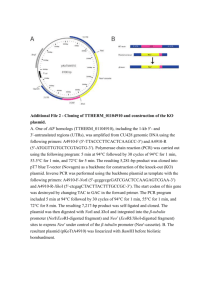
Online Only Appendix Manuscript DB-07-0092 Title: Long-Range Enhancers are Required to Maintain Expression of the Autoantigen IGRP in Adult Mouse Islets In Vivo Authors: Yingda Wang, Brian P. Flemming, Cyrus C. Martin, Shelley R. Allen, Jay Walters, James K. Oeser, John C. Hutton and Richard M. O’Brien Fusion Gene Plasmid Construction: (1) Generation of enhancer-IGRP-luciferase fusion genes. The construction of a human IGRP-chloramphenicol acetyltransferase (CAT) fusion gene containing promoter sequence from -324 to +3, relative to the transcription start site (TSS), in the pCAT(An) expression vector has previously been described (1). The CAT reporter gene was replaced with the more sensitive firefly luciferase reporter by re-isolating the IGRP promoter as a HinD III - Xho I fragment and ligating it into the pGL3-Mod vector (2). The putative IGRP enhancers A, B and C (Fig. 5) were synthesized by PCR using a previously described bacterial artificial chromosome (BAC) plasmid that contains the IGRP gene as the template (3). The putative IGRP enhancers E & F (Fig. 7) were synthesized by PCR using a second IGRPcontaining BAC plasmid (Accession number AC084429) as the template. The primers used incorporated HinD III restriction endonuclease sites (underlined): Enhancer A: 5'-CCCAAGCTT(-2477)CTCGAGGTAGCCTTAGCAAAGG-3' and 5'-CCCAAGCTT(-1787)CCATGGGCAATGACCTTGTCTC-3'; Enhancer B: 5'-CCCAAGCTT(-4766)GTGACCTAACACCTTAGCGCCACC-3' and 5'-CCCAAGCTT(-3691)CCAGTGATGGCCGATTAGACCATC-3'; Enhancer C: 5'-CCCAAGCTT(+3048)GTCAGCTTCTGAAGCATTAGCAAGAC-3' and 5'-CCCAAGCTT(+3831)GAGGATTCCTGACACTCAATACAGC-3'. Enhancer E: 5'-CCCAAGCTT(+8771)GTGCCCGGCTCTGCCACACACTCC-3' and 5'-CCCAAGCTT(+9372)AGGAAGGGACTCAACAGTCTTCC-3'. Enhancer F: 5'-CCCAAGCTT(+10622)AACCACAACTCATGAGAAAATTC-3' and 5'-CCCAAGCTT(+11022)CATTTATTCATCTCCCTGCCTTG-3'. The resulting PCR fragments were then digested with HinD III and ligated into the HinD III digested -324/+3 IGRP-luciferase fusion gene in either the correct or inverted orientation relative to their native orientation in the IGRP gene. IGRP enhancers C and F were also re-synthesized by PCR using the corresponding plasmids described above as the templates and primers that incorporated Sal I restriction endonuclease sites (underlined): Enhancer C: 5'-GCGTCGAC(+3048)GTCAGCTTCTGAAGCATTAGCAAGAC-3' and 5'-GCGTCGAC(+3831)GAGGATTCCTGACACTCAATACAGC-3'. Enhancer F: 5'- GCGTCGAC(+10622)AACCACAACTCATGAGAAAATTC-3' and 5'- GCGTCGAC(+11022)CATTTATTCATCTCCCTGCCTTG-3'. The resulting PCR fragments were then digested with Sal I and ligated into the Sal I digested 324/+3 IGRP-luciferase fusion gene in either the correct or inverted orientation relative to their native orientation in the IGRP gene. This places the enhancers 3' of the luciferase reporter gene (Figs. 5 & 7). Promoter fragments generated by PCR were completely sequenced to ensure the absence of polymerase errors. Plasmid constructs were purified by centrifugation through cesium chloride gradients (4). (2) Generation of IGRP-LacZ fusion genes. The construction of the -306/+3 IGRP-LacZ fusion gene has been previously described. To generate a -911/+3 IGRP-LacZ fusion gene this IGRP promoter region was amplified using PCR and ligated into a Sal I - HinD III digested, modified pCS2+ vector containing a LacZ reporter gene that encodes a cytoplasmic form of -galactosidase. The -911 IGRP-LacZ fusion gene was then isolated by digestion with Sal I and Not I for injection into pronuclei (see Research Design and Methods). The construction of a mouse IGRP-chloramphenicol acetyltransferase (CAT) fusion gene, containing promoter sequence from -911 to + 3, in the pCAT(An) expression vector, has been previously described (3). This plasmid was digested with BamH I and Bgl II to remove the IGRP promoter sequence between -911 and -508. A fragment of the IGRP gene promoter from -1342 to -508 was isolated from the pGEM-BAC 4.8 plasmid (3) as a BamH I - Bgl II fragment and subcloned into the BamH I - Bgl II digested -911 IGRP-CAT plasmid in the same orientation. The resulting plasmid contains IGRP promoter sequence from -1342 to +3, with a native IGRP Xba I restriction endonuclease site at -1342 (On-Line Fig. 1). We previously reported the isolation of a recombinant C plasmid, designated BAC IGRP-3, containing the entire IGRP gene (3). BAC IGRP-3 plasmid DNA was digested with BamH I and Xba I to isolate a 2566 bp fragment representing IGRP promoter sequence between -3911 and -1342 (On-Line Fig. 1). This fragment was ligated into the BamH I - Xba I digested -1342 IGRP-CAT plasmid to generate a plasmid containing IGRP promoter sequence from -3911 to +3 (On-Line Fig. 1). To generate a -3911/+3 IGRP-LacZ fusion gene the -911 to +3 IGRP promoter region was first isolated as a HinD III - Pst I fragment from -911 IGRP-CAT (3) and then ligated into the HinD III - Pst I digested pPD1.27 LacZ vector (5). The -3911 to -35 IGRP promoter region was then isolated from -3911 IGRP-CAT as a BamH I - Kpn I fragment and sub-cloned into BamH I - Kpn I digested pSP72 (Promega). The -3911 to -911 promoter region was then re-isolated as a HinD III fragment and ligated into HinD III digested -911 IGRP-LacZ in the correct orientation (On-Line Fig. 1). The -3911 IGRP-LacZ fusion gene was then isolated by digestion with Sph I and Not I for injection into pronuclei (see Research Design and Methods). The key restriction endonuclease recognition sites used in the construction of these fusion genes are shown in Online Only Appendix Figure 1. (3) Generation of an IGRP-BAC transgene by bacterial recombination. (i) Construction of the targeting vector. The pEGFP-IBGFTet plasmid, a generous gift from Doug Mortlock (Vanderbilt) was used for construction of the targeting vector. This plasmid contains, going 5' to 3', Nhe I and Pac I restriction endonuclease sites for introduction of recombination Arm A oligonucleotides, an enhanced green fluorescent protein (EGFP) cDNA, an internal ribosome entry site (IRES), a cDNA encoding a galactosidase-neomycin resistance fusion protein, a FRT (FLP recombination target) site, a tetracyclineresistance cassette (Tet), a second FRT site, and Spe I and Not I restriction endonuclease sites for introduction of recombination Arm B oligonucleotides (6). The Arm A oligonucleotides (Nhe I and Pac I compatible ends underlined): Sense strand 5'- CTAGC(+6664)TTTGCCCTCGGCTTTTACCTGCTTCTCCGACTGTTCGGTATTGACCTGCTG(+6 714)TTAAT-3' and Antisense strand 5'- TAA(+6714)CAGCAGGTCAATACCGAACAGTCGGAGAAGCAGGTAAAAGCCGAGGGCAAA(+ 6664)G-3' and Arm B oligonucleotides ( Spe I and Not I compatible ends underlined): Sense strand 5'- CTAGT(+6715)TGGTCCGTGCCCATCGCCAAAAAGTGGTGTGCCAACCCAGACTGGATCCAC( +6765)GC-3' and Antisense strand 5'- GGCCGC(+6765)GTGGATCCAGTCTGGGTTGGCACACCACTTTTTGGCGATGGGCACGGACC A(+6715)A-3' were introduced into the pEGFP-IBGFTet plasmid using standard methodology (4). The design of these oligonucleotides was such that recombination will insert the targeting vector into the IGRP locus in exon 5, at bp + 6715 relative to the TSS, 143 bp 3' of the exon 4/5 spice junction, such that the EGFP cDNA is in frame with the IGRP coding sequence but no IGRP sequence is removed (Fig. 6A). (ii) Introduction of a multi-component cassette into the IGRP locus by bacterial recombination. A BLAST search of the NCBI nr and database using the mouse IGRP cDNA sequence as the query identified a BAC clone (Accession number AC084429) derived from the RP23-291P1 library which contained the entire mouse IGRP gene as a contiguous sequence. This clone was purchased from BACPAC Resources, Children's Hospital Oakland Research Institute, Oakland, CA) and BAC DNA purified using the QIAGEN Large-Construct Kit (Valencia, CA) according to the manufacturer's instructions. SW105 recombination-deficient electrocompetent cells were prepared as previously described (7; 8). Purified BAC DNA (500-700 ng in 5 l water) was then mixed with 40l SW105 electrocompetent cells and electroporation performed as described (7; 8). SW105 cells containing the BAC plasmid were selected at 32˚C on LB agar plates containing 12.5 g / ml chloramphenicol. To confirm that the BAC plasmid was still intact following electroporation, BAC DNA was reisolated from individual colonies of SW105 cells using a mini-prep protocol described by BACPAC Resources (http://bacpac.chori.org/bacpacmini.htm). DNA was digested with BamH I or Nru I, and analyzed on 0.8% agarose gels or pulsed-field gels, respectively, to ascertain the presence of DNA fragments of the expected size. SW105 cells containing the intact BAC plasmid were then made recombination-competent, using heat shock to induce expression of the recombination machinery, and then electrocompetent, as previously described (7; 8). An 8422 bp fragment containing the targeting cassette (Fig. 6A) was isolated from the Arm ApEGFP-IBGFTet-Arm B plasmid by digestion with Nhe I and Not I, and gel purified twice using Freeze 'N Squeeze DNA Gel Extraction Spin Columns (Bio-Rad Laboratories, Hercules, CA). The purified fragment (800-1000 ng in 10 l water) was mixed with 40l of the SW105 recombination-competent and electrocompetent cells containing the BAC plasmid and electroporation performed as described (7; 8). Recombinants were selected on LB agar plates at 32˚C containing 12.5 g / ml chloramphenicol and 5 g / ml tetracycline. To confirm that recombination had occurred as expected, BAC DNA was re-isolated from individual colonies of SW105 cells using the BACPAC mini-prep protocol described above, digested with BamH I or Nru I, and analyzed on 0.8% agarose gels or pulsed-field gels, respectively, to ascertain the presence of DNA fragments of the expected size. DNA sequencing was subsequently performed to confirm that recombination had occurred at the Arm A and B junctions as expected. SW105 cells in which recombination had occurred as expected were then treated with Larabinose to induce expression of FLP recombinase so as to remove the Tet cassette, as described (7; 8). Cells were then plated on LB agar plates containing 12.5 g / ml chloramphenicol. Individual colonies were then re-plated in parallel on LB agar plates containing 12.5 g / ml chloramphenicol or containing 12.5 g / ml chloramphenicol and 5 g / ml tetracycline. To confirm that removal of the Tet cassette had occurred as expected, BAC DNA was re-isolated from individual tetracycline-sensitive colonies of SW105 cells using the BACPAC mini-prep protocol described above. DNA was then digested with BamH I or Nru I, and analyzed on 0.8% agarose gels or pulsed-field gels, respectively, to ascertain the presence of DNA fragments of the expected size. (iii) Isolation of the -5000 to +10051 region of the IGRP locus containing the multi-component targeting cassette by bacterial recombination. Transgenic mice containing the targeted IGRP locus could have been generated through microinjection of the modified BAC described above into the pronuclei of one-cell embryos (9). However, this BAC contains several other genes so we chose to generate transgenic animals containing only the region of the IGRP locus from -5000 to +10051. This region contains enhancers A, B, C and E in addition to the proximal promoter region, all exons and all introns (Fig. 4). Bacterial recombination was used to transfer this IGRP gene sequence, which also contains the targeting cassette, from the BAC plasmid to pGEM7 (Promega) (Fig. 6A). This fragment was designated the IGRP-BAC transgene. To achieve this recombination Arm Y, representing IGRP sequence between -5000 and -4766, and recombination Arm Z, representing IGRP sequence between +9815 and +10051, were synthesized and ligated into the pGEM7 vector (Fig. 6A). Recombination Arm Y was synthesized using PCR with BAC AC084429 as the template and primers that incorporated Xho I and Not I or HinD III restriction endonuclease sites (underlined): 5'-CCGCTCGAGGCGGCCGC(-5000)AGCCTTAGCTCAACCAGGAACC-3' and 5'-CCCAAGCTT(-4766)CTGAATTCATGTTACAGATGGC-3'. Recombination Arm Z was synthesized using PCR with BAC AC084429 as the template and primers that incorporated HinD III or Not I and BamH I restriction endonuclease sites (underlined): 5'-CCCAAGCTT(+9815)GCCTCTGCCTCCTAAGTGCTGG-3' and 5'-CGCGGATCCGCGGCCGC(+10051)CCAGTGATAACCTGCTATAGGC-3'. The resulting PCR fragments were then digested with Xho I and HinD III (Arm Y) or HinD III and BamH I (Arm Z). The Arm Y PCR fragment was ligated into Xho I and HinD III digested pGEM7. The resulting plasmid was then digested with HinD III and BamH I prior to ligation with Arm Z. The resulting Arm Y-Arm Z pGEM7 plasmid was digested with HinD III and gel purified twice using Freeze 'N Squeeze DNA Gel Extraction Spin Columns (Bio-Rad Laboratories, Hercules, CA). SW105 cells containing the IGRP-targeted, Tet-deleted BAC plasmid were made recombinationcompetent and electrocompetent as described (7; 8). The purified linearized Arm Y-Arm Z pGEM7 plasmid (800 ng - 1 g in 10 l water) was mixed with 40l of these cells and electroporation performed as described (7; 8). Recombinants were selected on LB agar plates containing 125 g / ml ampicillin. To confirm that recombination had occurred as expected, colonies were picked and DNA isolated by standard techniques (4). DNA was then digested with BamH I and analyzed on 0.8% agarose gels to ascertain the presence of DNA fragments of the expected size. DNA sequencing was used to confirm that recombination had occurred at the Arm Y and Z junctions as expected. To isolate the IGRP-BAC transgene 100 g of the pGEM7 plasmid was digested with Not I. A 0.8% pulse-field gel made using UltraPure Low Melting Point Agarose (Invitrogen; Hercules, CA) was used to separate the pGEM7 vector backbone from the IGRP-BAC transgene. The latter was cut from the gel and purified using GELase Agarose Gel-Digesting Preparation according to the manufacturer's instructions (Epicenter Inc., Madison, WI). The transgene was then precipitated with ethanol, dissolved in 6 ml TE (10 mM Tris pH 7.5, 1 mM EDTA) and dialyzed against 3 L TE for 48hr using a Slide-ALyzer (Pierce Biotechnology, Rockford, IL). The dialyzed fragment was precipitated with ethanol and the pellet washed three times with 70% ethanol before dissolving in 100 l injection buffer (10 mM Tris pH 7.5, 0.1 mM EDTA). A sample of the purified transgene fragment was then digested with HinD III and analyzed on 0.8% agarose gels to confirm the presence of DNA fragments of the expected size. Pronuclei injections were then performed using the intact IGRP-BAC fragment as described in (see Research Design and Methods). References 1. Martin CC, Bischof LJ, Bergman B, Hornbuckle LA, Hilliker C, Frigeri C, Wahl D, Svitek CA, Wong R, Goldman JK, Oeser JK, Lepretre F, Froguel P, O'Brien RM, Hutton JC: Cloning and Characterization of the Human and Rat Islet-Specific Glucose-6-Phosphatase Catalytic SubunitRelated Protein (IGRP) Genes. J Biol Chem 276:25197-25207, 2001 2. Martin CC, Oeser JK, Svitek CA, Hunter SI, Hutton JC, O'Brien RM: Identification and Characterization of a Human cDNA and Gene Encoding a Ubiquitously Expressed Glucose-6Phosphatase Catalytic Subunit-Related Protein. J Mol Endocrinol 29:205-222, 2002 3. Ebert DH, Bischof LJ, Streeper RS, Chapman SC, Svitek CA, Goldman JK, Mathews CE, Leiter EH, Hutton JC, O'Brien RM: Structure and promoter activity of an islet-specific glucose-6phosphatase catalytic subunit-related gene. Diabetes 48:543-551, 1999 4. Sambrook J, Fritsch, EF and Maniatis EF: Molecular Cloning: A Laboratory Manual. Plainview, NY, Cold Spring Harbor Laboratory Press, 1989 5. Fire A, Harrison SW, Dixon D: A modular set of lacZ fusion vectors for studying gene expression in Caenorhabditis elegans. Gene 93:189-198, 1990 6. Chandler KJ, Chandler RL, Broeckelmann EM, Hou Y, Southard-Smith EM, Mortlock DP: Relevance of BAC transgene copy number in mice: transgene copy number variation across multiple transgenic lines and correlations with transgene integrity and expression. Mamm Genome In press, 2007 7. Lee EC, Yu D, Martinez de Velasco J, Tessarollo L, Swing DA, Court DL, Jenkins NA, Copeland NG: A highly efficient Escherichia coli-based chromosome engineering system adapted for recombinogenic targeting and subcloning of BAC DNA. Genomics 73:56-65, 2001 8. Warming S, Costantino N, Court DL, Jenkins NA, Copeland NG: Simple and highly efficient BAC recombineering using galK selection. Nucleic Acids Res 33:e36, 2005 9. Chandler RL, Chandler KJ, McFarland KA, Mortlock DP: Bmp2 transcription in osteoblast progenitors is regulated by a distant 3' enhancer located 156.3 kilobases from the promoter. Mol Cell Biol 27:2934-2951, 2007 Online Only Appendix Figure Legend Online Only Appendix Figure 1. Diagramatic representation of key restriction enzyme sites in the mouse IGRP promoter and pPD1.27 vector.
 0
0
advertisement
Related documents
Download
advertisement
Add this document to collection(s)
You can add this document to your study collection(s)
Sign in Available only to authorized usersAdd this document to saved
You can add this document to your saved list
Sign in Available only to authorized users