Supplementary Material - Proceedings of the Royal Society B
advertisement

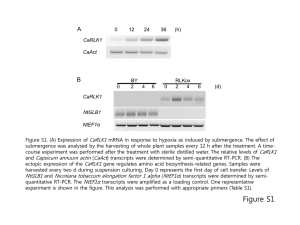
Supplementary information is linked to the online version of the paper. Methods ESEM, BSEI, and Catholuminescence. Samples were imaged at low magnification using a Nikon Optiphot Pol equipped with a Nixon Coolpix 4500 DX1 digital SLR (8 second exposure) and a Technosyn cold cathode cathodoluminiscope operated at 1 x 10-3 mbar vacuum with dry argon feed gas at 10 kV accelerating voltage and 450 A sample current. Higher power images were obtained using a Jeol f6400 scanning electron microscope equipped with a solid state 4-Quadrant Backscattered electron detector and PGT windowless EDS detector. The electron microscope was operated at 2 nA, and 20 kV at a working distance of 15 mm. XRD scans were completed on a Bruker D8; scans ran from 5 to 70o 2 with a step size of 0.02o and analysis time of 1 second per step. Beta-keratin sampled from the terminal ungual phalanx of a barnacle goose was used for peak and background comparison. Data analysis was completed using the Bruker EVA software. Electron microprobe analysis was performed using a Cameca SX100 with 5 wavelength dispersive spectrometers. Analytical conditions were 15 kV accelerating voltage and 100 nA beam current. FTIR absorption spectra and maps of the hadrosaur samples and pigeon down were recorded with a Biorad FTS 6000-IR which comprises an IR microscope, step-scanning spectrometer, and Sting-Ray focal plane array detector. The array is composed of a 64 by 64 detector grid (4096 pixels) with each pixel covering a spatial area approximately 7 m x 7 m. Entire field of view is 400 m2. Wavenumber range for this instrument is approximately 900 to 4,000 cm-1. Data presented here were collected using non-polarised light at a resolution of 16 cm-1. A supplementary FTIR reference spectrum of crocodile claw was taken using a Jasco 4100A (ATR) at a resolution of 4 cm-1. Amino Acid Analysis. Skin and terminal ungual phalanx sheath samples were analysed for D- and L- amino acids using a method developed by KAUFMAN and MANLEY (1998) and modified by Jones (2003). Samples (60-200 mg) were weighed in clean Pyrex culture tubes and 0.2 mL of 100 µM internal standard L-Homoarginine was added to each tube; Oxygen free nitrogen was then bubbled through the samples and a small amount of concentrated HCL (0.1mL) was added to destroy any carbonate present. When the gas bubbling had subsided, 0.8mL of 6M HCL was added to the culture tubes and nitrogen was bubbled through for (1min) prior to sealing with Teflon-faced screw-top lids. The sealed tubes were then hydrolysed at 110C for 6h, cooled, and subsequently freeze-dried. The freeze-dried hydrolysed amino acid samples were then resuspended in 1-4 mL of hplc grade water, centrifuged, and the supernatant was filtered (syringe filter, Cellulose nitrate, 0.45µm), prior to hplc analysis. The samples were analysed for amino acids using an Agilent 1100 Series high performance liquid chromatography system using a Thermo Electron Corporation BDS HyperClone BDS-C18 column (150 x 4.6 mm, 5 µ ,130 Å) and a fluorescence detector set with an excitation wavelength of 330 nm and an emission wavelength of 445 nm. Pre-column derivatisation of the sample (with O-phthaldialdehyde (OPA) and Iso-L-butyrylL-cysteine (IBLC) was performed using an automated derivatization and injector programme using Agilent Chemstation software. A gradient programme with a solvent flow of 0.5ml/min, with eluents: methanol (A), acetonitrile (B) and 23 mM sodium acetate buffer (C) was used as below). 2 min (A 5%, C 95%), 31 min (A 23%, B 0.4%, 76.6 %), 75min (A 50%, B 2%, C 48%), 85min (A 95%, B 5%) 105 min (A 5%, B 95%), 120 min (A5%, C 95%). 1 Amino acids were identified and quantified by comparison of their relative retention times and response factors with those of authentic standards. Full procedural blanks were also analysed with every batch of samples. Total organic carbon (TOC) and total nitrogen (TN) analyses were obtained using a Carlo Erba 1106 CHN analyser. Jones V., Characterisation of dissolved organic nitrogen compounds in aquatic samples. Ph.D. Thesis, The University of Liverpool (2003). Kaufman D.S. and Manley W.F. A new procedure for determining DL amino acid ratios in fossils using reverse phase liquid chromatography. Quaternary Geochronology, 17, 987-1000 (1998). Protein Analysis at Wolfson Molecular Imaging Center: Protein Separation and Analysis. Due to the dry nature of the sample it was possible to mechanically reduce the sample to dust within an Eppendorf tube using a sterilized metal implement. The powdered sample was transferred to another tube and 2 mL of an extraction solution was added. This solution was selected as the most efficient method of extracting keratins from solid material(1). The solution was prepared so that final concentrations were 5 M urea, 25 mM tris-urea and 5% 2-mercaptoethanol (Sigma Aldrich, Poole, UK) The mixture was left at 50˚C for up to two days. In order to remove low mass materials the solution was filtered using Zebra desalt spin columns (Pierce, Rockford, USA), the resulting solution was captured in another clean 1.5 mL tube and then analysed by gel electrophoresis. SDS PAGE gel and MALDI-MS The 1D protein separations were run on 4-20% TrisHCl precast gels (Bio-Rad, Hemel Hempstead, UK). 100 µL of sample were boiled with 100 µL of Laemmli buffer, and then 30 µL loaded onto the gel. Precision Plus Kaleidoscope protein standards (Bio-Rad) were used as mass markers. Two blanks were run with the samples, one containing earth material from the matrix surrounding the organic material, and another laboratory blank where a sterile plastic pipette tip was powdered under liquid nitrogen. Both were prepared as above for the organic sample. Following the electrophoresis the gel was stained using a silver staining kit (Invitrogen, Paisley, UK). After PAGE, gel bands were cut and analyzed via MALDI-MS (Kratos Axima CFR)following digestion with trypsin. When the proteins at ~12 kDa from the goose claw samples were analysed, the peptide mass fingerprints (PMFs) obtained correlated strongly within the online search engines with that of chicken beta-keratins. Skin and terminal ungual phalanx sheath samples from the dinosaur specimen were slowly heated in the extraction solution and an aliquot was further purified using size exclusion to isolate any proteins or peptides (>1000 Da), then mixed with the anionic detergent and analysed by PAGE. A separate aliquot was separated by PAGE without the initial purification by size exclusion step. Blank samples were prepared in the same way using the matrix component that was encasing (but not touching) the organic sample, to ensure no contamination was present within the original sample or introduced through out the analytical procedure. Independent trypsin digests of samples with and without the size exclusion step and/or PAGE were then analysed by MALDI-MS was also completed. Protein Analysis at Manchester Interdisciplinary Bioscience Center: Separate samples were extracted for protein following methods described in Asara et al. (2007). In brief, approximately 3 g material was manually ground using pestle and mortar, and demineralised with 0.5 M EDTA. The EDTA-insoluble residue was rinsed with ddH2O 2 and heated at 60oC in 4 M GuHCl, the supernatant removed and repeatedly heated in 4 M GuHCl three more times. The four supernatants were pooled, dialysed against distilled deionized H2O for 3 days, and lyophilized. The residue was then resuspended in 50 mM ammonium bicarbonate and an aliquot acidified to 0.1% trifluoroacetic acid, co-crystalised with α-cyano hydroxycinnamic acid and analysed by MALDI-MS for intact beta-keratin. A further aliquot was then digested with trypsin, following reduction and alkylation, and analysed by MALDI-MS (Brűker Ultraflex II) and nanoLC-ES-MS/MS (Dionex Ultimate 3000 coupled to a Waters QToF Ultima GLOBAL mas spectrometer). Peptides were bound to the C18 Peptide map column and underwent a slow gradient to 30% organic at 40 minutes followed by an increase up to 95% over 15 minutes. The organic component was then decreased to 0% at 60 minutes the column was reconditioned for a further 15 minutes before the next injection. The LC-MS/MS data was searched using the Mascot search algorithm to search the Swiss-Prot database, missed cleavages were set to 2 with peptide and tandem MS tolerances set to 100 ppm. Tandem MS data were manually interrogated to confirm peptide identification. An alternate approach to extract proteins from the fossil specimens was also carried out. Samples were briefly demineralised in 1 M HCl for 1 h, shown to be suitable for removing some recent contamination (i.e., mammal alpha keratins), and the HCl-soluble fraction removed and precipitated with four volumes of chilled acetone for 2 days. The precipitate was resuspended with heating at 60oC in 50 mM ammonium bicarbonate centrifuged, and the supernatant taken for digestion with trypsin. The HCl-insoluble fraction was then rinsed with ddH2O and further extracted using 0.5 M EDTA over 3 days. The EDTA-insoluble fraction was then heated at 60oC in 50 mM ammonium bicarbonate, centrifuged, and the supernatant taken for digestion with trypsin. Py-GCMS Analysis at Williamson Research Centre for Molecular Environmental Science, University of Manchester: Residues of the skin envelop and surrounding sedimentary matrix were analysed by normal flash pyrolysis gaschromatography mass spectrometry (Py-GCMS). Samples were pyrolysed using a CDS (Chemical Data Systems) ? 5200 series pyroprobe pyrolysis unit by heating at 650°C for 20 s to fragment macromolecular organic components. Fragments were analysed using an Agilent 7890A fitted with a HP-5 fused column (J+W Scientific; 5% diphenyl-dimethylpolyolsiloxane; 30m, 0.32 mm i.d., 0.25μm film thickness) coupled to an Agilent 5975C MSD single quadrupole MS operated in electron ionization (EI) mode (scanning a range of m/z 50-600 at 2.7 scans s1 ; ionisation energy 40 eV). The pyrolysis transfer line and injector temperatures were set at 350°C, the heated interface at 280°C, the EI source at 230°C and the MS quadrupole at 150°C. Helium was used as the carrier gas and the samples were introduced in split mode (split ratio 5:1). The oven was programmed from 40°C (held for 2 min) to 300°C at 10°C min-1 and held at this temperature for 25 min. Compounds were identified by comparison of spectra with those reported in the literature. Stable Isotope Analysis Samples were analysed for their carbon and oxygen stable isotopic composition at the University of Liverpool Stable Isotope Laboratory. Small volumes were reacted with anhydrous 100% orthophosphoric acid under high vacuum. Raw sample and standard 13C and 18O values were corrected following standard methods. Results are expressed as per mil deviation from the Pee Dee Belemnite (PDB) international standard. Sample reproducibilities were better than 0.1 per mil for both carbon and oxygen. 1. Nakamura A., Arimoto M., Takeuchi K., and Fujii T., A Rapid Extraction Procedure of Human Hair Proteins and Identification of Phosphorylated Species Biol. Pharm. Bull. 25, 3 569-572 (2002). Supplementary Figures Figure S1. Cathodoluminescence photomicrograph of the hadrosaur skin in thin section, showing grain sizes and a cellular structure identical to those imaged by electron microprobe. Figure S2. Electron Microprobe X-Ray fluorescence map of silicon distribution within the ossified tendon. The dark matrix is the mineral apatite. This image shows that a silica mineral coating has developed within the Haversian canals. Fig. S3. Longitudinal section of epaxial tendon showing a Haversian canal, A) showing the decrease in silica absorption defining the canal. B) Mapping carbon dioxide absorption shows that it is not uniformly lining the canal. (FTIR, reflection mode.) Fig. S4. Total reflectance map of a single canal in cross-section, showing the high reflectance zone with trapped carbon dioxide rimming the canal and two concentric zones within the canal. (FTIR, reflection mode.) 4