How to Read the Periodic Table
advertisement

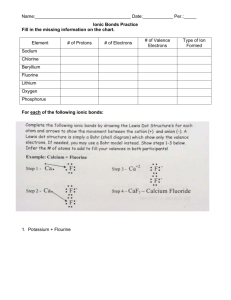
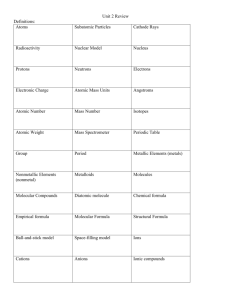
How to Read the Periodic Table The Periodic table is designed to help you predict what an element's physical and chemical properties are. You can also predict what elements will bond with each other. First, let's look at the columns and rows of the periodic table. Periodic Table Courtesy of Periodic Table of the Elements v. 4.0 by Kostas Tsigaridis (http://ptoe.move.to/) Groups or Families The vertical columns of the periodic table (there are 18) are called groups or families. Elements in the same group or family have similar but not identical characteristics. You will learn more about the 18 groups in a later section. You can know properties of a certain element by knowing which group it belongs to. Periods The horizontal rows of the periodic table are called periods. Elements in a period are not alike in properties. As a rule, the first element in a period is usually an active solid, and the last element in a period is always an inactive gas. Atomic size decreases from left to right across a period, but atomic mass increases from left to right across a period. Atoms on the left of the period, therefore, are usually larger and more lightweight than the smaller, heavier atoms on the right of the period. Think Inside the Box When you look at the periodic table, you should notice that each box represents a different element, and each box contains vital information about the element, including its name, symbol, atomic number, and atomic mass. Look at the sample box below for a description of each of these pieces of information. 6 C Carbon 12.011 The top number is the atomic number. Every element has its own unique atomic number. The atomic number tells how many protons are in one atom of that element. Since no two elements have the same atomic number, no two elements have the same number of protons. The large letter is the element's symbol, and just below that is the element's name. Each element has its own unique symbol and name. It is often very useful to memorize symbols and names for elements, especially the more commonly used elements. Below the name is the element's atomic mass. The atomic mass is the mass in atomic mass units for all possible isotopes of that element. The atomic mass essentially gives you an estimate of how massive one atom of that element is. Atoms of the element Chlorine have 17 protons in the nucleus and 17 electrons orbit the nucleus in the neutral atom. Chlorine atoms are never found singly in nature and only very rarely are they found sharing electrons with other elements. They are usually found with a negative charge and a full outer shell of electrons. common salt Sodium important in the production water, the pharmaceutical Chlorine gas is not encountered in nature as it is highly reactive. It is expensive manufacture. The element Chlorine is important both to life and commercially. As a negatively charged ion Chlorine makes up one half of the Chloride. Chlorine Gas, Cl2, is of plastics, treatment of drinking industry and cleaning products. and energy intensive to Isotopes of Chlorine There are 23 possible isotopes of Chlorine. All have 17 protons with the number of neutrons in the nucleus varying. Since the neutrons carry no charge they do not affect the number of electrons the isotopes have and so all the isotopes have the same chemical properties. The number of neutrons ranges from 11 to 34. This gives these isotopes a total mass of between 28 and 51 subatomic particles. Only two of these isotopes are stable. These are 35-Chlorine and 37-Chlorine which contain 18 and 20 neutrons respectively. The most common of these is 35-Chlorine which accounts for 75% of the Chlorine atoms in any natural sample. The Electron Structure of Chlorine The element Chlorine's 17 electrons are arranged into three shells of electrons. The first two shells are completely filled and the last shell has 7 electrons. We can obtain this by looking at the electron details of Chlorine which gives the result 1s2 2s2 2p6 3s2 3p5. This results in Chlorine having an identical outer shell electron pattern as Fluorine. This electron dot diagram is shown to the left. It tells us that Chlorine needs only one electron to fill its outer shell and achieve stability. The subshell detail of Chlorine's electron structure is shown below. The Chloride Ion Chlorine has one of the highest electronegativity values of any of the elements which means that its electrons are held very tightly to the nucleus. This makes Chlorine atoms prone to accepting a loose electron to complete its outer shell and therefore form the highly stable Chloride ion, Cl- as shown to the right. In this state it is quite nonreactive since it will not release any of its electrons to elements less electronegative than itself which are few and far between. Chloride ions will form compounds through Ionic Bonding with other elements. There is no sharing of electrons in these bonds, just attraction due to opposite charges. Sodium Chloride is an excellent example; it is made from Na+ ions and Cl- ions. Chlorine Gas It is possible to manufacture elemental or pure Chlorine gas from Chloride bearing solutions by supplying electricity. This requires a lot of electricity and the yellowish greenish Chlorine gas produced is very reactive and must be stored carefully and handled with caution. It holds the unenviable title as the first chemical weapon to be used in the First World War. This was initially deployed by the Germans against Allied troops, but was later used by both sides before being replaced by more lethal substances. Return from the Element Chlorine to Solar Chemistry 1 or return to the Green Planet home page for more Solar Power Facts. Share this page: What's This?</DIV< td> Enjoy this page? Please pay it forward. Here's how...