sequence-double dye
advertisement

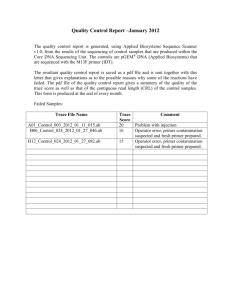
Double Dye Sequencing Protocol 1. Make master mixture with the primers (1pmol/l), sequencing buffer (3.54X), dNTP’s, and DNA polymerase (5U/ul). Order Master Mix CF A C G T 1 sequencing buffer (3.54X) 1X .4 .4 .4 .4 2 dNTP’s .5 .5 .5 .5 3 DNA polymerase (5U/ul) 0.25U .05 .05 .05 .05 4a 72nM 0.08 0.08 0.08 0.08 primer 1 (1pmol/l) 4b 72nM 0.08 0.08 0.08 0.08 primer 2 (1pmol/l) Total 1.1 1.1 1.1 1.1 X 2. Aliquot 0.5 - 0.7 ul of template into A, C, G, and T wells. Put into bottom of wells. 3. Aliquot 1.0 l of master mix into each well. Put on the sides of the wells. 4. Spin in centrifuge to 1.5 X 1000 rpm to combine template and master mix. 5. Thermocycle protocol: Primer Set 1 Primer Set 2 Primer Set 3 94.0°C 5 min 94.0°C 5 min 94.0°C 5 min 94.0°C 15 sec 94.0°C 15 sec 94.0°C 15 sec 65.0°C 15 sec 69.0°C 15 sec 66.0°C 15 sec 72.0°C 15 sec 72.0°C 15 sec 72.0°C 15 sec 72.0°C 5 min 72.0°C 5 min 72.0°C 5 min 4.0°C ∞ 4.0°C ∞ 4.0°C ∞ 6. After thermocycling is complete, add 1 l stop solution to side of each well. Centifuge at 1500 rpm to combine with sequencing reaction. 7. Keep sample in freezer until right before denaturing and loading. 8. Clean gel and pre-run the sequencer. 9. Denature for 3 minutes at 95C in thermocycler. 10. Immediately put in ice bath to quench the reaction and start loading. Load .7 l into each well. *Move quickly as possible for everything.