HEP_25828_sm_SuppInfo
advertisement

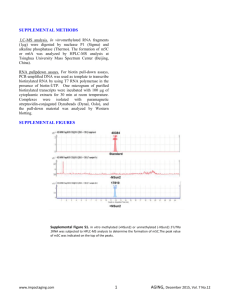
Supplementary Figure 1: HuR silencing attenuated liver fibrosis in BDL-mice. (A) qPCR of HuR mRNA levels, (B) Western blot (WB) of cytoplasmic HuR levels and (C) immunohistochemistry showing HuR expression in Sh control and Sh HuR in hepatocytes (arrows) and HSC (arrowheads) from BDL mice. (D) Serum ALT and (E) bilirubin levels. For OxiblotTM, 5 g of total protein sample were subjected to derivatization reaction in duplicate, following manufacturer instructions (OxiblotTM Protein Oxidation Detection Kit, Millipore). After protein determination, 20 g of total protein sample was run on a poliacrylamide gel and (F) coomassie staining performed to ensure equal loading prior to the derivatization of proteins. (G) Ponceau staining of the nitrocellulose membranes was performed to ensure equal loading of the derivatized proteins. Negative control (NC) of protein sample without derivatization was carried out to ensure specific binding to oxidized proteins. Supplementary Figure 2: Role of HuR in PDGF-induced responses. (A) HuR immunolabelling was done as described before (7) in primary HSC from BDL mice, transfected with control or HuR Sh RNA. (B) Activated HSC (cell line CFSC-8B), transfected with control or HuR Sh RNA, were incubated during the indicated times with or without PDGF (20 ng/ml), and protein extracts analyzed by WB with the indicated antibodies. (C,D) Cell scratch assay and (E) BrDU incorporation of control- or HuR-silenced primary HSC from BDL mice, treated with PDGF. Supplementary Figure 3: PDGF-induced response is mediated by HuR. (A) WB of nuclear protein extracts showing that ERK and PI3K inhibition prevents PDGFinduced nuclear translocation of p65. (C) Densitometry analysis of the effect of ERK and PI3K inhibition on PDGF-induced p65 nuclear translocation. (B) WB of total protein extracts showing that NFB inhibitor, BAY117802 prevents PDGF-induced increase in HuR expression. (D) Densitometry analysis of the effect of inhibition of p65 nuclear translocation (BAY117802) on PDGF-induced HuR protein levels. (E) Densitometry analysis of the effect of ERK inhibition and PI3K inhibition on PDGF-induced phosphorylation of pERK, pAKT, pLKB1 and HuR cytoplasmic localization. (F) Densitometry analysis of the WB showing V5 expression in cytoplasmic extracts of PDGF-treated CFSC-8B cells transfected with plasmids containing wild type or mutant HuR. (G) Primary HSC from BDL mice were plated during 16h, and then incubated with PDGF (20 ng/ml) or TGF- (8 ng/ml) during 1 hour. WB and densitometry analysis of HuR expression in cytoplasmic and nuclear extracts, and of pLKB1 (Ser428) expression in cytoplasmic extracts of PDGF or TGF--treated cells. Supplementary Figure 4: Role of LKB1 in ERK mediated-PDGF-induced HuR cytoplasmic translocation. (A) HSC (CFSC-8B), transfected with control or LKB1 Sh RNA, were incubated during the 30 minutes with or without PDGF (20 ng/ml), and cytoplasmic extracts analyzed by WB with the indicated antibodies. (B) Densitometry analysis of the effect of LKB1 knockdown on PDGFinduced HuR cytoplasmic localization. (C) HSC (CFSC-8B), transfected with control or LKB1 Sh RNA, were incubated during 24 hours with or without PDGF (20 ng/ml), and cytoplasmic extracts analyzed by WB with the indicated antibodies. (D) Densitometry analysis of the effect of LKB1 knockdown on PDGF-induced cyclin D1 protein expression. Supplementary Figure 5: Role of AMPK in LKB1 mediated-PDGF-induced HuR cytoplasmic translocation. (A) WB showing that PDGF stimulation induces LKB1 phosphorylation but not AMPK phosphorylation (Thr172) in HSC (CFSC-8B). (B) AMPK silencing does not reduce PDGFinduced HuR translocation. Supplementary Figure 6: Role of LKB1 in PDGF-induced response. (A) Primary HSC from BDL mice were transfected with control or LKB1 Sh RNA, and pLKB1 immunolabelling was done as described before (9). (B, C) Cell scratch assay and (D) BrDU incorporation of control- or LKB1-silenced primary HSC (from BDL mice), treated with PDGF. (E) IHC showing expression of pLKB1 (Ser428) (DAB+ cells, brown, arrowheads) in activated HSC (-SMA+ cells, alkaline phosphatase, red) in liver sections from BDL-mice and CCl4treated rats. Supplementary Figure 7: TGF--induced fibrogenesis response is mediated by HuR. (A) BrDU incorporation of control- or HuR-silenced primary HSC (from BDL mice), treated with TGF- (8 ng/ml). Time course of TGF--regulated expression of (B) Cyclin D1, Cyclin B1, and (C) p21 mRNA levels in activated HSC (CFSC-8B). (D) qPCR analysis of HuR mRNA expression and (E) WB analysis of HuR protein expression in activated HSC (CFSC-8B) 24h after TGF- stimulation (8 ng/ml). (F) Densitometry analysis of HuR protein in cytoplasmic and nuclear fractions after TGF- stimulation in activated HSC (CFSC-8B). Supplementary Table I: Specifications and conditions of the antibodies used. Supplementary Table II: Sequences of the primers used. SUPPLEMENTAL MATERIAL AND METHODS RNA isolation and real-time PCR (qPCR). For RNA isolation, total RNA was isolated using Trizol (Invitrogen, UK), 1 µg of total RNA was treated with DNAse (Promega, UK) and reverse transcribed into cDNA using Superscript III (Invitrogen, UK). qPCR was performed using Platinum® SYBR® Green qPCR SuperMix-UDG (Invitrogen, UK) using the LightCycler ® 2.0 Real-Time PCR System (Roche, UK). PCR was performed with primers described in Supplementary Table I. All reactions were performed in triplicate, and expression levels were normalised to the average level of 18s rRNA in each sample. Sub-cloning of wild type HuR and HuR mutants. The full-length cDNA of wild-type mouse HuR was purchased from (RZPD Deutsches Ressourcenzentrum für Genomforschung GmbH). The HuR-V5 (WT) was constructed by PCR using the 5´oligonucleotide containing the V5 tag sequence and subcloned into pCDNA 3.3 TOPO vector (Invitrogen). The HuR-V5 phospho mutants were constructed by using the QuickChange kit for site-directed mutagenesis (Stratagene) and as template the pCDNA-V5-HuR plasmid was used. Cell transfection. HSC (CFSC-8B) were transfected with 1 g of HuR-V5 phospho mutants or 100 nM si RNA of AMPK (Quiagen) using Lipofectamine 2000 (Invitrogen). Silencing mix was left overnight and afterwards medium was replaced with fresh 10%-FBS DMEM. Immunohistochemistry. Paraffin sections of formalin-fixed liver samples were rehydrated and subjected to antigen retrieval in 10 mM sodium citrate buffer (pH 6.0). The sections were incubated in 0.2% Triton-X in PBS containing 1% BSA and 10% horse serum for 1 h, followed by avidin-biotin block. To analyze hepatocyte proliferation or macrophage area, the sections were incubated with Ki67 (1:25, DAKO M7249) or F4/80 antibodies (AbD Serotec MCA497B) respectively, overnight at 4ºC. Next day, sections were incubated with relevant secondary antibodies for 30 minutes and color developed with Vector VIP substrate (Vector labs). Sections were then dehydrated, cleared and mounted in a permanent medium (DPX, Fluka). 5 fields (×20) of each section were analyzed using ImageJ software to identify Ki67 or F4/80positive area on each section. For double immunolabelling of -SMA with HuR or pLKB1 (Ser428) expression, the sections were incubated for 1 hr with -SMA conjugated with Alkaline Phosphatase (Sigma) and colour developed with Vector Red substrate (Vector labs). The sections were then incubated with either HuR or pLKB1 (Ser428) antibodies overnight at at 4°C, incubated with their respective secondary antibodies (Envision, Dako) and color developed with DAB (Dako). Sections were counterstained with Mayer´s Haematoxylin and then dehydrated, cleared and mounted in a permanent medium (DPX, Fluka). Pictures at 20, 40 and 100 times of magnification were taken. Statistical analysis. All data are presented as mean ± standard error of the mean (SEM). Statistical significance was estimated by Student’s t-test.