preparation-of-trialkoxyboranes
advertisement

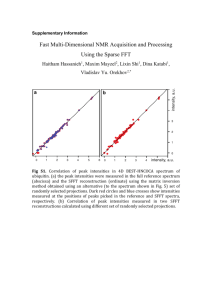
Preparation of Trialkoxyboranes Bobby Deimler Department of Chemistry and Physics, Georgia College and State University, Milledgeville, GA 31033 11/25/08 Abstract: Throughout the history of scientific study scientists have always sought to discover new compounds and apply them in a practical sense to the world around them. One set of compounds that have found uses in several areas are the Trialkoxyboranes. This report documents the preparation of two specific trialkoxyborane compounds, tri-npropyl borate and poly(vinylalcohol)-borate copolymer. These compounds were prepared separately in a two part experiment that was obtained prior to the experimentation date. This procedure required the use of several chemicals most importantly, boric acid, npropranol, poly(vinylalcohol) and NaOH. At the conclusion of this experiment, a clear liquid was obtained for the propyl borate and a magenta, rubbery, solid was obtained for the poly(vinylalcohol) – borate copolymer. The sample for the propyl borate weighed 3.4651 g and made up 87.03% of the total theoretical yield. Both a 1HNMR and an IR spectrum were taken of the propyl borate in order to confirm its identity. Both tests yielded spectra that were similar to the literature sources. This experiment is significant because it presents a way in which both IR and NMR can be utilized in the characterization of the production. This is critical because it allows hands-on experience with these instruments as well as allowing a relatively easy way to identify the product. This experiment is also important because it allows the creation of a definite product, especially in the case of the second part of the experiment. In many experiments only a fraction of a gram of product is obtained. In this reaction a large amount of a brightly colored product is obtained. The ability to interact with a tangible product helps to solidify the concepts of the experiment in a way that is easily memorable. Lastly, this experiment also demonstrates the concept of a dehydration reaction in the first part of the procedure. This is a useful organic reaction that will be utilized many times over the course of a career in chemistry. Introduction: One of the purposes of science is to find practical applications for the elements and compounds discovered through experimentation. One set of compounds that have found particular use in industry is trialkoxyboranes. These compounds have a wide variety of functions in photography, polymer chemistry, biochemistry and in the glass industry. (1) For example, in biochemistry these compounds have found use as chemosterilants for houseflies and screwworm flies. Trialkoxyboranes cause these insects to reproduce at a rate that is only one percent of their normal, limiting populations in areas where they can be a nuisance. (1) In the experiment described below, two separate trialkoxyboranes were prepared. The first was a tri-n-propylborate. This compound was prepared through the reaction of boric acid with an organic alcohol, which in this case was n-propanol. (Figure 1) B(OH)3 + 3(C3H7OH) B(OC3H7)3 + 3H20 (Figure 1) This is a dehydration reaction in which the hydroxyl group on the alcohol is prompted to leave by the hydrogen of the acid. Since the boric acid is an acid it is willing to give up protons (H+) because it is a proton donor. (Figure 2) By this same definition a base, which in this case is the hydroxyl group on the alcohol, is then a proton acceptor. This is known as the Bronsted-Lowry definition of acids and bases. The hydroxyl group on the alcohol is normally a very poor leaving group, meaning that it is very stable. It becomes necessary to protonate the hydroxyl group with one of the protons from the boric acid. This causes the hydroxyl group to become a -OH2+ which is a more feasible leaving group. This group then departs from the alcohol and goes on to form water, a byproduct of the reaction. (Figure 3) OH B HO + OH OH (Figure 2) H H H | | | + H- C ŠC ŠC - H | | | OH2- H H B HO H H H | | | H-CŠCŠC -H | | | OH H H O (Figure 3) H20 The space that is left open on the alcohol then reacts with the open oxygen on the boron to form the trialkoxyborane structure. (Figure 4) OH B HO O H H H | | | + H- C ŠC ŠC - H | | | H H OH B HO H H H | | | O- C Š C Š C - H | | | H H H (Figure 4) The second structure that was formed in this experiment was a tetraalkoxyborane. This structure was formed from a base trialkoxyborane structure. This compound was formed through the reaction of the trialkoxyborane, poly(vinylalcohol) and NaOH. (Figure 5) B(OC3H7)3 + NaOC2H4 (H4C2O)B(OC3H7)3- + 3H20 (Figure 5) The difference between a tri- and tetraalkoxyborane is that the first only has three of the alkoxyborane structures connected to it, while the latter has four. This reaction proceeds a little differently than that of the first reaction. In this reaction a tetrahydroxyborate is formed through the addition of a hydroxyl group to the original tri-n-propylborate structure. (2) This is achieved when the base, in this case NaOH, is added to the reaction solution near the end of the experiment. (Figure 6) OH | B OH OH (Figure 6) B- + NaOH OH OH OH OH Once this addition is achieved the same dehydration reaction that occurred in the primary part of the experiment happens here and a poly(vinylalcohol) group is attached in the place of the hydroxyl group that was just described above to give: H4C2O OC3H7 B H7C3O OC3H7 This structure is basically a textbook example the basis for a cross-linking of polymer. What occurs is that the two alcohols on either side of the center atom form a chain with one another and then with the center boron linking the two opposite sides together. (3) (Figure 7) CH CH O CH2 O CH O B CH2 (Figure 7) O CH This cross-linking can occur many times and in several places across parallel carbon chains. When looking at this picture one may wonder why there are only three carbons on each side of the boron when the structure above shows as many as six on one side. This is where the bonds on the corner carbons in Figure 7 come into play. They represent the rest of the carbon chain not present in the picture. Not all of the carbons in the chain are necessary in order to make the cross-linked structure. Instead some parts of the chain continue on in other directions creating the potential for other chains and cross-links. These various cross-links help to ensure structural stability. The B-O bond is an especially good bond to build a structure around because it is stable in air. (1) This is due to the fact that the oxygen can donate electrons into the p-orbital of the boron. (1) The more electrons that the boron possesses in it’s empty orbital the less it is going to want to accept and the less of an acid it is going to be, by the Lewis definition. This is a good because it helps the structure resist the water that is in the air. If it did not have these stable bonds than it would go through a hydrolysis reaction with the moisture in the air and turn back into boric acid effectively undoing the reaction. Procedure: This experiment was spilt up into two separate sections, one for each of the separate products. The experiment to prepare the propyl borate was performed first. To begin this experiment, 1.0179 g of boric acid was placed into a round bottom flask along with a boiling stone, 4 mL of dry n-propanol and 2 mL of toluene. This solution was attached to a reflux condenser which had a glass arm attached to the bottom. The flask was attached to the lower of the two holes of the arm. A small piece of tubing was placed inside the other hole on the arm, in a fashion that droplets from the reflux would be able to drain into it. The end of this piece of tubing was clamped using a gator clip to keep the product from spilling out. The entire apparatus was tilted sideways to make it easier for the reflux droplets to flow into the side arm. The solution was heated until reflux occurred. While the solution was refluxing the side arm was watched carefully. As the reflux continued this arm began to fill with two different liquid materials, water and toluene. Since water is denser than toluene the two liquids would form one layered liquid with water on the bottom. The side arm was allowed to fill until there was only water present within it. When this occurred, the water was removed using a pipette. This process was continued for one hour. At the end of this period the solution was removed from the apparatus. A pipette was used to transfer this solution to another 10 mL flask containing a new boiling stone. This new flask was attached to a hickman still, which had a reflux condenser on top of it. The solution in the flask was heated until it began to reflux. Any product that distilled up to 120 C was removed from the system with a Pasteur pipette. When the temperature reached 120 C the heat was turned off and the flask was allowed to return to room temperature. The solution that remained in the flask after cooling was the product that the experiment sought to obtain. This product was a clear liquid. An empty plastic vial was weighed and determined to be 6.6574 g. The sample was then added changing the mass to 10.1225 g. This means that the mass of the obtained sample was 3.4651 g. The sample was then placed into a plastic vial so that it could be safely transported the organic lab where both a 1HNMR and an IR were taken of the sample. Both of these spectra are included in the results section of this report. The second part of this experiment was designed to prepared the poly(vinylalcohol)-borate copolymer. To begin this portion of the experiment a 4% solution of boric acid was created by adding .2074 g of boric acid to 5 mL of water in a 20 mL beaker. In a separate beaker a 4% solution of poly(vinylalcohol) was created by slowly adding 1.3688 g of poly(vinylalcohol) to 30 mL of boiling water. Once all of the alcohol had been added the solution was allowed to return to room temperature. At this point the sample solution became very viscous and was similar to honey in consistency. Several drops of phenolphthalein were then added to the solution. The 4% boric acid solution was then added to the 4% poly(vinylalcohol) solution. These solutions needed to be stirred together with a stir rod due to the thick consistency of the poly(vinylalcohol) solution. A base, 6 M NaOH, was then added dropwise to the new solution. This caused the solution to turn magenta and begin to solidify. After about ten drops and continuous stirring, a pink solid was created that had the consistency of dry rubber cement. This was the final product of the reaction. Results: Figure 8 : By looking at this spectra one can see several important peaks. The first is the B-O peaks that appear between 1300 and 1500 cm-1. The second set is the C-O peaks, which occur at 1000 cm-1. The last important peak to note is the large rounded peak in the left side of the spectrum from around 3200 to 3500 cm-1. This peak is caused by water that was still in the system when the spectrum was taken (4,5). (6) Figure 9: The chart above is a 1HNMR spectrum and contains six different peaks. These peaks represent possible ways that the samples structure could be configured. From right to left, the first peak at 0 is most likely a saturated alkane with the structure R-H. The second, third and fourth peaks are probably ROH groups. These peaks go from .75 to 1.5, 1.5 to 2.0, and 2.2 to 2.5 respectively. The fifth peak from 3.5 to 4.0 is probably indicating an ester of the form RCO2-CH. The final peak from 7.0 to 7.5 normally indicates an aromatic structure. (6) Figure 10: This is a reference IR spectrum for the tripropylborate sample. (7) Figure 11: This is a reference 1HNMR spectrum for the tripropylborate sample. (8) Percent Yield = actual amount theoretical amount x 100 3.4651 g 3.9812 g x 100 = 87.03% yield for tripropylborate Discussion: The procedure for this experiment was performed successfully and both compounds were able to be prepared. The truth of this fact can be confirmed by the IR and 1HNMR spectra in the case of the tripropyl borate and by the physical characteristics in the case of the poly(vinylalcohol) copolymer. An IR spectrum was taken of the propylborate sample at the conclusion of the first part of the experiment. This spectrum showed several important peaks. The first was the B-O peaks that appear between 1300 and 1500 cm-1. These peaks are important because they show the connection between the propyl alcohol and the center boron in the structure. These peaks are not all conclusive though because they could also show the connection between the hydroxyl groups and the boron center in the boric acid structure. For this reason it is necessary to look at a second set of important peaks in this spectrum as well. These are the C-O peaks, which occur at 1000 cm-1. These two peaks are important because they indicate that there is a CO bond being formed within the structure. This set of peaks indicates the bond between the oxygen of the boron structure and the carbon of the alcohol. The item that best supports this conclusion are the two peaks that appear at 1000 cm-1. The appearance of these two peaks indicates that this is an ester C-O structure. Since the structure formed in this experiment is an ester, it supports the conclusion that the target structure has in fact been prepared. The last significant peak to note is the large rounded peak in the left side of the spectrum from around 3200 to 3500 cm-1. This peak is caused by water that was still in the system when the spectrum was taken. This water was probably left over from the time that the solution spent in reflux. The presence of water indicates that the solution needed to be refluxed for a slightly longer period of time, which would have removed more water from the system and narrowed this peak. In order to determine more information about the sample a 1HNMR was taken as well. This spectrum returned several peaks. The first peak indicated a saturated alkane with the structure R-H. This would make sense as the alcohol used in this part is a series of carbons in a chain. This chain easily forms a saturated alkane structure by filling it available space with hydrogens. The second, third and fourth peaks are probably ROH groups. This peak could occur for two separate reasons. The first is that there could be some unreacted reagents left in the solution. Both the hydroxyl in the boric acid and in the alcohol could cause this peak to occur. The second explanation is that this peak is for the water, which is produced as a byproduct of the reaction. When one sees the structure R-OH it is initially assumed to be an alcohol, but this structure could just as easily be an H-OH as well. The fifth peak indicates an ester of the form RCO2-CH. As stated before the target structure in this experiment was an ester so the appearance of an ester peak in the spectrum supports the fact that the desired structure has been formed. The final peak from 7.0 to 7.5 normally indicates an aromatic structure. This can be attributed to the toluene that was used in this experiment as a solvent. The appearance of an aromatic peak in the NMR indicates that there was probably some toluene left over in the final sample solution. Once a spectrum was taken of the sample in both media, the spectra obtained were compared to reference spectra for accuracy. When the IR obtained was compared against the reference it was determined that they were very similar. The major difference between the two was the width of the peak that occurs around 3000 cm-1. By comparison of the two spectra it is obvious that the sample obtained in the experiment performed contained much more water than the sample that was used for the reference. It is also possible that the sample contained some unreacted alcohol as well. This is due to the fact that the peak in the experimental IR is very similar to the peak that occurs in the 1propanol IR around that same area. (9) With this in mind, it may have been a combination of the vibrations of the OH groups of both the alcohol and the water that caused this peak to expand to the size that it was in the experimental spectrum. When the NMR spectra were compared it was determined that the NMR obtained from the experimental sample had several more peaks than the reference. The reference only possessed three peaks while the experimental sample contained six peaks. The three extra peaks in the experimental spectrum are attributed the aromatic peak, an unreacted alcohol peak, and the saturated alkane peak. The extra alcohol peak is attributed to the fact that my sample had some unreacted alcohol still left in solution. This is supported by the fact that this same peak occurs in the HNMR of unreacted 1-proponal. (9) The fact that the sample structure has the saturated alkane peak and the reference does not indicates that this peak is most likely due to unreacted material left in the sample. Perhaps some of the alcohol did not fully react with the acid and that which remained allowed this peak to rise. The last extra peak is the aromatic, which again is attributed to toluene that remained in the sample solution. At the conclusion of the first part of the experiment a percent yield was calculated for the sample and it was determined that 87.03% of the theoretical yield was produced. This was more than enough to run the required spectra and also showed a successful experimental reaction. In the second part of this experiment, a poly(vinylalcohol)-borate copolymer was prepared. This compound was identified through more qualitative than quantitative means. Success in this experiment was measured through two specific physical observations that occurred throughout the experiment. The first was when the poly(vinylalcohol) was added to the boiling water causing it to become more viscous and sticky. This gave some physical evidence that a reaction was occurring. This reaction helped to create an enviroment in which the final polymer could be created. The second important physical observation came when the base was added to the combination of the poly(vinylalcohol) and boric acid solutions. This caused the solution to react further and become a gummy solid with the consistency of rubber cement. This solid filled roughly one quarter of a 100 mL beaker. It also turned the solution magenta, which was a product of the bases interaction with phenolphthalein. Due to the large amounts of the colored solid that was produced it was safe to conclude that the reaction had been achieved successfully. This product was then compared against other instances where this or similar reactions had been done in literature and it was found that the characteristics of the product were similar. (3) Conclusion: The goal of this experiment was to prepare a solution of propylborate and a solid mass of poly(vinylalcohol)-borate copolymer. This was achieved successfully with a 87.03% yield of the propylborate and a successful synthesis of the copolymer. Through a comparison of IR and 1HMR spectra it was shown that the propylborate sample produced in the experiment was very similar to reference spectra obtained in literature. Through comparison of physical characteristics of the experimental borate copolymer and those produced in literature it was able to be determined that the sample produced in the second part of the experiment was the target copolymer. This experiment further shows that many compounds produced in the laboratory have great practical application in the outside world. Many of which are used in recognizable industries like photography or glass and glazing. A possible continuation of this experiment would be to react alcohols other than 1-proponal or poly(vinylalcohol) with the boric acid and see if they react in a different way. Perhaps they would form structures varying from the form that was seen in this experiment. An IR and NMR study of these compounds would help student’s ability to read and interpret both types of spectra. References: 1. Szafran, Zvi; Pike, Ronald M.; Singh, Mono M., Microscale Inorganic Chemistry: A Comprehensive Laboratory Experience; 1st Ed.; J. Wiley: 1991; pp. 248-251 2. Sorell, Thomas N., Organic Chemistry; University Science Books: 2006; pp. 290 3. Casassa, E.Z.; Sarquis, A.M.; Van Dyke, C.H. “The Gelation of Polyvinyl Alcohol with Borax”. J. Chem. Ed. 1986, 63, pg. 1-4. 4. Michigan State University. Infrared Spectroscopy. http://www.cem.msu.edu/ ~reusch/VirtualText/Spectrpy/InfraRed/infrared.htm (accessed11/30/08) 5. Socrates, George. Infrared and Raman Characteristic Group Frequencies: Tables and Charts; John Wiley and Sons: 2004; pp. 249-250 6. Michigan State University. Proton Chemical Shift Ranges. http://www.cem. msu.edu/~reusch/OrgPage/nmr.htm (accessed 11/30/08) 7. Pouchert, C., Ed., Aldrich Library of FT-IR Spectra, Aldrich Chemical Co.: Milwaukee, WI, Vol. II, pp. 1132 C 8. Pouchert, C., Ed., Aldrich Library of NMR Spectra, Aldrich Chemical Co.: Milwaukee, WI, Vol. III, pp. 725 B 9. National Institute of Advanced Industrial Science and Technology. Spectral Database for Organic Compounds. http://riodb01.ibase.aist.go.jp/sdbs/cgibin/direct_frame_top.cgi (accessed 11/30/08) Acknowledgements: I would like to acknowledge the Department of Chemistry and Physics for the opportunity to use the lab and the equipment necessary for this experiment to take place.