Australian Curriculum - Year 8 Program
advertisement

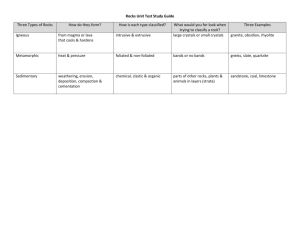
Organs and Systems (ACSSU150) Cells (ACSSU149) Biological Sciences Week Teaching Plan 1 Animal Cells – See Plant and Animal Cells Discuss as a class what makes up their body and what they know. Introduce the concept of cells giving an idea of size. Show students a picture of a labelled animal cell. Discuss the names and functions of the different structures and organelles. Compare the labelled diagrams to micrographs of real cells and have students identify the structures. 2 Plant Cells – See Plant and Animal Cells Compare the structure of animal cells, plant cells and fungal cells. Examine the function of cell structures and organelles that are unique to plant cells. Worksheet: Students label blank diagrams of plant and animal cells with the names and functions of the different components. 3 Viewing Cells – See Using a Microscope, Plant and Animal Cells. Review the parts of a microscope LO L7572: What can you see under the microscope? (Scootle Link) Practice preparing slides and viewing small objects under a microscope. Prac: Examine onion cells under a microscope and create a labelled scientific diagram of the cells. 4 Single Celled Organisms – See Disease – Bacteria Discuss single celled organisms and examine different types. Look at the importance of singled celled organisms as decomposers and how they cause disease. Examine what processes a cell must perform to be classified as an organism and look at how single celled organisms perform these functions – excluding reproduction 5 Mitosis Explain the reproduction of single celled organisms via mitosis and define asexual reproduction. Use a labelled flow chart to explain the stages of mitosis and explain that mitosis is also the process for growth and repair in multi-cellular organisms. ICT: Use a video to show the process. (Examples 1, 2) 6 Specialized Cells – See Cell Functions Discuss how cells make up multi-cellular organisms – defining tissues, organs and systems. Identify systems in the human body and identify the tissues and organs that make up each system. Examine the structure of specialized cells in familiar tissues such as bone cells, muscle cells and nerve cells. Research Project: Students produce a report on stem cells discussing what is special about stem cells and why there is controversy surrounding stem cell research. 7 The Musculoskeletal System – See Muscular System, Skeletal System I, Skeletal System II. Discuss the importance of bones and muscles and how they work together in the musculoskeletal system. Prac: Dissect a chicken wing and create a labelled scientific diagram. Compare endo- and exoskeletons. 8 The Digestive System – See Digestive System Examine the organs involved in the human digestive system and discuss the role of each component. LO L719: Body Parts – Digestive System (Scootle Link) 9 The Respiratory System – See Respiratory System Examine the organs involved in the human respiratory system. LO R11869: Assembling the respiratory system (Scootle Link) Research Activity: Students prepare a report or table to compare the lungs and hearts (or their equivalent) in mammals, birds, fish, amphibians and insects. Assessment 10 The Reproductive System – See Animal Reproduction Examine the organs involved in the male and female human reproduction systems. Examine the development of a child from fertilization to birth. ICT: Each student researches the gestation period of an animal and the average number of offspring. Compile the numbers as a class and have each student create a scatter plot using Excel and comment on any trends. Physical and Chemical Change (ACSSU225) The Particle Model (ACSSU151), (ACSSU152) Chemical Science Week Teaching Plan 1 The Particle Model – See States of Matter and the Particle Model Brainstorm different types of model and define a ‘scientific model’. Introduce the Particle Model as an example of a scientific model. Discuss particle models of solids liquids and gases and how the particle model explains certain properties of solids liquids and gases. (Video) 2 Changes of State – See States of Matter and the Particle Model Explain how heat affects the particles in the particle model and therefore how the particle model can be used to explain various changes of state. LO L3254: What the world is made of: particles, heat and movement (Scootle Link) Class Activity: Have students use themselves to simulate the particles in a solid, liquid and gas then simulate the different changes of state. 3 Elements – See Elements, Compounds and Mixtures Explain that the particle model cannot explain the chemical properties of substances. For this we need a more sophisticated model – Dalton’s Atomic Theory of Matter Review the assumptions of the Atomic Theory of Matter and define the elements and compounds. Examine elements in detail including chemical symbols and the periodic table Homework: Students come up with a mnemonic to help them remember the first 10 elements in the periodic table and read it to the class. 4 Compounds – See Elements, Compounds and Mixtures Examine the concept of a compound in detail. Compare and contrast molecular and lattice compounds. Explain the chemical formulae of compounds and discuss the meaning of the subscript numbers in the formulae for molecular and lattice compounds. Hands on Activity: Construct 3D models of compounds and relate these models to the chemical formulae. 5 Mixtures – See Elements, Compounds and Mixtures Contrast pure elements and compounds with mixtures. LO L5822: Types of matter: pure substances and mixtures (Scootle Link) Worksheet: Students are given table that classifies different types of mixtures according to whether the solvent and solute are solid, liquid or gas. Students must fill in the table with examples of each. 6 Physical Change – See Physical and Chemical Change Contrast physical and chemical changes. Have students identify physical and chemical changes. Revise the particle model for changes of state then use the particle model to describe other physical changes such as expansion and contraction, mixing, dissolving and diffusion. 7 8 Chemical Change – See Physical and Chemical Change Discuss what chemical change means and the evidence that a chemical change has taken place. Prac: Allow students to perform simple chemical reactions in test tubes that demonstrate the different signs of a chemical. Chemical Reactions Use the Atomic Theory of Matter to explain what is happening to the atoms in during a chemical reaction. Define reactants, products and the Law of Conservation of Mass. Have students simulate simple chemical reactions using 3D models. Only use reactions whose formula equations are balanced without the need for any coefficients (Examples). Have students write the word equation for each reaction and help students to write the formula equation. Homework: Provide students with a list of word equations with space to write in the formula equation underneath. Students should research the chemical formulae of the reactants and products to construct the unbalanced formula equations. Assessment 9 10 Chemical Properties of Metals – See Metals and Non-Metals Explain that how a substances reacts with other chemicals is determined by its chemical properties. Demonstrate the chemical properties of some metals such as burning magnesium or sodium in water. LO L927: Down to Earth: Metals Matter (Scootle Link) Prac: Flame test to show the colours produced by different metal ions. Research Assignment: Have students research an alloy to find out it’s composition, physical and chemical properties and relate these properties to its use. Students should also compare the properties and uses of the metals that make up the alloy. Chemical Properties of Non-Metals – See Metals and Non-Metals Compare some of the physical properties of non-metals with metals. Look at the distribution of metals and non-metals on the periodic table. Examine the properties of carbon and its allotropes – graphite and diamond. Look at molecular structure, properties and applications. Energy Transformation and Transfer (ACSSU155) Kinetic and Potential Energy (ACSSU155) Physical Sciences Week Teaching Plan 1 Kinetic Energy – See Kinetic and Potential Energy Revise the scientific definition of ‘energy’. Introduce the concept of kinetic energy. Relate the KE of an object to both its mass and velocity. Introduce the equation KE = ½ mv2. Worksheet: Students practice calculating KE, m or v from word problems. 2 Gravitation Potential Energy – See Kinetic and Potential Energy Introduce the concept of gravitational potential energy and explain the formula PE = mgh. Worksheet: Students practice calculating PE, m, g or h from word problems. 3 Rectilinear Motion – See Kinetic and Potential Energy Examine the scenarios of dropping a ball and throwing a ball straight up. Introduce the concept of energy transformation and conservation. Worksheet: Student practice calculating KE and PE at different points in the ball’s motion. Worksheet: Extend calculations to include calculations of other variables e.g. m, v, g or h. 4 Non-Linear Motion – See Kinetic and Potential Energy Examine scenarios where an object is not moving in a straight line but the object is completely stationary at its apex (e.g. pendulum or rollercoaster but not projectile). Prac: Swinging Pendulum Worksheet: Have student practice calculating KE, PE, m, v, g or h of objects not moving in a straight line. 5 Potential Energy – See Kinetic and Potential Energy Discuss other ways energy can be stored as potential energy e.g. elastic potential energy, chemical potential energy. Prac: Students use chemical potential energy to power a globe by creating their own battery from copper and zinc electrodes stuck in lemons connected in series. (Example) 6 Energy Transformation and Loss Introduce the concept of energy transformation and loss e.g. why does a bouncing ball eventually stop? Analyse mechanical and electrical systems to identify energy transformations and loss. Worksheet: Students identify energy transformations and loss from pictures of simple mechanical and electrical systems. 7 Power Plants Explore different types of power generation plants e.g. coal, nuclear, solar, wind, hydro, geothermic, tidal. Research Project: Have students research one form of power generation plant and construct a flowchart to show the energy transformations when the power is used in a household. 8 Heat Energy – See Heat Energy Highlight that often energy is lost from a system in the form of heat and find examples. Discuss the ways that heat can be transferred i.e. conduction, radiation and convection. Prac: heat potassium permanganate crystals in water to show convection. (Example) Examine the structure of a vacuum thermos flask to see how it is designed to keep things hot or cold. 9 1-Week Heat Investigation In groups, students are given the challenge to create the best thermal insulator in a box of a given size using materials they find around the home. As a class, agree on a fair method for testing which insulator box is the best. Test each and discuss the results. 10 Energy in Sustainable Living Discuss how homes can be made more energy efficient by reducing the need for heating and cooling. What other ways can a home be more energy efficient? Research Project: Students research energy efficient homes and design their own energy efficient home. Assessment Earth and Space Science Minerals, Ores and Mining (ACSSU153) Types of Rocks (ACSSU153) Week Teaching Plan 1 Igneous Rocks – See Rocks Review the structure of the Earth and the formation of igneous rocks. Discuss properties of igneous rocks and compare extrusive and intrusive igneous rocks. Prac: Examine various igneous rock samples under the microscope and record observations. 2 Sedimentary Rocks – See Rocks Define sedimentary rocks and discuss how they are formed. Explain different forms of weathering and erosion that contribute to the formation of sedimentary rocks. Discuss the properties and uses of sedimentary rocks. Prac: Examine sedimentary rock samples under the microscope and record observations. 3 Metamorphic Rocks – See Rocks Define metamorphic rocks and discuss regional and contact metamorphism. Review the properties of metamorphic rocks and some uses. 4 Identifying Rocks – See Rocks LO L3065: Get into geology: what rock is that? (Scootle Link) Activity: See Famous Rock Groups in Rocks 5 The Rock Cycle – See Rocks; Geological Change Review the formation of igneous, sedimentary and metamorphic rocks. Introduce the rock cycle (Video 1, 2, 3, 4, 5) LO L926: Down to Earth: rock back in time (Scootle Link) Hands on Activity: The Crayon Rock Cycle Poster: Have students create their own poster to demonstrate the rock cycle using photographs or drawings. 6 Minerals Discuss what minerals are and why they are important Discuss Moh’s hardness scale Activity: Minerals in Toothpaste Activity in Ground Rules 7 Ores Discuss that is meant by an ‘ore’ and how ores are formed. List metals and the ores they come from. Research Assignment: Students research where 5 metals or minerals are mined in Australia and plot them on a map using symbols and a key. 8 9 10 Mining – See Mining Review mining techniques e.g. surface, underground, dredging leaching. Discuss crude oil exploration and mining and the process of fractional distillations Worksheet: Provide students with a diagram showing the fractions that come from crude oil. Students complete the worksheet by writing what products come from each fraction. Impact of Mining – See Mining; Conservation of Resources Discuss the economic and environmental impact of mining. Activity: See Cookie Mining Activity in Mining. Research Report: Students prepare a report on the impact of mines on the environment and how governments and mining companies are trying to lessen the impact. Assessment