- Lorentz Center
advertisement

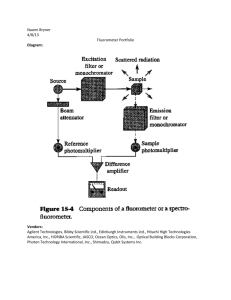
Agilent Low RNA Input Fluorescent Linear Amplification Kit Protocol (5184-3523). Fluorescent cRNA Synthesis Procedure cDNA Synthesis From Total RNA (Time required: ~3 hours) 1. Add 50 to 500 ng total RNA in a volume of 10.3 µL or less. The total concentration should be at least 5 ng/µL. □ 2. Add 1.2 or 5 µL of T7 Promoter Primer (from kit). □ 3. Use nuclease-free water to bring the total reaction volume to 11.5 µL. □ ng Sample for use 500 # Sample Name Conc. ng/µl µl Sample µl T7 primer 1 #DEEL/0! 2 #DEEL/0! 3 #DEEL/0! 4 #DEEL/0! 5 #DEEL/0! 6 #DEEL/0! 7 #DEEL/0! 8 #DEEL/0! 9 #DEEL/0! 10 #DEEL/0! 11 #DEEL/0! 12 #DEEL/0! 13 #DEEL/0! 14 #DEEL/0! 15 #DEEL/0! 16 #DEEL/0! 17 #DEEL/0! 18 #DEEL/0! 19 #DEEL/0! 20 #DEEL/0! µl H2O #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! #DEEL/0! □ 4. Incubate the reaction at 65°C in a heating block for 10 minutes. 5. 6. Place the reactions on ice and incubate for 5 minutes. □ Immediately—prior to use, gently mix the following □ components by pipetting, in the order indicated, at room temperature: Agilent Low RNA Input Fluorescent Linear Amplification Kit Protocol 16-2-2016 1 Be sure to always make a mix for 10% (round up to whole) reactions more or at least 1. Number of reactions 2 Lot # kit: cDNA Master Mix 5X First Strand Buffer 0.1 M DTT 10 mM dNTP mix MMLV RT RNaseOUT Total Volume (µl) 4 2 1 1 0.5 8.5 8 4 2 2 1 17 Pre-warm the 5X First Strand Buffer by incubating the vial in a 65°C waterbath for 3-4 minutes. Vortex briefly and spin the tube briefly in a microfuge. 7. To each sample tube, add 8.5 µL cDNA Master Mix. □ 8. Incubate samples at 40°C in a circulating water bath for 2 hours. □ 9. Move samples to a heating block or water bath set to 65°C and incubate for 15 minutes. □ 10. Move samples to ice. Incubate on ice for 5 minutes. □ 11. Spin samples briefly in a microcentrifuge. □ Fluorescent cRNA Synthesis: in vitro transcription and incorporation of cyanine 3or cyanine 5-CTP (Time required: 2.5 hours) 12. Immediately, prior to use, gently mix the following components by pipetting, in the order indicated, at room temperature: □ Be sure to always make a mix for 10% (round up to whole) reactions more or at least 1. Agilent Low RNA Input Fluorescent Linear Amplification Kit Protocol 16-2-2016 2 Number of reactions Number of reactions Cy-3 Number of reactions Cy-5 Transcription Master Mix Nuclease-free water 4X Transcription Buffer 0.1 M DTT NTP Mix 50% PEG RNaseOUT Inorganic Pyrophosphatase T7 RNA Polymerase Total 2 1 1 Volume (µl) 15.3 20 6 8 6.4 0.5 0.6 0.8 57.6 Cy3 CTP 30.6 40 12 16 12.8 1 1.2 1.6 115.2 NEL580 Lot# Cy5 CTP NEL581 Lot# 57.6 Mix 2.4 Cy3 CTP 60 Total 57.6 Mix 2.4 Cy5 CTP 60 Total Pre-warm the 50% PEG solution by incubating the vial in a 40°C waterbath for 1 minute. Vortex briefly and spin the tube briefly in a microfuge. 13. To each sample tube, add 60.0 µL of Transcription Master Mix. Gently mix by pipetting. Agilent Low RNA Input Fluorescent Linear Amplification Kit Protocol 16-2-2016 □ 3 # 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 14. Name Label with Incubate samples in a circulating water bath at 40°C for 2 hours. □ Purification of Amplified cRNA (Machery-Nagel NucleoSpin RNA II kit) 15. Add 20 µL of nuclease free-water to your cRNA sample □ 16. Add 350 µL of Buffer RA1 and vortex to mix. □ 17. Add 350 µL of ethanol (70%) and mix by vortexing. □ 18. Transfer 800 µL of cRNA sample to an nucleospin RNA II □ Agilent Low RNA Input Fluorescent Linear Amplification Kit Protocol 16-2-2016 4 column. Centrifuge the sample for 30 seconds at 11.000 RPM. Place the column in a new collection tube. □ 19. Add 200 µL of buffer RA2 to the nucleospin RNA II column. Centrifuge the sample for 30 seconds at 11.000 RPM. Place the column in a new collection tube. 20. Add 600 µL of buffer RA3 to the nucleospin RNA II column. □ Centrifuge the sample for 30 seconds at 11.000 RPM. Discard flowtrough and place column back into the collecting tube. 21. Add 250 µL of buffer RA3 to the nucleospin RNA II column. Centrifuge the sample for 2 min at 13.000 RPM to dry the membrane competely. Place the column into a nucleasefree 1,5 ml microcentrifuge tube. □ 22. Elute the RNA in 60 µL nuclease free H2O and centrifuge at 13.000 RPM for 1 min. □ 23. Add the eluate from step 22 again directly onto the nucleospin RNA II column. Centrifuge at 13.000 RPM for 1 min. □ 1. Perform wavescan on nanodrop to determine labelling efficiency and amplification. Save the report on disk. 2. If requested evaluate labelled sample on an Agilent 2100 bioanalyzer. Agilent Low RNA Input Fluorescent Linear Amplification Kit Protocol 16-2-2016 5 Agilent Low RNA Input Fluorescent Linear Amplification Kit Protocol 16-2-2016 6 Overview Concentrations and Hybridisation # 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 Name Cy5 Label Cy3 Conc. µg/ml µg on array Slide barcode µl required Batch number A B C D E F G H Agilent Low RNA Input Fluorescent Linear Amplification Kit Protocol 16-2-2016 7 Materiallist. Materials for general purposes. sterile nuclease-free reaction tubes (0,5 and 1,5 ml) nuclease-free water ice Microcentrifuge Incubator (waterbath or PCR machine 65ºC and 40ºC) vortex microplate centrifuge Magnetic stir plate Speedvac Nanodrop Bioanalyzer cDNA Synthesis From Total RNA 50 to 500 ng total RNA Low input fluorescent amplification kit (Agilent 5184-3523): cDNA Master Mix T7 Promoter Primer 5X First Strand Buffer 0,1 M DTT 10 mM dNTP mix MMLV RT RNaseOUT Fluorescent cRNA Synthesis: in vitro transcription and incorporation of cyanine 3or cyanine 5-CTP Low input fluorescent amplification kit (Agilent 5184-3523): Transcription Master Mix 4X Transcription Buffer 0,1 M DTT NTP Mix 50% PEG RNaseOUT Inorganic Pyrophosphatase T7 RNA Polymerase Cy3-CTP (Perkin-Elmer NEL580) Cy5-CTP (Perkin-Elmer NEL581) Agilent Low RNA Input Fluorescent Linear Amplification Kit Protocol 16-2-2016 8 Purification of Amplified cRNA (RNeasy kit) Buffer RLT ethanol (96-100% purity) RNeasy mini column buffer RPE Agilent Low RNA Input Fluorescent Linear Amplification Kit Protocol 16-2-2016 9