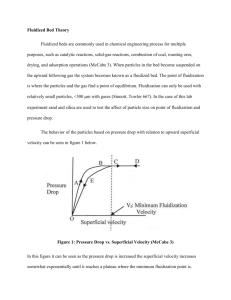
Basic Concepts in Fluidization Britt Halvorsen Telemark University College The phenomenon of fluidization Fluidization is a process whereby a granular material is converted from a static solid-like state to a dynamic fluid-like state. The process occurs when a fluid (liquid or gas) is passed up through the granular material (powder). 1 Fluidized beds In a fluidized bed gas is passing upwards through a bed of particles. The earliest applications of fluidization were for the purpose of carrying out chemical reactions. Since that time there have been a number of successful chemical processes involving fluidized bed reactors, but also other applications. Fluidized beds are applied in industry due to their large contact area between phases, which enhances chemical reactions, heat transfer and mass transfer. The efficiency of fluidized beds is highly dependent of flow behaviour Particle behaviour In a fluidized bed the frictional forces between particles are small Gas/particle assembly behaves like a liquid with a density equal to the bulk density. Behaviour of particles in fluidized beds depends on a combination of their mean particle size and density. 2 Various stages of fluidization Classification of particles Geldart fluidization diagram: Identify characteristics associated with fluidization of particular powders at ambient conditions. Geldart (1973) classification of particles according to their fluidization behaviour. 3 Group A powders Most commercial fluidized bed catalytic reactors use Geldart group A powders. Group A powders: Easily fluidized Bed expands considerably before bubbles appear due to: Inter-particle forces that are present in this group of powders Inter-particle forces are due to particle wetness, electrostatic charges and van der Waals forces. Bubble formation will occur when the gas velocity exceeds the minimum bubble velocity. The bubbles rice faster than the gas percolating through the emulsion. Maximum bubble size usually less than 10 cm and independent of the bed size. Kunii and Levenspiel (1991) Group B particles For group B particles Inter-particle forces are negligible Bubbles are formed as the gas velocity reaches the minimum fluidization velocity. Bed expansion is small compared to group A particles. Small bubbles are formed close to the air distributor and the bubble size increase with distance above the distributor. Bubble size also increases with the excess gas velocity Excess gas velocity: difference between the gas velocity and the minimum fluidization velocity, Geldart (1986). Coalescence is the dominating phenomena for group B powders and bubble size is roughly independent of mean particle size. Most bubbles rise faster than the interstitial gas velocity. 4 Group C and D powders Group C powders: Cohesive. Fluidization is extremely difficult Bubble formation will not occur. Geldart group D powders: Large and/or dense particle powders Undesirable for fluidization operations. Large gas flows are needed to get these particles fluidized Geldart C and D powders give a low degree of solid mixing and gas back-mixing. Phenomenon of fluidization Gas or liquid Fixed and expanded bed (low fluid velocity) Fluid percolates through the void between the particles. Expanded bed (higher fluid velocity) Particles move apart Bed at minimum fluidization Further increased velocity→particles are suspended by the upward-flowing fluid Frictional force between particle and fluid counter balances. Pressure drop equals weight of particles+fluid Minimum fluidization velocity 5 Liquid: Smoothly Velocity above minimum fluidization velocity Gas and liquid Lean fluidized bed phase fluidization High velocity, fine particles Particles carried out of the bed with the fluid Pneumatic transport Gas Bubbling fluidized bed Gas velocity increased above minimum fluidization velocity Bed height about minimum fluidization Bubbles growing with hight Slugging Deep bed with small diameter Bubble diameter about bed diameter Fine particles, axial sluggs Coarse particles, flat slugs 6 Gas Turbulent fluidization Very high gas velocity Terminal velocity of solids exceeded Upper surface of bed disappears Entrainment of particles significant Turbulent motion of solid clusters and voids of gas with various size and shape When will the bed start to fluidize? At the velocity where the buoyant forces equals the drag forces. (wall friction and solids stress are neglected) Minimum fluidization velocity developed from the : (1 − ε )(ρ g s − ρ g )⋅ g = Φ sg εg (u g − u s ) ε g = void (gas) fraction [ ρs , ρg = solid and gas density kg/m3 [ g = acceleration of gravity m/s 2 ] ] [ Φ sg = Gas − particle drag coefficient m/s 2 ] 7 Minimum fluidization velocity At minimum fluidization: Solid velocity is zero: U mf = ε mf 2 (1 − ε mf )(ρ s − ρ g )g Φ sg Calculated minimum fluidization velocity: Deviation from experimental; Void fraction Particle size distribution Mean particle diameter Drag models Different drag models are developed Ergun equation : (1 − ε ) µ 2 Φ sg = 150 g ε g (d s ψs ) g 2 + 1.75 ρ g v g − vs ε s d s ψs , for ε g ≤ 0.8 ε s = Solid volume fraction d s = Particle diameter ψ s = Form factor µ g = Gas viscosity v g , vs = Gas and solid velocity 8 Syamlal & O’Brien drag model Empirical model Φ sg = 3 1 C Dρ g 2 ε g (1 − ε g ) v g − v s 4 ds Rt ( R t = 0.5 A − 0.06Re s + 0.0036Re s2 + 0.12Re s (2B − A) + A 2 A=ε ) 4.14 g B = 0.8ε 1.28 g , for ε g < 0.85 B = ε 2.65 g , for ε g > 0.85 R t C D = 0.63 + 4.8 Re s ε g ⋅ ρ g ⋅ (v g − v s )⋅ d p Re s = µg Calculated and experimental minimum fluidization velocities Minimum fluidization velocity [m/s] 0.6 0.5 Ex. Small+mix small 0.4 Ex. Medium + mix medium Ex. Large Calc., void=0.37 0.3 Calc., void=0.38 Calc., void=0.40 0.2 Calc., void=0.42 Calc., void=0.44 0.1 0 0 200 400 600 800 1000 1200 Mean particle diameter [µ µ m] •Particle size distribution influences on: Minimum fluidization velocity, Bubble formation, Segregation, Pressure drop 9 Pressure drop a. Uniform distributed sand ( Figure from: Kuuni and Levenspiel, Fluidization Engineering) Wide distribution of particle sizes (180-1400 µm) ( Figure from: Kuuni and Levenspiel, Fluidization Engineering) Pressure drop (Experiments performed at TUC) 10 Efficiency dx Bubble size dy Bubble Distribution Efficiency Bubble velocity Bubble frequency V=dy/dt Particle segregation Experiment: Mixture of two powders. Particle range: 100-200 µm (153) and 750-1000 µm (960) Simulation: Two particle phases: Mean diam. 153 µm and 960 µm 11 Experimental set-up, 3-D bed 2.0 m 0.75 m 0.35 m 0.25 m Fibre optical probe Probe head Light reflection Signal [V] distance between lights: 2.7 mm 2 1.5 1 0.5 0 -0.5 -1 -1.5 -2 Signal 1 Signal 2 0 0.1 0.2 0.3 0.4 0.5 Time [s] 12 Labview program Pneumatic transfer Test facility [Akilli H et al., 2001] Lab scale set-up for experimental study of pneumatic transport. Characteristic for industrial transport systems: •Long pipes with rather small diameters. •Pipe walls influence significantly on the flow behaviour. •Elbows highly influence the flow behaviour 13 Conveying system Horizontal pipes Vertical pipes Bend Horizontal-vertical Vertical-horizontal Horizontal-horizontal U-bend Distance between bends Ratio diameter/length Different flow regimes can arise Flow regime can change during the transport Change in velocity direction influence the flow behaviour Particle collisions with walls Particle motion Influenced by: Gravitational settling in horizontal pipes Inertial behaviour in bends and branches Turbulent dispersion Lift forces, friction forces Particle-wall collisions Rough walls/smooth walls Particle-particle collisions (Huber and Sommerfeld, 1998) 14 Flow regimes Flow regimes in horizontal pipes, Crowe et al.(1997) Homogeneous flow: Dilute flow transport particles are well mixed homogenious state by turbulent mixing Dune flow: Particles start to settle Slug flow: Includes regions with settled particles – and regions with particles in suspension. Slug flow is used in dense flow transport. Higher pressure drop and solid loading. Low velocity less material degradation and line erosion. Packed bed: Lower velocity. No transport Flow regimes Pressure drop as a function of gas velocity, Kunii and Levenspiel (1991) Saltation velocity: Superficial gas velocity at which particles begin to separate from the gas phase and slide or roll along the bottom of the pipe. →Changes from suspended to non-suspended transport 15 Wall roughness Smooth pipes Particles settle Stronger coupling with gas phase Deformation of stream-wise velocity profile (ref) Rough walls Increase of rebound angle compared to compact angle Particles re-suspended Gravitational settling reduced Secondary flow Increasing pressure loss Huber and Sommerfeldt, 1998 Wall roughness Figure: Particle mass flux in smooth and rough pipe. (Huber and Sommerfeld, 1998) 16 Rope Formation and disintegration: •Centrifugal forces •Secondary flows •Gas velocity •Bend radius •Solid loading •Pipe orientation •Particle size Figure: Rope formation and dispersion [Akilli et al., 2001] •Particle density Mc Clusky et al. (1989) Levy and Mason (1998) Yilmaz and Levy (2001) Rope Centrifugal forces in the elbow cause gas and particles to segregate The rope formation and dispersion is dependent of solid particles impinging on the outer wall of the bend, forming relatively dense phase structure (rope). Centrifugal forces Secondary flows Gas velocity Bend radius Solid loading Pipe orientation Particle size Particle density The particles are decelerated in the bend due to particle-particle collisions and particle-wall collisions, and have to be re-accelerated downstream the bend to the conveying velocity. Mc Clusky et al. (1989), Levy and Mason (1998), Yilmaz and Levy (2001) 17 Pressure drop Pressure drop influenced by: Gas velocity Dictates flow regime Great effect on total pressure drop Increase in gas velocity strongly increases the total pressure drop Operation at low gas velocity Increasing slip velocity Increasing total pressure drop Particle size Particle concentration cause problems in suspending particles. Particle density cause high energy losses. Slight decrease in slip velocity Increase in total pressure drop Particle-wall collision Bend, radius ratio Pipe dimensions Hidayat and Rasmuson, (2005) Turbulence Particle influence Displacement of flow field Generation of wakes Dissipation to the motion of dispersed phase Modification of velocity gradients Additional length scales Particle-particle interaction (0.1) 18 Concluding remarks Fluidization behaviour depends on: Particle size and particle size distribution Particle density Fluid properties Efficiency of fluidsized bed depends on: Contact between particles and fluid Economy Bubble size Bubble frequency Bubble velocity Bubble distribution Low pressure drop Good mixing High degree of conversion Low degree of energy loss Challenges (A lot of research still remaining) Develop good models (Empirical) Physical CFD simulations Scaling Reduce simulation time 19