10b Harvesting Chemical Energy-student
advertisement

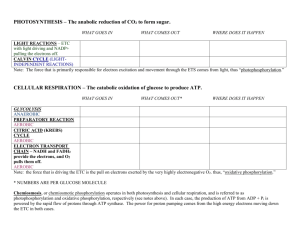
Harvesting Chemical Energy As open systems, cells require _________ energy sources to perform cellular work. Only photosynthetic organisms have the ability to ______________ the energy from the sun. During Photosynthesis: ________ and _______ are the raw materials used to make __________ _______________ energy is converted into _____________________ energy __________________________ energy can be released to drive _____________ reactions by cellular respiration. Note: the chemical elements are recycled, but the energy is not! The storage and release of Free Energy The free energy is __________ and ______________ to ATP (adenosine triphosphate). Recall, the _______________ bonds have stored energy; ____________ these bonds releases energy (to drive reactions); while ____________ new phosphate bonds temporarily stores the chemical energy. The compound receiving the phosphate group from ATP is said to be ______________________ and becomes more reactive in the process. The ________________________ compound loses its newly acquired ______________ group as work is performed. There are two types of phosphorylation: 1. ____________________________ phosphorylation an enzyme catalyzed reaction of Pi to ADP 2. ____________________ phosphorylation forming of ATP directly through a series of enzyme catalyzed _________ reactions where oxygen is the ____________________________ (more about this in a bit). REDOX Reactions Also Release Energy _____________________________________ reactions = chemical reactions which involve a partial or complete transfer of electrons from one reactant to another; called «____________________________ » for short. 1. Oxidation = ____________________________________________________________ 2. Reduction = ____________________________________________________________ “LEO says GER” Loss of electrons = oxidation Gain of electrons = reduction The electron transfers require both a ___________ (becomes oxidized) and an _____________________ (becomes reduced). Since electrons lose ___________________________ when they shift towards _________ electronegative atoms, redox reactions that move electrons closer to elements with a higher electronegativity (like oxygen) _______________ energy. Cellular Respiration is a ___________ process that (ultimately) transfers _____________________, including electrons with high potential energy, from sugar (glucose) to _____________________. Released energy is used to convert ADP to ATP. How is the energy transferred to a molecule of ATP? Essentially, glucose is broken down in a series of sequential steps so that the ____________________ released from ________________ bonds can be harnessed (captured) bit by bit. Eventually this harnessed free energy is ______________________ to molecules of ATP as they are phosphorylated (i.e from ADP + Pi ATP) Why does the process need to occur in a sequence of steps? Activation energy = requires extreme heating to start combustion of glucose Exergonic reation = large amount of energy is released; would damage cells Therefore, the combustion of glucose occurs by a series of _______________________________ steps, that result in: At each step of the way, __________________ (along with electrons) are transferred to another substance (enzymes & coenzymes) and eventually a final acceptor (oxygen) When this happens, a _____________of the reaction’s total energy is released and stored in ATP (ie. ATP is produced). An Overview of Cellular Respiration There are two types: ___________________ respiration (fermentation) = ATP-producing pathway in which sugars are only partially degraded (low energy yield); proton donors and acceptors are organic molecules. __________________ respiration = ATP-producing pathway in which sugar is fully oxidized (high energy yield); ultimate electron acceptor is oxygen (inorganic molecule) Overall Equation: (Aerobic) Aerobic Cellular Respiration occurs in four stages: 1. 2. 3. 4. Glycolysis pyruvate oxidation (occurs only in eukaryotic cells) Citric Acid or Kreb’s Cycle Electron Transport Chain (ETC) Glucose + oxygen carbon dioxide + water + energy C6H12O6 + 6O2 6CO2 + 6H2O + 38 ATP (total) Breaking down glucose to get H+ (and electrons) Using H+ (and electrons) by releasing them slowly trapping released energy in ATP Stepwise fall of Electrons occurs due to special electron acceptors (______ and ______) and the ______________________________________. _______________ (and electrons) stripped from glucose are NOT transferred directly to oxygen, but are first passed to a special electron acceptor ex. NAD+ NAD+ (nicotinamide adenine dinulceotide) and FAD (flavin adenine dinucleotide = are dinucleotides that function as a coenzyme in the redox reactions of metabolism. NAD+ and FAD are found in all cells assist enzymes in electron transfer during redox reactions What happens? During the ____________________ of glucose, the electrons are ________________ and temporarily trapped in NAD+ and FAD. This transferred is catalyzed by enzymes called ______________________, which: o remove a pair of hydrogen atoms (2 electrons and 2 protons) from glucose (substrate) o deliver the two electrons (______) and one proton (______) to NAD+ (or 2eand 2H+ to ________) o ____________ the remaining proton (H+) into the surrounding solution. Overall reactions: The high energy electrons transferred to NAD+ and FAD are then passed down the _______________________________ to oxygen, powering the production of ATP by oxidative phosphorylation. Electron Transport Chains Consist of ____________________________ molecules built into cellular membranes _____________ energy rich electrons from reduced coenzymes (NADH and FADH2) Since electrons lose potential energy when they are transferred to a more electronegative atom, this series of reactions _______________ energy. ______________ energy from energy-rich electrons in a controlled stepwise fashion powering the production of ATP Each successive carrier in the chain has a ____________ electronegative then the carrier before, so the electrons are pulled downhill towards ________________ (the molecule with the ______________ electronegativity), and the final electron ______________.