Chemical reactions
advertisement

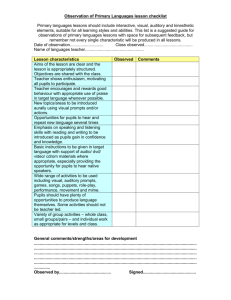
1 of 4 The National Strategies Secondary Secondary Framework in Science, 3.2 Chemical reactions 3.2 Chemical reactions Year Yearly learning objectives Amplification – pupils could learn to Strategies for progression Rich questions 7 sort some reactions into reversible and irreversible explain the difference between reversible and irreversible changes What is a chemical reaction? recognise that new materials are made during chemical reactions use experimental evidence to explain that a chemical reaction has taken place Provide pupils with the opportunity to evaluate the criteria used to decide if changes are chemical or physical, and reversible or irreversible. describe the ways in which materials can vary in their appearance and state using a range of features Model for pupils how to identify and record the range of evidence that a chemical reaction has taken place. Create opportunities for pupils to discuss how well their simple particle model can be used to explain chemical reactions and whether it needs to be developed. Is the iron in a car the same as the iron in blood? Is carbon good news for iron? Are elements always simple? Can a compound be pure? Is a compound a mixture with glue? Explore with pupils some of the common misconceptions about elements, compounds and mixtures, and why people might have these. Provide pupils with the opportunity to investigate and classify different types of reactions. 8 recognise that materials can be made up of one or more kinds of particles describe the type and arrangement of atoms in elements, compounds and mixtures describe and develop a particle 00215-2009PDF-EN-01 describe how elements combine to form the building blocks of all substances describe elements as consisting of only one type of atom use a simple model or analogy to show similarities and differences between elements, compounds and mixtures Create opportunities for pupils to experience and reflect on the search for patterns that underpinned the development of the periodic table. Provide pupils with opportunities to evaluate different models used in published materials to Is burning always destructive? What makes a chemical reactive? Is the periodic table complete? Why is the periodic © Crown copyright 2009 2 of 4 Year The National Strategies Secondary Secondary Framework in Science, 3.2 Chemical reactions Yearly learning objectives Amplification – pupils could learn to model to explain the differences between the terms atoms, elements, compounds and mixtures Strategies for progression Rich questions explain atoms, elements, compounds and mixtures. table such an odd shape? Explore with pupils the value of representing chemical reactions using the model of symbols and numbers. Are all recipes based on chemical reactions? Provide opportunities for pupils to investigate and classify a wider range of reactions and identify common features linked to their formula, name or behaviour. Provide opportunities for pupils to explore how the model of an atom has developed over time. 9 use a particle model to construct predictions for simple chemical reactions and to produce word equations use a simple modelling kit to interpret the rearrangement of atoms during a chemical reaction describe some simple generalisations about the characteristics of groups of elements based on their position in the periodic table distinguish between metals and other materials using their properties Create opportunities for pupils to manipulate the models of chemical reactions and energy transfer to explain how reactions are initiated. Provide opportunities for pupils to use a range of evidence to investigate bonding patterns in relation to the periodic table. Create opportunities for pupils to investigate the conservation of matter practically and explore its implications. Challenge pupils to explain how the conservation of matter is represented in symbol equations. Provide pupils with the opportunity to explore reasons 00215-2009PDF-EN-01 Could the Fire Brigade be renamed the Oxygenation Brigade? Where do elements come from? Is purity overrated? Are endothermic reactions energy thieves? Can we design new atoms? Can we classify every reaction? Could you have an atom with a thousand protons? Is a chemical reaction like shuffling a deck of cards? Is a chemical reaction substance abuse? © Crown copyright 2009 3 of 4 Year The National Strategies Secondary Secondary Framework in Science, 3.2 Chemical reactions Yearly learning objectives Amplification – pupils could learn to Strategies for progression Rich questions for developments in the model of the atom. Encourage pupils to discuss and develop ideas about how the properties of subatomic particles might explain different chemical reactions. 10 use a particle model to construct predictions for chemical reactions and to produce symbol equations explain the evidence that a chemical reaction has taken place in terms of energy transfer and rearrangements of bonds between atoms explain chemical reactions in terms of new products being made and energy changes taking place represent simple reactions by word equations and use these to predict the products of chemical reactions use a formula to explain why a substance is a compound or an element Create opportunities for pupils to evaluate the efficiency of a chemical process by calculating yields from evidence gathered. Create opportunities for pupils to evaluate the range of models used to represent different types of chemical bonds and possible misconceptions they could cause. Structure opportunities for pupils to apply their understanding of chemical bonds to explain chemical reactions and to predict the behaviour of materials. Why are insulators possessive about their electrons? Is a semiconductor half as good as a conductor? Are alloys designer metals? Are solutions of ionic compounds shocking? Provide pupils with the opportunity to explore the limitations of models of materials and reactions and how these can be developed. Challenge pupils to use a range of evidence to explain how formulae and other models are used to make sense of chemical reactions. 00215-2009PDF-EN-01 © Crown copyright 2009 4 of 4 The National Strategies Secondary Secondary Framework in Science, 3.2 Chemical reactions Year Yearly learning objectives Amplification – pupils could learn to Strategies for progression Rich questions 11 use a particle model to predict the outcome of chemical reactions and to produce balanced symbol equations use a modelling kit to interpret the rearrangement of atoms during chemical reactions to develop an appreciation of size and shape of molecules, and use this to illustrate bonding to explain changes in the characteristics of products and reactants as a result of chemical reactions Create opportunities for pupils to integrate the energy-transfer and bonding models to explain the concept of bond energy and how this explains the behaviour of materials and reactions. Can chemical bonds set your hair? explain the evidence that a chemical reaction has taken place in terms of rearrangements of bonds between atoms, using the model of the differences of electron structure between elements explain chemical reactions as the rearrangement of atoms, link this to conservation of mass and use it to predict the amount of product obtained from a reaction based on conservation of mass use a subatomic particle model to explain how atoms join together to form different substances Challenge pupils to explore how intermolecular interactions can be used to explain the properties and applications of materials. Explore with pupils how they can devise and evaluate analogies to explain the concept and application of chemical equilibrium. What can you learn about intermolecular forces from an iceberg? Are polymers the spaghetti of the chemistry lab? Why do some atoms go around in pairs? Provide opportunities for pupils to investigate electrolysis and explain the process by using qualitative and quantitative models. Extension use a particle model to predict the outcome of complex chemical reactions and to produce balanced symbol equations and ionic half equations when appropriate explain the evidence that a chemical reaction has taken place (in a system at equilibrium) in terms of energy transfer and rearrangements of bonds between atoms 00215-2009PDF-EN-01 explain the link between electron arrangement, bonding and chemical reactions explain how different atomic structures affect the properties of a range of substances, e.g. crystals, polymers, water use the concept of bond energy to explain exothermic and endothermic reactions, including catalysis, and evaluate the efficiency of experimental procedures by comparing the predicted and actual amounts of products obtained © Crown copyright 2009



![afl_mat[1]](http://s2.studylib.net/store/data/005387843_1-8371eaaba182de7da429cb4369cd28fc-300x300.png)