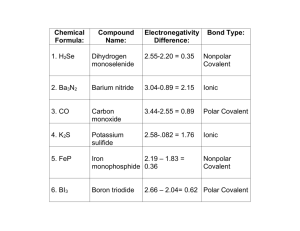
Covalent Bond 14
advertisement

Name: Block: Covalent Bonds Practice Introduction: In this activity you will create electron dot diagrams. illustrate one way that atoms can bond. These diagrams will Prelab: Define the following terms. 1. Chemical Bond: 2. Valence Electrons: 3. Octet: 4. Covalent Bond: Answer the following questions. 1. When chemical bonds form between atoms, what are the three things that electrons do? 2. When is an atom chemically stable? Compound #1 Element Number of valence electrons Electrons needed to form an octet? Electron Dot Diagram of Element Structure & Compound Formula Electron Dot Diagram of Compound Fluorine F Fluorine F Element Number of valence electrons Electrons needed to form an octet? Compound #2 Electron Structure Dot & Diagram of Compound Element Formula Hydrogen H Hydrogen H Hydrogen H Hydrogen H Carbon C Compound #3 Electron Dot Diagram of Compound Element Number of valence electrons Electrons needed to form an octet? Electron Dot Diagram of Element Structure & Compound Formula Electron Dot Diagram of Compound Electrons needed to form an octet? Compound #4 Electron Structure Dot & Diagram Compound of Formula Element Electron Dot Diagram of Compound Chlorine Cl Chlorine Cl Chlorine Cl Chlorine Cl Carbon C Element Number of valence electrons Hydrogen H Hydrogen H Oxygen O Post Lab Analysis Questions: 1) Do covalent bonds tend to form with metals, nonmetals, or metalloids? Explain. 2) Does hydrogen follow the octet rule? Explain. 3) How does covalent bonding differ from ionic bonding? Explain.