Chemistry Worksheet: Science Reasoning

Chemistry Worksheet: Science Reasoning
Short Answer
Figure 14-1
1. Arrow C in Figure 14-1 is best described as ____.
2. Arrow A in Figure 14-1 represents ____.
3. Arrow B in Figure 14-1 represents ____.
4. Figure 14-1 represents the ____.
A mass spectrometer is a device for separating atoms and molecules according to their mass. A substance is first heated in a vacuum and then ionized. The ions produced are accelerated through a magnetic field that separates ions of different masses. The graph below was produced when a certain element (element X) was analyzed in a mass spectrometer. Use the graph to answer the questions below.
5. How many isotopes of element X exist?
6. What is the mass of the most abundant isotope?
7. What is the mass of the least abundant isotope?
8. What is the mass of the heaviest isotope?
9. What is the mass of the lightest isotope?
10. Without performing any calculations, predict the approximate atomic mass for element X. Explain the basis for your prediction.
11. Using the data given by the graph and identify the unknown element.
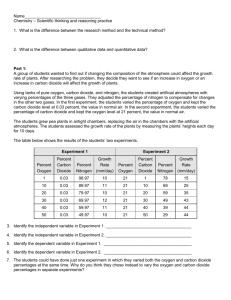
A group of students wanted to find out if changing the composition of the atmosphere could affect the growth rate of plants. After researching the problem, they formed two hypotheses.
Hypothesis 1: Will increasing the percentage of oxygen in the atmosphere make plants grow more rapidly?
Hypothesis 2: Will increasing the percentage of carbon dioxide in the atmosphere makes plants grow more rapidly?
Using a mixing valve and tanks of pure oxygen, carbon dioxide, and nitrogen, the students created artificial atmospheres with varying percentages of the three gases. They adjusted the percentage of nitrogen to compensate for changes in the other two gases. In the first experiment, the students varied the percentage of oxygen and kept the carbon dioxide level at 0.03 percent, the value in normal air. In the second experiment, the students varied the percentage of carbon dioxide and kept the oxygen level at 21 percent, the value in normal air.
The students grew pea plants in airtight chambers, replacing the air in the chambers with the artificial atmospheres. The students assessed the growth rate of the plants by measuring the plants’ heights each day for 10 days.
The table below shows t he results of the students’ two experiments.
Experiment 1 Experiment 2
Percent
Oxygen
1
Percent
Carbon
Dioxide
0.03
Percent
Growth
Rate
Nitrogen (mm/day)
98.97 10
Percent
Oxygen
21
Percent
Carbon
Dioxide
1
Percent
Growth
Rate
Nitrogen (mm/day)
78 15
10
20
30
40
0.03
0.03
0.03
0.03
89.97
79.97
69.97
59.97
11
10
12
11
50 0.03 49.97 10
12. Identify the independent variable in Experiment 1.
13. Identify the independent variable in Experiment 2.
14. Identify the dependent variable in Experiment 1.
21
21
21
21
21
10
20
30
40
50
69
59
49
39
29
25
35
43
44
44
15. Identify the dependent variable in Experiment 2.
16. The students could have done just one experiment in which they varied both the oxygen and carbon dioxide percentages at the same time. Why do you think they chose instead to vary the oxygen and carbon dioxide percentages in separate experiments?
17. Are these graphs the proper way to graph the data?
Two students are presented with a problem in science class. They need to identify specific compounds in an unknown mixture. This “mystery” mixture could include one or more of the following compounds: cornstarch, baking powder, and powdered sugar. All of these compounds are white and are difficult to distinguish using sight alone.
To analyze the mixture, the students must first find ways to identify the individual compounds. Their teacher gives them five test tubes; distilled water; two bowls for the water samples; samples of cornstarch, baking powder, and powdered sugar; dropper bottles; iodine solution; white vinegar; three shallow pans; and a candle with matches.
The students place small amounts of cornstarch, baking powder, and powdered sugar in separate piles in the shallow pan. Then they place some distilled water in a small bowl. They add a drop of vinegar to each sample and record what happens in a data table. Only the baking soda shows any change. The soda begins to fizz due to a gas being given off.
Next they place small amounts of each compound into the other shallow pan and distilled water in another small bowl. This time, they add a drop of iodine to each sample and record their results. Only the sample of cornstarch turns blue.
They finally place a small amount of each compound in three separate test tubes and distilled water in a fourth test tube. They use a lit candle to gently heat the bottom of each test tube. Only the sugar shows any signs of melting. The other compounds are unaffected.
After finding unique ways of testing for each compound, students then repeat all three tests on the mystery mixture. The following data table is a record of their results.
Addition of Addition of Heated with
Material tested
Distilled water
Cornstarch
Sugar
Baking soda
Vinegar
No change
No change
No change
Fizzing takes place
Iodine
No change
Turns blue
No change
No change
Candle
Boiled
No change
Compound melted
No change
Mystery mixture Fizzing takes place Turns blue No change
18. What kind of research did the students conduct?
19. What was the control that was used in the experiment?
20. Which compounds are present in your mystery mixture?
In Figure 1, a simple mercury barometer is made by filling a long glass tube with mercury and then inverting the open end of the tube into a bowl of mercury. Answer the following questions about the simple mercury barometer shown here.
21. What occupies the space above the mercury column in the barometer’s glass tube?
22. When the barometer in Figure 1 is moved to a higher elevation, such as an altitude of 5000 meters, the column of mercury changes as shown in Figure 2. Why is the mercury column lower in Figure 2 than in
Figure 1?
23. Suppose the barometer in Figure 1 was carried into an open mine 500 meters below sea level. How would the height of the mercury column change? Explain why.
24. What prevents mercury from flowing out of the glass tube into the bowl of mercury?
25.
Suppose a tiny crack formed at the top of the barometer’s glass tube. How would this event affect the column of mercury? Explain why.
Chemistry Worksheet: Science Reasoning
Answer Section
SHORT ANSWER
1. heat trapped near Earth's surface
2. solar radiation
3. reflected sunlight
4. greenhouse effect
5. 7
6. 202 amu
7. 196 amu
8. 204amu
9. 196 amu
10. 201 amu
11. Mercury
12. oxygen
13. carbon dioxide
14. growth
15. growth
16. Any changes could have been due to one of them and not the other and you would not know which one it was that did it.
17. Yes
18. qualitative
19. Distilled water
20. baking soda and cornstarch
21. vacuum
22. air pressure on the Hg in the bowl is lower at higher elevation
23. the Hg in the tube will be higher due to increase pressure
24. air pressure on surface of Hg in bowl
25. air will enter tube and Hg will run out