Experiment 6 Chromatography Handout
advertisement

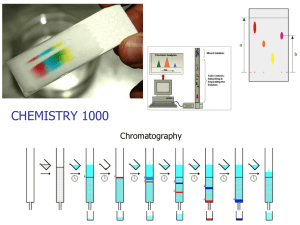
Experiment 6: Chromatography Laboratory Handout Purpose: In this experiment you will explore the technique of chromatography, a very important method for the separation and identification of substances. This technique is widely used for evidence analysis dealing in forensic science. Background: Chromatography separates the components of a mixture based on polarity. The principle method, by which chemists separate compounds within a mixture, was invented a century ago by the Russian scientist Mikahil Tswett to separate colored plant pigments. The name chromatography is derived from the Greek word for color, chromatos. Methodology: Different types of chromatography have similar principle, containing a mobile phase and a stationary phase that differ in their relative polarities. The sample mixture to be separated is dissolved in the mobile phase and then applied to the stationary phase. As the components of the mixture travel through the stationary phase, they interact differently with the two phases. The more polar components will interact more with the polar phase because “like dissolves like”. Definitions: Chromatography – method by which chemists separate compounds within a mixture. Polarity – the behavior of electrons created by forming chemical bonds by a molecule. One atom will have a stronger tendency to withdraw electrons from the other atom. The greater the electron withdrawing atom is the more polar the molecule. Nonpolar – the electrons are exactly shared between atoms because there is no tendency to withdraw electrons from each other. Like dissolves Like – in order for compounds to mix, they must have the same polarity. Water is a polar molecule but oil is nonpolar and they do not mix. However, HCl in water will mix because they are both polar and octane in oil will mix because they are both nonpolar. Stationary Phase – separates the analyte (or substances) to be measured from other molecule in the mixture and allows it to be isolated. Mobile Phase – is the part of the chromatographic system which carries the solutes through the stationary phase.