7-5 The Chemical Nature of Matter
advertisement

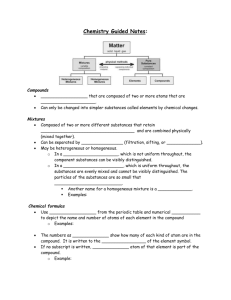
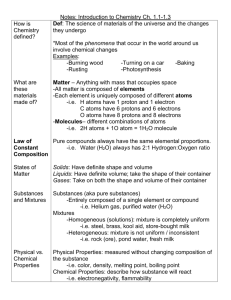
Academic Standard 7-5 7-5 Topic: The Chemical Nature of Matter The student will demonstrate an understanding of the classifications and properties of matter and the changes that matter undergoes. (Physical Science) Key Concepts Matter: atoms, elements, compounds, mixtures Physical Properties: melting point, boiling point, density, color Chemical Properties: reactivity, ability to burn, ability to rust Elements: metals, nonmetals Periodic Table: groups and families of elements Common substances: chemical symbols, chemical formulas Law of Conservation of Matter: balanced chemical equation, reactants, products Acids and Bases: indicators, litmus paper, pH paper, phenolphthalein, pH Physical changes: changes in size, shape, and state Chemical changes: changes in color or temperature, formation of precipitate or gas Indicators: 7-5.1 Recognize that matter is composed of extremely small particles called atoms. Taxonomy level: 1.1-B Remember Conceptual Knowledge Previous/Future knowledge: Students have been introduced to the concept of matter being composed of very small particles in 5th grade (5-5.1). Students have not been introduced to the concept of atoms in previous grades. Students will further develop the concept of atoms in 9th grade Physical Science (PS-2.1 and PS-2.2). It is essential for students to know that matter is composed of extremely small particles called atoms and that atoms are the smallest part of pure substances that cannot be broken down into simpler substances. It is not essential for students to know the subatomic particles, for example, protons, neutrons, and electrons, which compose atoms. Assessment Guidelines: The objective of this indicator is to recognize that matter is composed of extremely small particles called atoms; therefore, the primary focus of assessment should be to recall from long-term memory the information that atoms are the extremely small particles of matter. However, appropriate assessments should also require students to identify extremely small particles of matter as atoms. 1 Academic Standard 7-5 7-5.2 Topic: The Chemical Nature of Matter Classify matter as element, compound, or mixture on the basis of its composition. Taxonomy Level: 2.3-B Understand Conceptual Knowledge Previous/Future knowledge: Students have been introduced to the concept of mixtures in 5th grade (5-4.3). Students have not been introduced to the concept of elements or compounds in previous grades. Students will further develop the concepts of elements, compounds, and mixtures in high school Physical Science (PS-3.4). It is essential for students to know that matter can be classified on the basis of its composition as follows: Elements Elements are pure substances that cannot be changed into simpler substances. Elements are composed of one type of atom. Compounds Compounds are pure substances that are composed of two or more types of elements. Compounds can only be changed into simpler substances called elements by chemical changes (7-5.10), which are not as easy as separating mixtures into their components. Mixtures Mixtures are composed of different substances that are mixed together. They can be separated easily into their component substances by processes described in 5th grade (5-4.4). It is not essential for students to know that atoms combine to form molecules, which are the smallest part of compounds. Students also do not need to know that mixtures can be heterogeneous or homogeneous. Assessment Guidelines: The objective of this indicator is to classify matter as element, compound, or mixture on the basis of its composition; therefore, the primary focus of assessment should be to categorize matter as elements, compounds or mixtures depending on its composition. However, appropriate assessments should require students to recognize the classifications of matter based on its composition; to exemplify (give examples of) matter based on its composition; to illustrate with words, pictures, or diagrams categories of matter; to infer the types of matter based on characteristics; to compare the types of matter based on their characteristics. 2 Academic Standard 7-5 Topic: The Chemical Nature of Matter 7-5.3. Compare the physical properties of metals and nonmetals. Taxonomy Level: 2.6-B Understand Conceptual Knowledge Previous/Future knowledge: Students have been introduced to the concept of physical properties in 5th grade (5-4.2). Students have not been introduced the concept metals and nonmetals in previous grade levels. Students will further develop the concepts of metals and nonmetals in 9th grade Physical Science (PS-2.3) It is essential for students to know that metals and nonmetals are two major groups of elements that have different physical properties as follows: Metals Nonmetals Physical properties of metals include the following: Luster Having a shiny surface or reflecting light brightly Conductors Heat and electricity move through them easily. Malleable Ability to be hammered into different shapes Ductile Ability to be drawn into a wire High density Heavy for their size Physical properties of nonmetals include the following: Dull Not shiny Nonconductors Heat and electricity do not move through them easily Brittle Break or shatter easily It is not essential for students to know the chemical properties of metals versus nonmetals, or the electron arrangement in atoms of metals versus nonmetals. Assessment Guidelines: The objective of this indicator is to compare the physical properties of metals and nonmetals; therefore, the primary focus of assessment should be to determine the differences between metals or nonmetals based on their physical properties. However, appropriate assessments should also require students to infer from a list of physical properties whether an object is metal or nonmetal; to recall physical properties of metals and nonmetals; or to summarize physical properties of metals and nonmetals. 3 Academic Standard 7-5 Topic: The Chemical Nature of Matter 7-5.4 Use the periodic table to identify the basic organization of elements and groups of elements (including metals, nonmetals, and families). Taxonomy Level: 3.2-B Apply Conceptual Knowledge Previous/Future knowledge: Students have not been introduced to the periodic table in previous grade levels. Students will further develop the concept of the periodic table in 9th grade Physical Science (PS-2.3). It is essential for students to know how to use the periodic table to identify the basic organization of elements. The elements on the periodic table are identified with numbers increasing from left to right across each row of the table. Along with this number (atomic number) there is also a symbol that identifies each element on the table. There are two large sections of elements on the periodic table. On the periodic table there is a zigzag line on the right side of the table. Metals are to the left of this line. Nonmetals are to the right. Families Also called groups, are columns of elements on the periodic table that have many similar properties; they are usually numbered 1-18. A family of elements has some chemical characteristics that are similar. Metals Groups A major classification of elements generally located on the left side of the periodic table. Examples on metals are: Sodium (Na), Calcium (Ca), Iron (Fe), and Aluminum (Al). Nonmetal Families A major classification of elements generally located on the right side of the periodic table. Examples of nonmetals are: Chlorine (Cl), Oxygen (O), Sulfur (S), and Iodine (I). It is not essential for students to know how each element gets its atomic number (no subatomic particles are part of this standard). They do not need to know the names of the individual families or groups of elements or to identify metalloids. Assessment Guidelines: The objective of this indicator is to use the periodic table to identify the basic organization of elements on the periodic table, the metal and nonmetal divisions, and to locate families of elements; therefore, the primary focus of assessment should be to retrieve knowledge about the periodic table as identified. However, appropriate assessments should also require students to exemplify elements that are metals and nonmetals given a periodic table; recognize the location of an element on the periodic table; recognize the location of groups of metals and nonmetals; to recognize that families are columns of elements; or identify an element based on its atomic number or symbol. 4 Academic Standard 7-5 Topic: The Chemical Nature of Matter 7-5.5 Translate chemical symbols and the chemical formulas of common substances to show the component parts of the substances (including NaCl [salt], H20 [water], C6H1206 [simple sugar], 02 [oxygen gas], C02 [carbon dioxide], and N2 [nitrogen gas]). Taxonomy Level: 2.1-B Understand Conceptual Knowledge Previous/Future knowledge: Students have not been introduced to the concepts of chemical symbols and the chemical formulas in previous grades. Students will further develop the concepts of chemical symbols and chemical formulas in 9th grade Physical Science (PS-4.5). It is essential for students to know that chemical symbols show the atoms of the elements composing a substance. Atoms of common elements have different symbols as follows: Symbol Na Cl H 0 C N Element Sodium Chlorine Hydrogen Oxygen Carbon Nitrogen Chemical formulas are constructed from the symbols of the elements composing the substances. Elements are made up of one type of atom and their formulas have one symbol. Compounds are composed of more than one element and their formulas have more than one type of symbol showing the different atoms that compose the compound. In a chemical formula, the numbers as subscripts show the ratio of the number of atoms of the different elements in the compound. It is not essential for students to know chemical symbols and chemical formulas for substances not listed in 7-5.5. Assessment Guidelines The objective of this indicator is to translate chemical symbols and chemical formulas of common substances listed above to show the component parts; therefore, the primary focus of assessment should be to interpret a chemical symbol and formula to identify the element(s) and the number of atoms of that element in a formula of the substance. However, appropriate assessments should also require students to recognize the chemical symbols and formulas of common substances to recall the components. 5 Academic Standard 7-5 Topic: The Chemical Nature of Matter 7-5.6 Distinguish between acids and bases and use indicators (including litmus paper, pH paper, and phenolphthalein) to determine their relative pH. Taxonomy level: 4.1-B Analyze Conceptual Knowledge Previous/Future knowledge: Students have been introduced the concept of solutions, solvents, solutes, and concentrations in 5th grade (5-4.5). Students have not been introduced to the concept of solutions of acids and bases nor their concentrations in previous grades. They will further develop these concepts of acids and bases in 9th grade Physical Science (PS-3.8). It is essential for students to know that acids and bases are solutions usually with water as the solvent: Acids Acids can be identified by their sour taste (lemons and oranges contain acids)—but students should not use a taste test on laboratory chemicals; by their reaction with some metals (for example, zinc), and by their reaction with bases to form a neutral pH solution. Bases Bases can be identified by their bitter taste (soapsuds have a bitter taste)—but students should not use a taste test on laboratory chemicals; by their slippery feels when rubbed between thumb and fingers---but some strong bases burn the skin so this test should not be used; and by their reaction with acids to form a neutral pH solution. pH pH is a measure of the concentration of solutions of acids and bases and is measured on a scale of 1-14. A basic solution has a pH greater than 7 and an acidic solution has a pH less than 7. If a solution is neither an acid nor a base, it has a pH of 7 and is called neutral. Pure water has a pH of 7. The stronger the acid, the lower the pH (less than 7) while the stronger the base, the higher the pH (greater than 7). Indicators Indicators are substances that can be used to determine the whether a solution is an acid or base. The following indicators can be used to determine relative pH Litmus paper Blue litmus paper turns red in an acid, and stays blue in a base. Red litmus paper turns blue in a base, and stays red in an acid. Both red and blue litmus paper turn purple in a solution that is neutral (neither an acid nor a base). pH paper pH paper can turn a series of colors in solutions of pH. The color of the paper is compared to the chart of the vial to determine the pH. Phenolphthalein Phenolphthalein turns magenta in a strong base and stays clear in an acid It is not essential for students to know the specific colors of pH paper at given pH ranges, that a solution is called “neutral” if it is neither an acid or a base, or how to neutralize a solution to form water and salt. 6 Academic Standard 7-5 Topic: The Chemical Nature of Matter Assessment Guidelines: One objective of this indicator is to distinguish between acids and bases, therefore, the primary focus of assessment should be to analyze properties of solutions to determine whether they are acids or bases. Another objective is to use indicators to determine relative pH; therefore, the primary focus of assessment should be to apply a procedure that uses indicators to determine the relative acidic or basic concentration of the solutions. However, appropriate assessments should also require students to identify solutions as acidic or basic based on properties; to identify solutions as more acidic or more basic depending on pH results with indicators; summarize the use of indicators in determining the pH of a solution; or infer the pH of a solution given the results of a particular indicator. 7 Academic Standard 7-5 Topic: The Chemical Nature of Matter 7-5.7 Identify the reactants and products in chemical equations. Taxonomy Level: 1.1-B Remember Conceptual Knowledge Previous/Future knowledge: Students have been introduced to the concept of forming new substances when they chemically combine in 5th grade (5-4.7). Students have not been introduced to the concept of reactants or products or chemical equations in previous grades. Students will further develop the concept of chemical reactions and equations in 9th grade Physical Science (PS4.6 and PS-4.7). It is essential for students to know that when substances are mixed and at least one new substance is formed, a chemical reaction has occurred. Chemical equations Are used to represent chemical reactions that have occurred. They contain the chemical formulas (7-5.5) for the substances that are mixed and the new substance(s) that are formed. An arrow separates them. Reactants The substances that are mixed in a chemical reaction are called the reactants and appear on the left side of the arrow in a chemical equation. Products The new substances that are formed in a chemical reaction are called the products and appear on the right side of the arrow in a chemical equation. For example, the following chemical equation shows the formation of water (H20) from oxygen (02) and hydrogen (H2). The reactants are oxygen (02) and hydrogen (H2), located on the left side of the arrow. The product, water (H20), is on the right side of the arrow. 2H2 + 02 → Reactants → 2H20 Products It is not essential for students to know how to balance chemical equations by adding coefficients in front of the chemical formulas or to name the various types of chemical reactions that occur (for example, single displacement, double displacement, decomposition, or synthesis). Assessment Guidelines: The objective of this indicator is to identify reactants and products in chemical equations; therefore, the primary focus of assessment should be to recognize the reactants and products in a chemical equation. However, appropriate assessments should also require students to recall that reactants are before the chemical reaction and products are after the chemical reaction shown by the arrow of the equation. 8 Academic Standard 7-5 Topic: The Chemical Nature of Matter 7-5.8 Explain how a balanced chemical equation supports the law of conservation of matter. Taxonomy Level: 2.7-B Understand Conceptual Knowledge Previous/Future knowledge: Students have been introduced to the concept of the law of conservation of energy during energy transformations in 6th grade (6-5.2). Students have not been introduced to the concept of the law of conservation of matter in previous grades. Students develop the concept of chemical equations in this grade (7-5.7). Students will further develop this concept of balanced chemical equations and conservation of mass in 9th grade Physical Science (PS-4.7). It is essential for students to know that the amount of matter does not change during a chemical reaction; only the atoms are rearranged to form new substances. The law of conservation of matter is based on countless experiments in which the mass of the reactants has been found to equal the mass of the products. The law of conservation of matter states that matter can neither be created nor destroyed, but can be changed in form. In other words, the total mass of the material(s) before the reaction is the same as the total mass of material(s) after the reaction. A balanced chemical equation has the same number of each kind of atom on both sides of the arrow. When students multiply the number in front of the chemical formula in the equation by each number written below the symbol of the atoms in the formula, the number of each kind of atom on the left side of the arrow must equal the number of each kind of atom on the right side of the arrow for the equation to be balanced In the chemical equation for the reaction of water (liquid) changing to hydrogen and oxygen (gas) 2H20 2H2 + 02 There are 2 x 2 Hydrogen (H) = 4 Hydrogen (H) atoms on the left of the arrow and 2 x 2 Hydrogen (H) = 4 Hydrogen (H) atoms on the right of the arrow; There are 2 x 1 Oxygen (O)= 2 Oxygen (O) atoms on the left of the arrow and 1 x 2 Oxygen (O) atoms on the right of the arrow; (if no number appears in front of the formula or below the atom, it is an understood “1”). Therefore, there are the same numbers of atoms of hydrogen (4) and oxygen (2) on both sides of the equation and the equation is said to be balanced. Since there are the same number of each kind of atom on both sides of the arrow and atoms represent kinds of matter, the amount of matter is the same on both sides of the equation, which supports the law of conservation of matter. It is not essential for students to know how to add numbers in front of chemical formulas to balance chemical equations. They must only recognize a balanced equation. They do not have to know how to create the chemical formulas for substances. They must only analyze given formulas for common substances to show the component parts. Assessment Guidelines: The objective of this indicator is to explain how a balanced equation supports the law of conservation of matter; therefore the primary focus of assessment should be to construct cause and effect models of how a balanced equation supports the law of conservation of matter. However, appropriate assessments should also require students to recall the law of conservation of matter; summarize how the law of conservation of matter relates to a balanced equation; or recognize whether or not a chemical equation is balanced or not. 9 Academic Standard 7-5 Topic: The Chemical Nature of Matter 7-5.9 Compare physical properties of matter (including melting or boiling point, density, and color) to the chemical property of reactivity with a certain substance (including the ability to burn or to rust). Taxonomy level: 2.6-B Understand Conceptual Knowledge Previous/Future knowledge: Students have been introduced to the concept of properties of matter in 2nd grade (2-4.1), in 3rd grade (3-4.1), and physical properties of the states of matter in 5th grade (5-4.2). Students have not been introduced to the concept of physical properties or chemical properties of matter in previous grades. Students will further develop the concept of physical and chemical properties in 9th grade Physical Science (PS-3.1). It is essential for students to know that physical properties of matter can be observed and measured without changing the kind of matter being studied. Chemical properties are not usually visible. They can be recognized only when substances react chemically with one another, that is, when they undergo a change in composition. The following physical properties can be used to help identify a substance: Melting Point Melting point is the temperature at which a solid can change to a liquid. The temperature at which a pure substance melts is unchanging. Therefore, the melting point of a pure substance can be used as a physical property for identifying it. Ice melts to form water at 00C or 32oF. Boiling Point Boiling point is the temperature at which a liquid changes state into a gas. The liquid starts to form bubbles throughout, which grow larger, rise to the surface, and burst. This temperature is unchanging for a given substance, at sea level, and can be used to identify that substance. The boiling point for pure water is 100oC or 212oF. Density Density, defined qualitatively, is the measure of the relative “heaviness” of objects with a constant volume, is the mass of a given volume of a substance. The density of a given substance is unchanging and can be used as a physical property to identify it. The density of water is a universal standard: 1 milliliter of water (at 4oC) equals 1 cubic centimeter of water equals 1 gram of water. The density of water is 1 g/mL or 1 g/cm3 or 1g/cc. Lead for example is a very heavy, dense metal. The density of lead is much greater than the density of the very light metal, aluminum. Color Color of a given substance is distinctive and can be used to help identify a substance. Chemical properties can also be used to help identify a substance. Chemical properties are not usually visible. Chemical properties can be recognized only when substances react chemically with one another, that is, when they undergo a change in composition. Chemical reactivity is the tendency of a substance to undergo a chemical reaction usually with other specific substances. When a substance reacts with oxygen quickly and releases heat and light energy, the process is called burning. When a substance, for example iron, reacts with oxygen and water slowly, the process is called rusting. 10 Academic Standard 7-5 Topic: The Chemical Nature of Matter It is not essential for students to calculate the density of substances if given the mass and the volume of the substances. Assessment Guidelines The objective of this indicator is to compare physical and chemical properties of matter; therefore, the primary focus of assessment should be to determine the differences between physical and chemical properties of matter. However, appropriate assessments should also require students to classify properties as being physical or chemical; to summarize how physical or chemical properties help identify substances; or exemplify (give examples of) physical and chemical properties used to identify substances. 11 Academic Standard 7-5 Topic: The Chemical Nature of Matter 7-5.10 Compare physical changes (including changes in size, shape, and state) to chemical changes that are the result of chemical reactions (including changes in color or temperature and formation of a precipitate or gas). Taxonomy Level: 2.6-B Understand Conceptual Knowledge Previous/Future knowledge: Students have not been introduced to the concepts of physical or chemical changes before this grade level. They will further develop the concept of physical and chemical changes in 9th grade Physical Science (PS-4.6). It is essential for students to know that physical changes do not change the identity of a substance. When a substance changes state, the substance does not change identity. A change in state, therefore, is a physical change. When substances change in size or shape, they do not change identity; therefore, these changes are also physical changes. When the following changes occur, however, the substance usually changes identity; therefore, the changes are chemical changes: Color change When a substance changes color, the chemical composition of the substance has usually changed; therefore, this is a chemical change. Temperature change When substances change temperature after being mixed with other substances, the chemical composition of the substances has usually changed; therefore, this is a chemical change. Precipitate formation When two or more liquids are mixed and a solid (precipitate) forms, the chemical composition of the substances has usually changed; therefore, this is a chemical change. Gas formation When substances are mixed or heated and a gas is given off, the chemical composition of the substances has usually changed; therefore, this is a chemical change. It is not essential for students to identify the type of chemical change that has occurred, nor to identify a reaction as exothermic or endothermic depending on the temperature change. Assessment Guidelines The objective of this indicator is to compare physical and chemical changes; therefore, the primary focus of assessment should be to determine how physical and chemical changes are alike and different. However, appropriate assessments should also require students to exemplify (give examples of) physical and chemical changes; to infer if a physical or chemical change has occurred based on the conditions given; or to classify changes as chemical or physical based on descriptions given. 12