Key_Heat_and_Thermal_Energy_Review_Quiz
advertisement

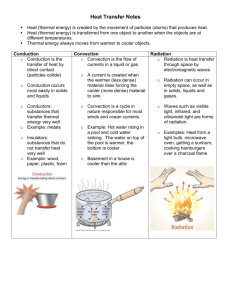
Heat and Thermal Energy Review Quiz Key 1. In order to have a transfer of thermal energy between two objects in contact, they must have different __temperatures___. 2. Temperature is defined as the (average / total) kinetic energy of the particles. 3. The transfer of thermal energy is called __heat__. 4. If the temperature of a substance rises by 10o C then it must have risen by (more than / less than / exactly) 10 K and (more than / less than / exactly) 10 oF. 5. Convert the following temperatures: a. 311 K = __100.4_oF b. 63 oF = __17.2__ oC c. 37oC = _310___ K 6. Two steel objects each have 1000 J of thermal energy in them. If object A is at a higher temperature than object B then object A must have (more / less / the same) mass as compared to object B. 7. Water has a (high / low) specific heat and therefore changes temperature (quickly / slowly). Gold has a (high / low) specific heat and therefore changes temperatures (quickly / slowly). Based on this, if you burn your hand, it is better to put it in contact with 1000 g of (water / gold) at room temperature. 8. If objects A and B are only in contact with one another and object A loses 100 J of thermal energy then object B gains (more than / less than / exactly) 100 J of thermal energy. 9. At the boiling point, what phase or phases of matter exist? _liquid and gas___ 10. At the freezing point, what phase or phases of matter exist? __solid and liquid____ 11. Lead has a specific heat of 130 J / kg oC and aluminum has a specific heat of 903 J / kg oC. If the same amount of thermal energy is added to both, the (aluminum / lead) will have a larger increase in temperature. 12. Heat from the sun travels through the vacuum of space by (conduction / convection / radiation). 13. The classroom is heated by (conduction / convection / radiation). 14. A pot on the stove gets hot due to (conduction / convection / radiation). 15. Thermal energy always moves from (high to low / low to high) temperatures. 16. As a substance changes phase the temperature (increases / decreases / remains the same). 17. As the temperature of a substance increases, the speed of the molecules (increases / decreases / remains the same), and the kinetic energy of the molecules (increases / decreases / remains the same). 18. The temperature of a substance only changes when it is in (one / two) phase(s). 19. As the entropy of a substance increases, the substance has the ability to do (a larger / a smaller / the same) amount of work. 20. Two specimens of water are mixed together. If the initial temperature of one is 40 oC and the other is 90 o C then the final temperature must be (greater than 90 oC / less than 40 oC / between 40 oC and 90 oC) 21. If equal amounts of water at 0 oC and 80 oC are mixed together then the equilibrium temperature will be _40___ oC. 22. A (solid / liquid / gas) has a definite shape and volume, on the other hand, a (solid / liquid / gas) has a definite volume but assumes the shape of the container. 23. When an object is heated, the vibration of the molecules (increases / decreases / remain the same). 24. The transfer of energy that doesn’t involve matter moving is (conduction / convection / radiation), the transfer of energy that involves particles bumping into each other is (conduction / convection / radiation), and the transfer of energy through the movement of fluids is called (conduction / convection / radiation). 25. Changing a solid into a liquid is called (melting / freezing / condensing / boiling), changing a liquid into a gas is called (melting / freezing / condensing / boiling), changing a gas into a liquid is called (melting / freezing / condensing / boiling), and changing a liquid into a solid is called (melting / freezing / condensing / boiling). 26. When dry ice changes from a solid directly to a gas without going through the liquid phase, this process is called _sublimation___. 27. At thermal _equilibrium___, the temperature of the objects in contact is the same and there (is / is not) a flow of thermal energy between them. 28. If 1500 J of thermal energy raises the temperature of 2 kg by 5oC, the specific heat of the substance is __150____ J/kg oC. 29. If 200 g of brass (specific heat 376 J / kg oC) at 75 oC is placed into 100 g of water (specific heat 4180 J / kg oC) at 0 oC, the equilibrium temperature is __11.4___ oC. 30. If 300 g of an unknown substance at 50 oC is placed into 100 g of water (specific heat 4180 J / kg oC) at 100 oC and they reach an equilibrium temperature of 90 oC. The specific heat of the unknown substance is _348.3__ J / kg oC. 31. Five hundred grams of lead are raised 100 m and dropped, if the specific heat of lead is 130 J / kg oC and all of the potential energy is converted into thermal energy then the lead will increase in temperature by _7.54__ oC. 32. The boiling point for isopropyl alcohol is _80__ oC. 33. The melting point for isopropyl alcohol is _-90__ oC. 34. At - 20 oC, isopropyl alcohol is a (solid / liquid / gas). 35. At – 120 oC, isopropyl alcohol is a (solid / liquid / gas). 36. At what temperature can isopropyl alcohol be either a liquid or a gas? __80 oC___. 37. At what temperature can isopropyl alcohol be either a solid or a liquid? __-90 oC___.