Supplementary Materials and Methods (doc 72K)
advertisement

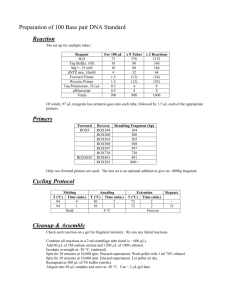
SUPPLEMENTARY MATERIALS AND METHODS Promoter cloning. The HCMV IE promoter was obtained from pCK plasmid.1 Human cellular promoters were amplified from HT1080 or K562 cells using the indicated primers. Promoters Primers EF1α (-341~+1007) 5’- ACGCGTGTAAGCCAGCAATGGTAGAGGGAAGATTCTGCACG-3’ GAPDH (-350~+315) 5’-ACGCGTTTCATCCAAGCGTGTAAGGG-3’ RPL10 (-350~+143) 5’-ACGCGT AGGCCCACCTAGGGTACTTTCCTTT-3’ Source of genomic DNA 5’-GGATCCTTTT GGCTTTTAGG GGTAGTTTTCACGACACC-3’ 5’-GTTTAAACGGTGTCTGAGCGATGTGGCT-3’ 5’-GGATCCGGCGACACCAGGATCTTCAGTGGCT-3’ 5’- ACGCGTAGAATTGTTTGAACCCAGGAGGCGG-3’ LENG8 (-385~+305, +1907~+2121) 5’-GTTTAAACAAAGTAGAAGACGACGGCGCACGCG-3’ 5’-GTTTAAACCCACACCCAGAACTCTTCAGATCCT-3’ 5’-GAATTCCTGGACCTTGGGGTATAAGGGGTGG-3’ SNX3 (-353~+338) 5’-GAATTCAATCCAGACGCGTGTCTGGTGCAA-3’ 5’-GGATCCTTCGCTGTAGCTGCTG-3’ 5’-ACGCGTGTAGCTCCTCCCCCAGACCAATTGTTTTAAG-3’ CNOT3 (-350~+654, +5076~+5266) 5’-GGATCCTCCATCCTTCCAGCCAGGAGCCAATACCGAC-3’ 5’-AGATCTTGGGGCTGGTCTCTTGTCAGATAGC-3’ HT1080 5’-GGATCCCTTCCCTGCCCTACAGACGCACTCT-3’ 5’-ACGCGTGTCCATTTAATCCTCAAAAAACTTA-3’ CPNE1 (-300~+489, +5612~+5999) 5’-GGATCCTTTTTACTGCAGTCCCCGTTATTAGCTC-3’ 5’-AGATCTAGCTGTGAAGCTGAGCTTTATGACT-3’ 5’-GGATCCCTGATAAAACAAGAGATGAATTTCC-3’ HYPO (-350~+66) 5’-ACGCGTTCTTTTACACGTTTGGTTTTATGGT-3’ DKC1 (-473~+91) 5’-ACGCGTGCACACTACTCCTATTGGC-3’ VPS72 (-466~+43) 5’-ACGCGTACAAAAATTAGTTGGGCAT-3’ ITGB4BP (-350~+304) 5’-ACGCGTTCTGTCCCTCAAGGCACAGCT-3’ β-actin (-387~+944) 5’- ACGCGTGAGATGTCCACACCTAGGATGTCC-3’ 5’-GGATCCGGCTGCAACAGGCCAGGAAACCTTC-3’ 5’-GAATTCGTTACCCTGCACCGCGTGC-3’ 5'-GAATTCACCGCCTACCGAGACTGCG-3’ 5’-GTTTAAACGAGGCCTAGGGGCGGCGGAGGCGGGAGTTCAA-3’ 5’-GGATCCGGTGAGCTGCGAGAATAGCCG-3’ All amplified fragments were initially cloned into pGem T easy. K562 Deletion analysis of RPL10 promoter. -50, -100, -200, -350, -500, -1000 bp RPL10 promoters were amplified using forward primers RPL F50 (5’ACGCGTACGCGCGCAGACAGACCGCCTATATAAGCCAT-3’), RPL F100 (5’ACGCGTTGACGTCTGACAGAGCGTCCACCCGTCTTCG-3’), RPL F200 (5’ACGCGTCTGGCCGCCCGCGGCCCTGGTACCCGGTCACC-3’), RPL F (5’ACGCGTAGGCCCACCTAGGGTACTTTCCTTT-3’), RPL F500 (5’ACGCGTGTCTCCCCCTCCGGCCTCCCGGGTTGACAAAGG-3’), and RPL F1000 (5’ACGCGTGTGCGCTCGAGCAGGATTTCCTCCCGTCCTTCC-3’), respectively, and the reverse primer RPL R (5’-GGATCC GGCGACACCAGGATCTTCAGTGGCT-3’). Genomic DNA from HT1080 was used as a template. The amplified fragments were initially cloned into pGem T easy. The BamHI-BglII fragment of pGem T easy-eGFP was cloned to the BamHI site of pGem T easy-RGpA, resulting in pGem T easy-eGFP-RGpA. The MluIBamHI fragments from pGem T easy-RPL10 promoters were cloned to the MluI-BamHI site of pGem T easy-eGFP-RGpA. The respective vectors were transfected to 293T using FuGene6, according to the manufacturer’s instructions. Two days later, cells were analyzed by FACS. Construction of retroviral vectors. Retroviral plasmid MT has been described previously.2 ID was derived from MT by deleting 310 bp from the 447 bp-long U3 present in the 3’ LTR. The 3’LTR of MLV was amplified using primers SCV3LB (5’GGATCCCTCGAGCGATAAAATAAAAGATTTTATTTAGTCTCC-3’) and SCV3LRI (5’GAATTCGTCGACTGAAAGACCCCCGCTGACGG-3’) with MT as a template. The amplified fragment was initially cloned into pGem T easy (Promega, Madison, WI), resulting in pGem T easy-3’LTR. A deleted form of 3’LTR was amplified using primers 3’LTR-1 (5’GCTAGCCCCTGTGCCTTATTTGAA-3’) and SCV3LRI (5’GAATTCGTCGACTGAAAGACCCCCGCTGACGG-3’) with MT as a template. The amplified fragment was cloned into pGem T easy, resulting in pGem T easy-3’dLTR-1. The NheI-SalI fragment was cloned into the NheI-SalI site of pGem T easy-3’LTR, resulting in pre SIN2. The BamHI-SalI fragment from pre SIN2 was cloned into the BamHI-SalI site of MS3 to make ID. A 52bp-length fragment of new MCS was made by the polymerase reaction using primers NEWMCSF (5’-ACGCGTTTAAACCGCGGAATTCGGATCCACATCGTG3’) and NEWMCSR (5’-CTCGAGATCTAGGCCTCACGATGTGGATCCGAATTC-3’) without template. The amplified fragment was introduced into pGem T easy, and the MluIXhoI fragment was cloned into the MluI-XhoI site of ID. To make the retroviral vector convenient for linearization, two restriction enzyme sites were introduced to ID. The L1 fragment was amplified using primers L1F (5’GCTCTTCCGCTCACGTGTGATCAATTTAAATTTCGAA-3’) and L1R (5’AGCGGAAGAGCTTCGAAATTTAAATTGATCACACGTG-3’) without template, and cloned into pGem T easy, resulting in pGem T easy-L1. The SapI fragment released from pGem T easy-L1 was inserted into the SapI site of ID. The L2 fragment was amplified using primers L2F (5’-AGGCCTGGTCACCGGCCATTATGGCCACGTGATCATTTAAATTTG3’) and L2R (5’-TATTCGCGCGTTTCGGTGATGAATATT-3’) with pUC18 (Promega, Madison, WI) as a template. The amplified L2 fragment was cloned into pGem T easy, generating pGem T easy-L2. The StuI-SspI fragment released from pGem T easy-L2 was introduced into the SspI site of ID. To select cells transfected with vector DNA, a selectable marker gene cassette was inserted into ID. To construct the neomycin resistant gene cassette, the promoter of human β-actin was amplified using primers BApF (5’GTCGACATTAATGCCGGTGAGTGAGCGGCGCGGGGCCAA-3’) and BApR (5’GGATCCGGTGGCGCGTCGCGCCGCTGGGTTTT-3’) with gDNA from K562 as a template. The amplified fragments were initially cloned into pGem T easy, resulting in pGem T easy-pBA. Neo-polyadenylation signal was amplified using primers NeoF (5’- AGATCTATGGGATCGGCCATTGAACAA-3’) and pAR (5’CATATGTCATAATCAGCCATACCACATTT-3’) with pGem T easy-SVneo-pA as a template. The amplified fragments were cloned into pGem T easy, producing pGem T easy-SVNeo-pA. The BglII-NdeI fragment released from pGem T easy-SVNeo-pA was inserted into the BamHI-NdeI site of pGem T easy-pBA, to make pGem T easy-pBA-SVNeo-pA. The MluIEcoRI fragment filled in with Klenow was cloned into the SspI site of ID. The MluI-BamHI fragment of pGem T easy- RPL10 promoter (1 kb) was cloned to the MluI-BamHI site of ID, resulting in RKD. The eGFP gene was amplified using primers eGFP5 (5’-ACGCGTGGATCCATGGTGAGCAAGGGCGAG-3’) and eGFP3 (5’CTCGAGAGATCTTTACTTGTACAGCTCGTC-3’) with pIRES2-EGFP (CLONTECH Laboratory, Palo Alto, CA) as a template. The amplified eGFP sequence was cloned into pGem T easy to generate pGem T easy-eGFP. The BamHI-BglII fragment was cloned into the BamHI site of RKD, resulting in RKD-eGFP. The human gp91phox gene was amplified using primers gp91 phox 5 (5’-GGATCCATGGGGAACTGGGCTGTGAAT-3’) and gp91 phox 3 (5’-GGATCCCTCGAGTTAGAAGTTTTCCTTGTT-3’) with cDNA isolated from human peripheral blood lymphocytes as a template. The amplified fragment was cloned into pGem T easy to generate pGem T easy-gp91. The BamHI fragment was cloned into the BamHI site of RKD, resulting in RKD-gp91. To construct ID containing an expressing cassette in a reverse orientation, the reverse new MCS was cloned. Two 56 bp oligomers, R-new MCS F (5'TCGAACGCGTTTAAACCGCGGAATTCGGATCCACATCGTGAGGCCTAGATCTCGA G-3') and R-new MCS R (5'CGCGCTCGAGATCTAGGCCTCACGATGTGGATCCGAATTCCGCGGTTTAAACGCGT R-3'), were annealed and cloned into the MluI-XhoI site of ID. The polyadenylation signal sequence of rabbit β-globin gene (RGpA) was amplified using primers RGpA F (5’– GGATCCTTTTCCCTCTGCCAAA-3’) and RGpA R (5’ – ACTAGTATAAGAGAAGAGGGACAGC-3’) with cosmid vector pAxCAwt (Takara Bio, Japan) as a template. The amplified fragment was cloned into pGem T easy to generate pGem T easy-RGpA. The EcoRI fragment (Klenow-blunted) of pGem T easy-RGpA was inserted in the BglII site (Klenow-blunted) of ID, resulting in ID-RGpA. The RPL10 promoter (1 kb) and gp91phox cDNA sequence were subsequently cloned into this plasmid, resulting in ROKD-gp91. To construct ROKT-gp91 and ROK-gp91, deleted forms of 3’LTR were amplified using forward primers 3’LTR-2 (5’-GCTAGCTTCGCGCGCTTCTGCTCC-3’) and 3’LTR-3 (5’-GCTAGCCCCACAACCCCTCACTCG-3’), respectively, and the reverse primer SCV3LRI (5’-GAATTC GTCGACTGAAAGACCCCCGCTGACGG-3’). MT was used as a template. The amplified fragments were initially cloned into pGem T easy, resulting in pGem T easy-3’dLTR-2 and pGem T easy-3’dLTR-3. The NheI-SalI fragments were cloned into the NheI-SalI site of pGem T easy-3’LTR, resulting in pre-SIN3 and pre-SIN4. The BamHI-SalI fragments from pre-SIN3 and pre-SIN4 were cloned into the BamHI-SalI site of ID, resulting in ID3 and ID4. The reverse new MCS, the RPL10 promoter (1 kb), gp91phox cDNA sequence, and RGpA were subsequently introduced into these plasmids, resulting in ROKT-gp91 and ROK-gp91. Staining of gp91phox-positive cells. 5 X 105 cells tranduced with gp91phox expressing retroviral vector were incubated on ice with monoclonal antibody 7D5 (MBL Medical & Biological Laboratories, NaKa-ku Nagoya, Japan). After 30 min, cells were washed and incubated for 30 min on ice with R-PE-labeled goat-anti-mouse IgG1 antibody (Southern Biotech, Birmingham, AL). After incubation, cells were analyzed by FACS. Determination of vector copy number. gDNA was prepared from transduced cells using Puregene Blood Core Kit A (Qiagen, Hilden, Germany). Real-time quantitative PCR was performed according to the ABI 7300 system’s protocol (Applied Biosystems), using the following primers: GFP forward, 5’CAGGAGCGCACCATCTTCTT -3’, GFP reverse, 5’- GCGGCGGTCACGAACTC -3’, gp91phox forward, 5’- TGGTGGCATGGATGATTGC -3’, gp91phox reverse, 5’CAAAGTAAGACCTCCGGATGGT -3’, β-actin forward, 5’CAGCGGAACCGCTCATTGCCAATGG -3’, β-actin reverse, 5’TCACCCACACTGTGCCCATCTACGA -3’. The average vector copy number was calculated from the slope of the standard curve made from dilutions of K562 gDNA containing single copy of GFP or gp91phox gene. SUPPLEMENTARY REFERENCES 1. Lee Y, Park E, Yu S, Kim D, Kim S. Improved Expression of Vascular Endothelial Growth Factor by Naked DNA in Mouse Skeletal Muscles: Implication for Gene Therapy of Ischemic Diseases. Biochemical and Biophysical Research Communications 2000; 272: 230-235. 2. Youngtae Hong SSY, Jong-Mook Kim, Karim Lee, Young Soon Na, Chester B. Whitley, Yoshikazu Sugimoto, Sunyoung Kim. Construction of a high efficiency retroviral vector for gene therapy of Hunter’s syndrome. J Gene Med 2003; 5: 18-29. 3. Kim S, Lee K, Kim M, Kang S, Joo C, Kim J et al. Factors affecting the performance of different long terminal repeats in the retroviral vector. Biochemical and Biophysical Research Communications 2006; 343: 1017-1022.