mixture fragment
advertisement

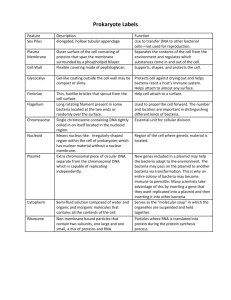
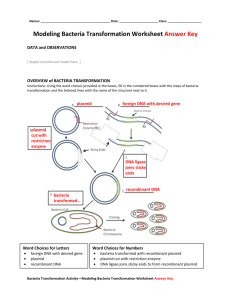
Fescue Chloroplast Genome Project - Part II Lab 2120 Chapter 13 addendum E. coli transformation Transformation of chemically competent Escherichia coli by heat shock. Goal – Transform E. coli with a newly engineered plasmid. Materials Competent E. coli strain JM109 Plasmid ligation mix LB liquid LB agar + ampicillin + IPTG + X-Gal Description and Protocol 1. The ligation mixture. Since the last lab your PCR reaction was completed and the products were separated using gel electrophoresis (you will experience gel electrophoresis first hand in two weeks). You will be given a picture of the gel containing all the samples from your lab section. Place the picture in the area below. Lab section ____________ Meeting time/day ___________ My PCR product(s) is in lane___________. It’s size is approximately ___________. The DNA band seen in your sample was cut from the gel and purified. The most efficient way to analyze your DNA fragment is to place it into a plasmid cloning vector with a known sequence. This vector allows us to use bacteria to cheaply and efficiently make trillions of copies (clones) of our DNA fragment. Since we know the sequence of the vector we now have several options regarding manipulation of the piece of DNA. The one we will be exploiting to sequence the DNA is defined primer sequences (marked as T7 and SP6 on the plasmid map below) that now flank our fragment. We will explore this topic more in lecture and when we get to the sequencing portion of this series. The ligation mix contains… Purified DNA fragment amplified from fescue pGEM-T Easy - plasmid designed to receive PCR products (plasmid is produced by Promega) Ligase – an enzyme which links the free 3’-OH of a linear strand of DNA to the 5’-PO4 of another. Ligation Buffer Tris (pH 7.8) - pH buffer MgCl2 – ligase cofactor DTT – reducing agent which helps prevent promiscuous ligations ATP – Ligase expends energy when linking strands Map of pGEM-T Easy PCR products are inserted here. Notice that your fragment will be inserted into a gene called lacZ. lacZ produces an enzyme called β–galactosidase. If the plasmid is reconnected to itself without the DNA insert the lacZ gene will be reconnected and will produce a functional enzyme in the bacterial cell. There is a dye precursor (X-Gal) in the culture plates you will use. If the enzyme is present it will catalyze X-Gal into a blue substrate making blue colonies. Alternatively, plasmids that do receive the DNA fragment will have an interrupted lacZ gene that will not produce an active enzyme and the colonies will remain white. 2. The bacterial cells The tube labeled JM has 50μl of high efficiency chemically competent E. coli cells, strain JM109. The term “chemically competent” means that the cells have been carefully grown, washed, and resuspended in a solution of calcium chloride (CaCl2). Bacterial cells have natural processes to take up DNA but for the purposes of screening plasmid clones they are very inefficient. Bacterial cells are much more likely to receive our engineered plasmids if we… Keep them ice cold Bathe them in a solution of CaCl2 Briefly heat them to 42°C in the presence of the plasmid (aka heat shock) The cells in the tube are ready to transform. It is very important that you keep them ice-cold until it is time to heat shock. 3. Transformation Protocol Add 2μl of ligation mixture to your JM109 E. coli and leave in the cooler for 20 minutes. Place the JM109 cells in the 42°C heat block for 45-50 seconds. Put the cells back into the cooler for 2 minutes Add the SOC bacterial growth medium and incubate at 37°C for ~30 minutes. Remove 100μl of transformed bacteria, squirt it onto an LB agar plate (LB + ampicillin, IPTG, and X-Gal), and spread the bacteria around the plate using a bent glass rod. LB - nutrient rich medium commonly used to grow E. coli Ampicillin - an antibiotic used to select bacteria that receive the plasmid from ones that do not IPTG – induces expression of the lacZ gene. X-Gal – dye precursor Label the plate with your group and lab section and incubate at 37°C. Chapter 13 addendum Laboratory Report 1. What is transformation? 2. What manipulations were performed outside of lab (beginning with the PCR products) to produce the plasmid you used to transform bacteria? 3. Make a drawing/diagram showing the plasmid cloning vector with your expected insert (it is not necessary to list all of the restriction sites shown on the map – label the region as the MCS for Multi-Cloning Site). 4. Why are we putting our DNA fragment into bacteria? 5. What is the purpose of “heat shocking” the bacteria? 6. When you examine your plates next week how will you know which bacteria have been successfully transformed? When you look at the colonies on the plate how will you know which ones received a plasmid carrying your fragment?