Supplementary File S1: cell surface protein isolation and
advertisement

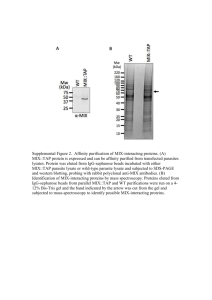
Supplementary File S1: cell surface protein isolation and mass spectrometry protocol. Cell surface protein isolation For the isolation and collection of surface proteins, we used the Pierce® Cell Surface Protein Isolation Kit (Thermo Scientific), generally following the protocol described by de Wit et al. [de Wit M. et al., 2012] (see Figure 1 of the main manuscript). Per biological replicate, 3x107 cells were cultured in five 75 cm2 flasks. Prior to surface protein biotinylation, all reagents were cooled to 4ºC. The cells were washed four times with ice-cold phosphate buffered saline (PBS) followed by incubation with 0.25 mg/mL Sulfo-NHS-SS-Biotin in 48 mL ice-cold PBS per flask on a rocking platform for 30 minutes at 4ºC. The biotinylation reaction was quenched by adding 500 μL of the provided Quenching Solution (Pierce). Cells were washed with ice-cold PBS, harvested by gentle scraping and pelleted by centrifugation. The cells were lysed using the provided Lysis Buffer (Pierce) containing a protease inhibitor cocktail (Sigma) for 30 minutes on ice with intermittent vortexing. To rid cell remnants, the lysates were centrifuged at 16,000 x g for 2 minutes at 4ºC. The clarified supernatant was used for purification of biotinylated proteins on NeutrAvidin Agarose. Before use, 500 μL of NeutrAvidin Agarose slurry was washed thrice with Pierce Wash Buffer in a provided column (Pierce). The clarified supernatant was added to the slurry and incubated for 2 h at 4ºC in the closed column using an endover-end tumbler to mix vigorously and allow the biotinylated proteins to bind to the NeutrAvidin Agarose slurry. The unbound proteins, representing the intracellular fraction, were collected by centrifugation of the column at 1,000 x g for 2 minutes and stored at -20ºC to serve as an internal control for the surface protein isolation process. Any remaining unbound proteins were removed by repetitive washing; thrice with 1% Nonidet-P40 and 0.1% SDS in 500 μL PBS and thrice with 0.1% Nonidet-P40 and 0.5 M NaCl in 500 μL PBS. Finally, the captured surface proteins were eluted from the biotin-NeutrAvidin Agarose by incubation with 330 μL of 50 nM dithiothreitol (DTT) in PBS containing 62.5 mM Tris-HCl for 1 h at room temperature (RT) in the end-over-end tumbler. The eluted proteins, representing the cell surface proteins, were collected by column centrifugation at 1,000 x g for 2 minutes. For all cell lines, three biological replicates were obtained; the three cell surface protein lysates per cell line were pooled and concentrated ten times using a Microcon YM-10 filter (Millipore) with a Mw cut-off of 10 kDa that was operated at 14,000 x g for 30 minutes at 25ºC to obtain adequate protein concentrations for gel-electrophoresis. Protein concentrations were quantified using the BCA protein Assay Kit (Pierce) and the lysates were stored at -20ºC until use. Gel-electrophoresis Of each sample, approximately 30 μg of cell surface protein lysate was separated by 1D gelelectrophoresis on a NuPAGE 4-12% Bis-Tris Gel (Invitrogen). The obtained gel was fixed in 50% ethanol containing 3% phosphoric acid for 1 h, rinsed for three consecutive times in Milli-Q water (MQ) and subsequently stained with Coomassie-R250 O/N on a rocking platform to visualise the protein bands (Figure 1). After staining, the gel was washed vigorously with MQ to rid the Coomassie and ORT-1 Hum31 Hum54 LM7 Cal-72 U2OS MG-63 SaOS-2 stored in MQ at 4ºC until further processing. kDa 250 — 150 — 100 — 75 — 50 — 37 — 25 — 20 — 15 — 10 — Figure S1: The Coomassie stained gel with the eight separated cell surface protein samples. This figure illustrates that the protein levels loaded onto the gel are equal across samples, which is of paramount importance when performing comparative proteomic studies based on spectral counting. Also, a different band pattern between the different cell lines, and, more importantly, different cell types can be observed. In-gel digestion The proteins were further processed into tryptic peptides according to the protocol described by Piersma et al., [Piersma et al., 2010] which was modified so that the pre-treatment phase of this protocol was applied to the whole gel in stead of to protein fractions. This allows for as good retrieval of peptides whilst reducing laboriousness of this procedure. [Pham et al., 2012] In brief, the entire gel was washed and dehydrated in 50 nM ammonium bicarbonate pH 7.9 (ABC) and 50 nM ammonium bicarbonate (ABC) containing 50% acetonitrile (ACN), respectively. Cysteine bonds were reduced by incubation with 10 mM DTT for 1 h at 56ºC and alkylated with 50 mM iodoacetamide for 45 minutes at RT in the dark. The gel was washed, dehydrated, and washed again. Then, the gel was sliced into 10 bands per lane using a clean razor blade under keratin-free conditions. Thus, 10 protein fractions per cell line were obtained (Figure 1). Each fraction was further cut into 1 mm 3 dimensioned gel pieces and washed in ABC/ACN. The slices were dried completely in a vacuum centrifuge (Thermo Fisher) and then incubated O/N with 100 µL of 6.25 ng/µL trypsin in 50 nM ABC at 25ºC. The peptides were extracted in 100 µL of 1% formic acid once, followed by extraction in 100 µL of 50% ACN in 5% formic acid twice. The combined volume of peptide fractions to be subjected to mass spectrometry by nanoliquid-chromatography tandem mass-spectrometry (nanoLC-MS/MS) was reduced to 50 µL in a vacuum centrifuge. Nano-LC separation Peptide separation was performed using an Ultimate 3000 nanoLC system (Dionex LC-Packings, Amsterdam, The Netherlands) equipped with a 20 cm × 75 µm ID fused silica column custom packed with 3 µm 120 Å ReproSil Pur C18 aqua (Dr Maisch GMBH, Ammerbuch-Entringen, Germany). After injection, peptides were trapped at 6 µL/min (1.6% ACN in 0.05% formic acid (FA)) on a 1 cm × 100 µm ID precolumn packed with 5 µm ReproSil Pur C18 aqua. Peptides were separated in a 60 minutes gradient (8-32% ACN in 0.05% FA) at 300 nL/min followed by washing (72% ACN in 0.05% FA) and equilibration (4% ACN in 0.05% FA). The inject-to-inject time was 90 minutes. Mass spectrometry Intact peptide MS spectra and MS/MS spectra were acquired on a LTQ-FT hybrid mass spectrometer (Thermo Fisher, Bremen, Germany) as described in detail in [Albrethsen et al., 2010;Piersma et al., 2010]. Intact masses were measured at 50,000 resolution in the ICR cell. In parallel, following an FT pre-scan, the top 5 peptide signals (charge-states 2+ and higher) were submitted to MS/MS in the linear ion trap (3 amu isolation width, 30 ms activation, 35% normalized activation energy, Q value of 0.25 and a threshold of 5,000 counts). Dynamic exclusion was applied with a repeat count of 1 and an exclusion time of 30 seconds. Protein identification and quantification To identify proteins from the acquired data, MS/MS spectra were searched against the human IPI database 3.62 (83,947 entries) using Sequest (version 27, rev 12) with a maximum allowed deviation of 10 ppm for the precursor mass and 1 amu for fragment masses. Methionine oxidation and cysteine carboxamidomethylation were allowed as variable modifications; two missed cleavages were also allowed. Scaffold 3.00.04 (Proteomesoftware, Portland, OR, USA) was used to organize the gel-slice data and to validate peptide and protein identifications. First, peptide identifications with a Peptide Prophet probability >95% were retained. Subsequently, protein identifications with 2 peptides or more in at least one of the samples and a Protein Prophet probability of >99% were retained. The proteins that contained similar peptides and could not be differentiated based on MS/MS analysis alone were grouped. For quantitative protein analysis spectral counting (the number of assigned MS/MS spectra for each identified protein) was used. For quantification across samples, the spectral counts were normalised to the sum of the spectral counts per biological sample. Differential analysis of samples was performed using the Beta-Binominal test as described previously. [Pham et al., 2010] Protein identification and quantification details can be found in [Pham et al., 2010;Piersma et al., 2010]. The obtained dataset was exported to Excel for further use. Data mining Subcellular protein localisations were verified using the Uniprot Knowledgebase (www.uniprot.org), searching under the header “GO annotation” for evidence of expression at the cell and/or plasma membrane. Protein-protein interactions (PPIs) were investigated using the Search Tool for the Retrieval of Interacting Genes/Proteins (STRING) version 9.0 (www.string-db.org). [Szlarczyk et al., 2011] For cluster and gene ontology (GO) analyses we used the Cytoscape platform for network analysis (www.cytoscape.org), using the plug-ins CluterONE version 0.91 (http://chianti.ucsd.edu/cyto_web/plugins/) for clustering and BINGO version 2.44 (http://www.psb.ugent.be/cbd/papers/BiNGO/) for the analysis of biological processes within our obtained networks, based on GO annotations. Additionally, verification of subcellular protein localisation was performed using Ingenuity pathway analysis software (IPA, Ingenuity Systems, Inc. CA, USA) and Biomart portal, version 7.0 (www.biomart.org). Analysis of surface protein enrichment To verify enrichment of cell surface proteome in our biotinylated fraction, we compared our surfaceenriched (biotinylated) protein lysates to intracellular (unbound) protein lysates and analysed the percentage of surface proteins in both fractions. The intracellular fractions of the osteosarcoma cell lines and human primary osteoblasts were pooled prior to work-up and mass spectrometric analysis. In this quality control, we only considered proteins with > 3 spectral counts per OS cell line. Proteins with a transmembrane and/or a signal peptide annotation using the Biomart portal version 7.0 (www.biomart.org) were considered predicted surface proteins. Within our surface protein enriched fraction, we retrieved 37% predicted surface proteins and compared to the whole lysate (10%). Thus, we indeed enriched our protein samples with surface molecules. Figure S2: Enrichment of surface proteins in whole lysate (intracellular fraction) and surface enriched (biotinylated) fraction. Analysis of the intracellular proteome also allowed for a comparison of intracellular EPHA2 expression in OS cell lines and primary human osteoblasts. The average spectral counts for intracellular EPHA2 in the OS cell lines was 11 compared to 0 in the human primary osteoblasts. Thus, apart from differential surface expression of EPHA2, the total protein expression of EPHA2 is also differentially regulated between OS cells and healthy bone cells. Reference List Albrethsen J, Knol JC, Piersma SR, Pham TV, de Wit M, Mongera S, Carvalho B, Verheul HM, Fijneman RJ, Meijer GA, Jimenez CR (2010) Subnuclear proteomics in colorectal cancer: identification of proteins enriched in the nuclear matrix fraction and regulation in adenoma to carcinoma progression. Mol Cell Proteomics 9: 988-1005 de Wit M., Jimenez CR, Carvalho B, Belien JA, Delis-van Diemen PM, Mongera S, Piersma SR, Vikas M, Navani S, Ponten F, Meijer GA, Fijneman RJ (2012) Cell surface proteomics identifies glucose transporter type 1 and prion protein as candidate biomarkers for colorectal adenoma-to-carcinoma progression. Gut 61: 855-864 Pham TV, Piersma SR, Oudgenoeg G, Jimenez CR (2012) Label-free mass spectrometry-based proteomics for biomarker discovery and validation. Expert Rev Mol Diagn 12: 343-359 Pham TV, Piersma SR, Warmoes M, Jimenez CR (2010) On the beta-binomial model for analysis of spectral count data in label-free tandem mass spectrometry-based proteomics. Bioinformatics 26: 363369 Piersma SR, Fiedler U, Span S, Lingnau A, Pham TV, Hoffmann S, Kubbutat MH, Jimenez CR (2010) Workflow comparison for label-free, quantitative secretome proteomics for cancer biomarker discovery: method evaluation, differential analysis, and verification in serum. J Proteome Res 9: 19131922 Szklarczyk D, Franceschini A, Kuhn M, simonovic M, Roth A, Minguez P, Doerks T, Stark M, Muller J, Bork P, Jensen LJ, von Mering C (2011) The STRING database in 2001: functional interaction networks of proteins, globally integrated and scored. Nucleid Acids Res 39: D561-D568