Praluent
advertisement

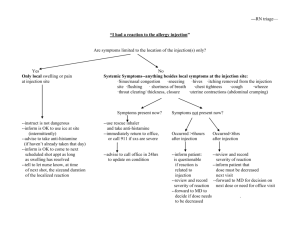
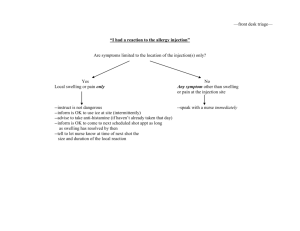
New Drug Introduction: Praluent®/ alirocumab Pharmacology Manufacturer Approval Date Indications Contraindications Black Box Warnings Warnings & Precautions Pregnancy/Lactation Pharmacokinetics Drug Interactions – Precipitant drugs Adverse Effects (Treatment%) [Placebo%] Monitoring Efficacy Dosing - Initial Dosing - Max Renal Adjustment Hepatic Adjustment Praluent is a human monoclonal antibody (IgG1 isotype) that inhibits proprotein convertase subtilisin kexin type 9 (PCSK9). Sanofi and Regeneron Pharmaceuticals July 24, 2015 Indicated as an adjunct to diet and maximally tolerated statin therapy for the treatment of adults with heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease, who require additional lowering of LDL-C History of serious hypersensitivity reaction to alirocumab, including vasculitis and hypersensitivity reactions requiring hospitalization None Hypersensitivity reactions (pruritus, rash, urticarial), including some serious events, have been reported. If signs or symptoms of serious allergic reactions occur, discontinue treatment. Potential for immunogenicity with alirocumab, which may lead to a higher incidence of injection site reactions or loss of efficacy Pregnancy Category: C Lactation Recommendation: unknown A – F = 85%, tmax 3-7 days. Steady state reached after 2 to 3 doses D – Vd = 0.04-0.05 L/kg M – Expected to degrade to small peptides and individual amino acids. Does not affect CYP450 enzymes, PGP, or OATP. E – T1/2: 17 to 20 days. Statins reduce half-life of alirocumab to 12 days; not clinically meaningful Allergic reactions (8.6%) [7.8%] UTI (4.8%) [4.6%] Injection site reactions (7.2%) Diarrhea (4.7%) [4.4%] [5.1%] Bronchitis (4.3%) [3.8%] Influenza (5.7%%) [4.6%] Myalgia (4.2) [3.4%] LDL-C levels within 4 to 8 weeks of initiating or titrating alirocumab 75 mg SC every 2 weeks Administration: Warm to room temperature for 30 to 40 min prior to use. Do not keep at room temperature for more than 24 hours. Inject subcutaneously into stomach, upper arm, or thighs. Injection may take up to 20 seconds. Rotate injection sites. 150 mg SC every 2 weeks No dose adjustment is needed for patients with mild or moderate renal impairment. No data are available in patients with severe renal impairment. No dose adjustment is needed for patients with mild or moderate hepatic impairment. No data are available in patients with severe hepatic impairment. Cost: Source: NY Times – accessed 08/11/2015 Dose(s) Brand – Generic ® 75 mg, 150 mg injection Praluent - alirocumab 1 year $14,600 Summary Praluent® (alirocumab) is the first-in-class PCSK9 inhibitor that reduces LDL-C. Alirocumab is indicated for the treatment of adults with heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease, who require additional LDL lowering despite diet and maximally tolerated statin therapy. Dosing of alirocumab is 75 mg or 150 mg every 2 weeks given as a subcutaneous injection. The most common adverse effects observed include hypersensitivity reactions. LDL should be monitored at 4 to 8 weeks after initiating or titrating alirocumab. References: 1. www.praluent.com 2. Praluent [package insert]. Bridgewater, NJ: Sanofi and Regeneron; 2015. 3. Robinson JG, Farnier M, Krempf M, et al. Efficacy and safety of alirocumab in reducing lipids and cardiovascular events. N Engl J Med 2015; 372:1489-99. 4. Pollack A. The New York Times Website. New Drug Sharply Lowers Cholesterol, but it’s Costly. http://www.nytimes.com/2015/07/25/business/us-approves-drug-that-cansharply-lower-cholesterol-levels.html. Published July 24, 2015. Accessed August 11, 2015. Date Prepared: 8/11/2015 Editor: Peter G. Koval, Pharm.D., BCPS Author: Megan Shah, Pharm.D. Candidate, UNC Eshelman School of Pharmacy