Final Exam key - Workforce3One
advertisement

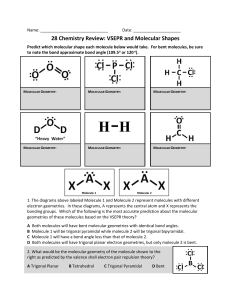
Final Exam 135 Points Name:____________________ Date: _______________ 1. (20 points) for the CO32- and ICl2+ ions give the following a. Lewis structure b. AXE notation c. Electronic geometry d. Molecular geometry e. Bond angles f. Sketch molecule in 3-D g. Indicate the hybridization h. Indicate if the molecule has polar bonds (if so how many?) i. Indicate whether the entire molecule is polar or non polar (57 Points, 3 points each) Multiple choice. Show your work for partial credit 2. The molecular geometry of the H3O+ ion is __________. A) linear B) tetrahedral C) bent D) trigonal pyramidal E) octahedral Answer: D 3. The molecular geometry of the CS2 molecule is __________. A) linear B) bent C) tetrahedral D) trigonal planar E) T-shaped Answer: A 4. The molecular geometry of the CHCl3 molecule is __________. A) bent B) trigonal planar C) trigonal pyramidal D) tetrahedral E) T-shaped Answer: D 5. The molecular geometry of the SF2 molecule is __________. A) linear B) bent C) trigonal planar D) tetrahedral E) octahedral Answer: B 6. The O–S–O bond angle in SO 2 is slightly less than __________. A) 90° B) 109.5° C) 120° D) 180° E) 60° Answer: C 7. The F–N–F bond angle in the NF3 molecule is slightly less than __________. A) 90° B) 109.5° C) 120° D) 180° E) 60° Answer: B 8. The hybridization of the carbon atom in carbon dioxide is __________. A) sp B) sp2 C) sp3 D) sp3d E) sp3d 2 Answer: A 9. There are __________ σ and __________ π bonds in the H–C≡C–H molecule. A) 3 and 2 B) 3 and 4 C) 4 and 3 D) 2 and 3 E) 5 and 0 Answer: A 10. There are __________ σ and __________ π bonds in the H2C=C=CH2 molecule. A) 4, 2 B) 6, 4 C) 2, 2 D) 2, 6 E) 6, 2 Answer: E 11. The total number of π bonds in the H–C≡C–C≡C–C≡N molecule is __________. A) 3 B) 4 C) 6 D) 9 E) 12 Answer: C 12. There is/are __________ σ bond(s) in the molecule below. A) 1 B) 2 C) 12 D) 13 E) 18 Answer: C 13. There is/are __________ π bond(s) in the molecule below. A) 0 B) 1 C) 2 D) 4 E) 16 Answer: C 14. The molecular geometry of the left-most carbon atom in the molecule below is __________. A) trigonal planar B) trigonal bipyramidal C) tetrahedral D) octahedral E) T-shaped Answer: C 15. The molecular geometry of the right-most carbon in the molecule below is __________. A) trigonal planar B) trigonal bipyramidal C) tetrahedral D) octahedral E) T-shaped Answer: A 16. The hybridization of the carbon atom labeled x in the molecule below is __________. A) sp B) sp2 C) sp3 D) sp3d E) sp3d 2 Answer: B 17. The Lewis structure of carbon dioxide is given below. The hybridization of the carbon atom in carbon dioxide is __________. A) sp3 B) sp2 C) sp D) sp2d E) sp2d 2 Answer: C 18. A 2.25-g sample of magnesium nitrate, Mg(NO3)2, contains __________ mol of this compound. A) 38.4 B) 65.8 C) 148.3 D) 0.0261 E) 0.0152 Answer: E 19. Of the following, which gives the correct order for atomic radius for Mg, Na, P, Si and Ar? A) Mg > Na > P > Si > Ar B) Ar > Si > P > Na > Mg C) Si > P > Ar > Na > Mg D) Na > Mg > Si > P > Ar E) Ar > P > Si > Mg > Na Answer: D 20. The list that correctly indicates the order of metallic character is __________. A) B > N > C B) F > Cl > S C) Si > P > S D) P > S > Se E) Na > K > Rb Answer: C 21. (16 points) A Lewis structure for SO2 is shown. O S O Based on the Lewis structure shown and the VSEPR model, answer the following questions. A. (4 pts) What is the electronic geometry? a. linear pyramidal b. bent c. trigonal planar d. trigonal c. trigonal planar d. trigonal B. (4 pts) What is the molecular geometry? a. linear pyramidal b. bent C. (4 pts) What is the approximate O–S–O bond angle? a. 90˚ b. 109.5˚ c. 120˚ d. 180˚ D. (4 pts) Given that the S-O bond is polar, which of the following describes the SO2 molecule? a. polar b. non-polar F P F 22. (16 points) A Lewis structure for PF3 is shown. F Based on the Lewis structure shown and the VSEPR model, answer the following questions. A. (4 pts) What is the electronic geometry? a. tetrahedral pyramidal b. square planar c. trigonal planar d. trigonal c. trigonal planar d. trigonal B. (4 pts) What is the molecular geometry? a. tetrahedral pyramidal b. square planar C. (4 pts) What is the approximate F–P–F bond angle? a. 90˚ b. 109.5˚ c. 120˚ d. 180˚ D. (4 pts) Given that the P–F bond is polar, which of the following describes the PF3 molecule? a. polar b. non-polar 23. (10 points) Lithium and nitrogen react in a combination reaction to produce lithium nitride: 6Li (s) + N2 (g) 2Li3 N (s) In a particular experiment, 3.50-g samples of each reagent are reacted. What is the theoretical yield of lithium nitride in g? 5.85 24. (8 points) What is the empirical formula of a compound that contains 29% Na, 41% S, and 30% O by mass? Na 2S2O3 25. (5 points) How many grams of H3 PO4 are in 175 mL of a 3.5 M solution of H3 PO4 ? 60 26. 3 bonus points Pe rio d ic Ta b le of t he Ele m e nt s 1A 1 8A 18 3A 13 4A 14 5A 15 6A 16 7A 17 2 He 4 .0 0 5 B 1 0 .8 6 C 1 2 .0 7 N 1 4 .0 8 O 1 6 .0 9 F 1 9 .0 14 Si 2 8 .1 15 P 3 1 .0 16 S 3 2 .1 17 Cl 3 5 .5 30 Zn 6 5 .4 13 Al 2 7 .0 31 Ga 6 9 .7 10 Ne 2 0 .2 18 Ar 3 9 .9 32 Ge 7 2 .6 33 As 7 4 .9 34 Se 7 9 .0 35 Br 7 9 .9 36 Kr 8 3 .8 47 Ag 108 48 Cd 112 49 In 115 50 Sn 119 51 Sb 122 52 Te 128 53 I 127 54 Xe 131 79 Au 197 80 Hg 201 81 Tl 204 82 Pb 207 83 Bi 209 1 H 1 .0 1 2A 2 3 Li 6 .9 4 4 Be 9 .0 1 11 Na 2 3 .0 12 Mg 2 4 .3 3B 3 4B 4 5B 5 6B 6 7B 7 8 9 10 1B 11 2B 12 19 K 3 9 .1 20 Ca 4 0 .1 21 Sc 4 5 .0 22 Ti 4 7 .9 23 V 5 0 .9 24 Cr 5 2 .0 25 Mn 5 4 .9 26 Fe 5 5 .8 27 Co 5 8 .9 28 Ni 5 8 .7 29 Cu 6 3 .5 37 Rb 8 5 .5 38 Sr 8 7 .6 39 Y 8 8 .9 40 Zr 9 1 .2 41 Nb 9 2 .9 42 Mo 9 5 .9 43 Tc (9 8 ) 44 Ru 101 45 Rh 103 46 Pd 106 55 Cs 133 56 Ba 137 57 La 139 72 Hf 178 73 Ta 181 74 W 184 75 Re 186 76 Os 190 77 Ir 192 78 Pt 195 87 Fr (2 2 3 ) 88 Ra 226 89 Ac 227 8B 85 86 84 At Rn Po (2 0 9 ) (2 1 0 ) ( 2 2 2 ) 104 105 106 107 108 109 110 Rf Db Sg Bh Hs Mt Ds (2 6 1 ) ( 2 6 2 ) (2 6 3 ) (2 6 2 ) ( 2 6 5 ) (2 6 6 ) (2 8 1 ) Lant ha nide s 58 Ce 140 59 Pr 141 Act inide s 90 Th 232 91 Pa 231 60 Nd 144 92 U 238 61 62 63 64 65 66 67 68 69 70 71 Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu (1 4 5 ) 1 5 0 152 157 159 162 165 167 169 173 175 93 94 95 96 97 98 99 100 101 102 103 Np Pu Am Cm Bk Cf Es Fm Md No Lr (2 3 7 ) (2 4 4 ) (2 4 3 ) ( 2 4 7 ) (2 4 7 ) (2 5 1 ) ( 2 5 2 ) (2 5 7 ) (2 5 8 ) (2 5 9 ) (2 6 0 ) This project is funded by a grant awarded under the President’s Community Based Job Training Grant as implemented by the U.S. Department of Labor’s Employment and Training Administration (CB-15-162-06-60). NCC is an equal opportunity employer and does not discriminate on the following basis: against any individual in the United States, on the basis of race, color, religion, sex, national origin, age disability, political affiliation or belief; and against any beneficiary of programs financially assisted under Title I of the Workforce Investment Act of 1998 (WIA), on the basis of the beneficiary’s citizenship/status as a lawfully admitted immigrant authorized to work in the United States, or his or her participation in any WIA Title I-financially assisted program or activity. This product was funded by a grant awarded under the President’s High Growth Job Training Initiative, as implemented by the U.S. Department of Labor’s Employment & Training Administration. The information contained in this product was created by a grantee organization and does not necessarily reflect the official position of the U.S. Department of Labor. All references to non-governmental companies or organizations, their services, products, or resources are offered for informational purposes and should not be construed as an endorsement by the Department of Labor. This product is copyrighted by the institution that created it and is intended for individual organizational, non-commercial use only.