Protein measurement on graphite electrodes (Bicinchoninic acid
advertisement

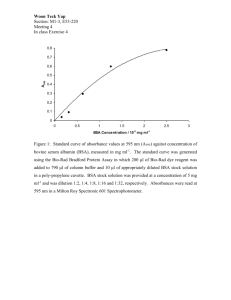
Richter, Hanno 1-3 Protein Measurement on Graphite Electrodes (Bicinchoninic acid method) Reagents: 1. 10 % SDS solution (10 g SDS / 100 mL H2O) (don’t autoclave), (Ambion #9822) 2. Mg/Ca mix: a. 3 g MgSO4 x 7 H2O b. 0.1 g CaCl2 x H2O c. Adjust volume to 1 L d. Autoclave, not necessary to be anaerobic 3. Isotonic wash buffer: a. 800 mL dH2O b. 4.19 g MOPS c. 0.6 g NaH2PO4 d. 0.1 g KCl e. 5.0 g NaCl f. 10 mL Mg/Ca mix g. Bring to pH 7.0 with 2 N NaOH h. Adjust volume to 1 L i. Autoclave, not necessary to be anaerobic 4. Blunt P-1000 tips a. To prevent lysis during resuspension of the cells in wash buffer 5. Mixed reagent for protein measurement: a. 50 mL Bicinchoninic acid b. 1 mL Cu(II) sulfate solution c. Use 2 mL per sample 6. Ampule of BSA standard a. 1.0 mg/mL b. 0.5 N NaOH Sample preparation: 1. Scrap the graphite electrode for protein and DNA: a. Weigh an empty 50 mL Falcon tube if extracting DNA (w1). b. Scrap the graphite electrode i. Use a sterile DNA free scraping dish and razor ii. Use 20 mL washing buffer to rinse electrode and razor during scraping iii. Scrap all sides of the electrode iv. Flush the razor blade repeatedly throughout c. Collect the cells in the wash buffer d. Transfer all 20 mL into Falcon tube e. Wash scraping dish again with 5-10 mL washing buffer f. Add this to Falcon tube g. Weigh tube again if extracting DNA (w2) h. Take a 0.5 mL sample for DNA-extraction i. Weigh tube once more if extracting DNA (w3) Richter, Hanno 2-3 j. k. l. m. n. Centrifuge tube at 4000g for 15 minutes If supernatant is clear, discard it If supernatant is not clear, centrifuge tube again at 4000g for 15 minutes Freeze tube with pellet Lyophilize the tube i. Use a needle to make holes into the blue cap 2. Measure the protein concentration: a. Add 3 mL 5% SDS to the Lyophilisate. i. The volume can be larger (10 mL) for flow through potentiostats which produce high current. b. Vortex to resuspend c. Spin everything down in the centrifuge for only a few seconds i. Cells must be in solution. ii. Homogenate should still look homogenous d. Sonicate on setting No. 3 for 1 minute or setting No. 4 for 2 minutes i. The tip shouldn’t touch the side or bottom of the tube e. Clean the tip with a paper towel afterward. f. Incubate lysates for 5 minutes in the steamer. i. Close valve at the bottom ii. Turn water on iii. Fill with water up to the line of the stick iv. Close hood v. Turn on the steam vi. Wait 5 minutes vii. Turn off the steam viii. Open the lid ix. Insert the sample x. Close the lid xi. Turn on the steam xii. Wait 5 minutes xiii. Turn off the steam xiv. Retrieve the sample xv. Turn off steamer xvi. Drain the water. g. Get the protein standard (BSA, 1 mg/mL) out of the 4oC fridge. h. Crack an ampule. i. Add BSA to 4 mL Cuvettes for standards: i. 0 µL 1.0 mg/mL BSA ii. 20 µL 1.0 mg/mL BSA iii. 40 µL 1.0 mg/mL BSA iv. 60 µL 1.0 mg/mL BSA v. 80 µL 1.0 mg/mL BSA vi. 100 µL 1.0 mg/mL BSA j. For each cuvette complete the volume with Milli-Q water to a total of 100 µL. k. Make sample dilutions: Richter, Hanno 3-3 l. 5 replicates per dilution m. Pipette sample into 4 mL cuvettes i. Dilution series example: 1. 100 µL 2. 50 µL 3. 25 µL 4. 10 µL ii. Notes for Geobacter cells: 1. For current up to 0.8 mA no dilution is needed 2. For current above 0.8 mA dilute by about how many times greater the current was above the base level 0.8 mA a. e.g. 1:10 for 8 mA. n. For each cuvette complete the volume with Milli-Q water to a total of 100 µL. o. Add 2 mL of mixed reagent to each sample p. Incubate for 30 minutes in the 37oC room q. Filter samples (HPLC filters) into the 1.5 mL cuvettes r. Read the samples in the spectrophotometer at 562 nm i. Std.-curve Test nameProteinCRa ii. Edit standards iii. Clear measurements iv. Put standard cuvettes in v. Measure the standards vi. Press enter vii. Measure samples. 3. Calculate the protein concentration in mg/mL a. Total amount of protein = Protien concentration (mg/mL) * dilution factor * Volume SDS (mL) * (w2 – w1) / (w3 – w1)