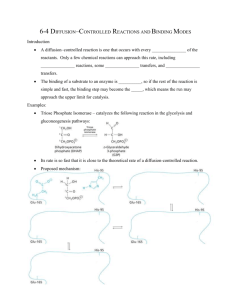
Purification and characterization of variants of
advertisement

Purification and characterisation of variants of Trp278 and Trp279 in the surface site of barley -amylase Ahmet Alsac s002135 July 2005 1 CHAPTER 1 LIST OF ABBREVATIONS 3 CHAPTER 2 Summary Preface Introduction 4 4 5 6 CHAPTER 3 THEORY 7 CHAPTER 4 MATERIALS AND METHODS Method Scheme 4.1 Expression of foreign genes in Pichia pastoris 4.2 Expression and purification of enzyme variants 4.2.1 Shake flask cultivation 4.2.2 Purification 4.3 Characterization of proteins 12 12 13 15 15 17 20 CHAPTER 5 RESULTS AND DISCUSSION 5.1 Expression 5.2 Purification 5.3. Characterization 33 33 35 39 CHAPTER 6 CONCLUSION 43 BIBLIOGRAPHY 44 CHAPTER 7 APPENDIX Appendix 1 Expression and purification overview Appendix 2 Affinity Chromatogram Appendix 3 DP440 Appendix 4 Cl-pNPG7 Appendix 5 Starch Binding Appendix 6 Stock Solutions for Media Preparation Appendix 7 Coomassie blue and silver staining 46 46 49 51 53 55 57 58 2 Chapter 1 List of Abbrevations A: Alanine AMY1: barley -amylase low pI isozyme 1 AMY2: barley -amylase high pI isozyme 2 AMS: Ammonium sulfate BMGY: Buffered glycerol complex medium BMMY: Buffered methanol complex medium Cl-pNPG7: 2-Chloro 4-nitrophenyl maltoheptaoside DMSO: Dimethylsulfoxide DP440: Degree of polymerization 440 DP17: Degree of polymerization 17 OD: Optical Density MD: Minimal dextrose medium MES: 2-morpholine ethansulfonicacid rpm: revolutions per minute Trp:Tryptophan W: Tryptophan wt: wild type YPD: Yeast extract peptone dextrose medium. YNB: Yeast nitrogen base with ammonium sulfate without amino acids 3 Chapter 2 Summary Variants of barley -amylase isozyme AMY1 were expressed, purified and their kcat, Km and Kd values were determined. Two single mutants one mutated at tryptophan at position 278 (W278A) and one mutated at tryptophan at position 279 (W279A), the double mutant (W278A/W279A) and wild type (wt) were expressed form the yeast Pichia pastoris using shaker flasks. The variants of proteins were purified from the culture supernatants by ultrafiltration and affinity chromatography on -cyclodextrinsepharose. For determination of kcat and Km the substrate amylose DP17, amylose DP440 and 2-chloro-4-nitrophenyl D--maltoheptaoside (Cl-pNPG7) were used. There were no significant differences in these kinetic values between the wt and the single mutants. For determination of the dissociation constant Kd the binding to barley granules was used. Here was observed a difference between the wt and the single mutant. Thus single mutants have higher Kd than the wt. W278A has a Kd of 2.67 (mg/ml), W279A has a Kd of 3.14 (mg/ml) and wt has Kd of 0.70 (mg/ml). The double mutant gave the same kinetic values as the wt. This was not an expected result and probably variant used was not mutated at the right position. 4 Preface This report is based on experimental research made in the Biochemistry and Nutrition Group (BNG) of BioCentrum, Technical University of Denmark. The work for the report corresponds to 30 ECTS points and it is the final project for the civil engineer study. This study was limited and had a duration of 5 months, beginning 1. February 2005. My supervisors were Henrik Næsted and Birte Svensson. 5 Introduction -Amylases from cereals are involved in mobilization of seed storage starch. They are essential in the production of beer and other beverages and in the baking industry. Designer enzymes having improved specificity, activity, or synergy with enzyme partners in the biotechnological area provide important prospects. Finding domains with influence on -amylase binding to carbohydrates via mutation and engineering of the domain surface binding site in barley -amylase would most probably enhance their catalytic power on raw starch and related substrates. Combining the catalytic properties of, for example psychrophilic -amylases, which hydrolyze polysaccharides at low temperature, and the capacity of enhanced starch binding of barley -amylase isozyme 1(AMY1) could result in an -amylase with high efficiency on starch at low temperature. amylase has three binding sites for binding of starch; one at the active site, one at the so called “old” starch granule binding surface site and one at the so called sugar tongs. I have expressed and purified AMY1 wt and three variants, mutated at “old” starch granule surface binding site, of which determination of kinetic profiles reveal the importance of these mutations and how important the “old” starch granule surface binding site is for the function of AMY1. The amino acids that are mutated are tryptophan at position 278 replaced with alanine, W278A, and tryptophan at position 279 replaced with alanine, W279A, and the double mutat W278A/W279A. It is known that these tryptophans are the functional residues that bind starch so it is expected that mutation of these residues will result in a decrease in affinity towards starch. 6 Chapter 3 Theory -amylases (1,4- -D-glucan glucanohydrolase) are monomeric enzymes widely occurring in animals, plants, and microorganisms. They catalyze the hydrolysis of internal -D(1,4)-glucosidic linkages in starch [1] (amylose and amylopectin), glycogen, and related oligo- and polysaccharides to produce maltodextrins, maltooligosaccharides, and glucose (fig. 3.1). Most of the glucose residues in starch are unbranched and are linked by -D(1,4)-glucosidic bonds. The (1,4) linear molecule in starch is called amylose. The branches are formed by -D-(1,6)- glucosidic bonds which occur for approximately every thirty -D-(1,4)-glucosidic linkage, this molecule is called amylopectin. The branches serve to increase the solubility and make its sugar easily accessible. For glycogen the -D(1,6)- glucosidic bonds occur about once in ten units. Therefore glycogen is highly branched and more soluble than starch [1]. Seed germination [2] is triggered by an increase in temperature and humidity and causes the embryo to synthesize gibberellic acid, which induces de novo synthesis of -amylase and an array of other hydrolases from the living outer tissue of barley seeds called aleurone layers(fig. 3.2). These activated hydrolases release sugar units from starch in endosperm. The progressive release of sugars from the storage starch provides energy to the growing embryo until the plant can feed itself by photosynthesis. In germinating barley seeds, different -amylase isozymes, encoded by two multigene families and referred to as AMY1 and -amylase isozyme 2 (AMY2), are distinguished. The two isozymes, which contain 414 and 403 amino acid residues, respectively, display 80% sequence identity, and are distantly related to -amylases from microorganisms and animals. AMY1 has a molar weight at 45.5 kDa. Despite the high sequence identity, AMY1 and AMY2 show distinctly different physicochemical and biochemical properties. AMY1 is known as the low-pI (pI 4.9) isozyme, and AMY2 is known as the high-pI (pI 5.9) isozyme. AMY1 has highest affinity for calcium ions [3], is the most stable at acidic pH, and is the least stable at elevated temperature. 7 Figure 3.1: Overview of the structure of starch. Above the building units and organization of starch. Below a diagram showing how the side branching chains are clustered together within the amylopectin molecule. 8 Figure 3.2: Mechanisms of release of sugar into the embryo from endosperm. (1) The endosperm signals via gibberellic acid that it needs carbohydrates. (2) The signal is received in aleurone which then activates carbohydrate degrading enzyme. (3) Enzymes degrade the starch to sugar units in endosperm. (4) Sugar units are consumed by embryo While AMY2 is also most stable in urea at pH 6.7, AMY1 has highest stability in urea below pH 6 or in the presence of NaCl. Moreover AMY1 is most stable in guanidinium chloride. Charge screening thus destabilizes AMY2 but stabilizes AMY1. Isozyme sequence comparison suggests that AMY1 lacks four of the 20 salt-bridges identified in the crystal structure of AMY2. Moreover, AMY1 has the highest affinity and activity toward starch granules, whereas on soluble substrates AMY1 still has the highest affinity, 9 but AMY2 has the highest turnover rate. Finally, a most remarkable difference is the unique capacity of AMY2 in binding the endogenous bifunctional inhibitor BASI [4] (barley -amylase/subtilisin inhibitor). AMY1 contains three domains [5]. A major central domain with a parallel (/)8 barrel super secondary structure called domain A. An irregular loop of 65 residues bulges in domain A between 3 and 3, called domain B. Finally, the 61 residue long loop at the Cterminal is organized as a five – stranded antiparallel -sheet, called domain C AMY1 has three binding site areas. The “sugar tongs” in domain C, the “old” starch granule binding site on the surface of domain A and the binding site at the active site. It has been shown by site-directed mutagenesis and crystallography, that AMY1 contains a separate surface binding area on the basis of two contiguous tryptophans [5], Trp278 and Trp279 (Trp276 and277 in AMY2) (fig. 3.3). This site has low affinity for acarbose (Kd=5mM) and binds -cyclodextrin [5]. The binding of -amylase to -cyclodextrin is used in affinity chromatography purification. No mutant was previously obtained at Trp279Ala which is invariant in cereal -amylases. Sequence comparison suggests that this starch binding site is unique to -amylases from higher plants, whereas other surface binding sites are reported in certain microbial or mammalian enzymes [5]. Calcium ions at the active site are critical for folding and conformational stability and hence for the enzyme activity [3]. The starch granule binding surface binding site from the structure of AMY1/4I,4II,4IIItrithiomaltotetraoside (thio-DP4) is shown at fig. 3.3. It is seen that the two rings from the thio-DP4 stack onto indole rings of a pair of tryptophans, Trp278 and Trp279. This site is called the starch granule binding surface site. The two rings of thio-DP4 form six hydrogen bonds to Trp278 and neighboring residues in the structure [5].The angle between the planes formed by the two indole rings in Trp278 and Trp279 is around 135 and is constant between AMY1 and AMY2 in both native and complexed structures. The closer the planes determined by the two indole rings and the plane of sugar rings the stronger the forces in the hydrophobic stacking. The surface-binding site is suggested to possess the special capacity of selecting substrates according to their geometrical characteristics probably governed by the surroundings of the site. The two tryptophans perform a locked conformation because of a very tight packing with neighboring 10 residues. This structure constitutes a geometric filter that favors binding of structurally complementary molecules. Because the granular starch binding site and sugar tong are distant to the active site they may play a more important role when the substrate is longer and minor role when the substrate is shorter. When the substrate is long the two binding sites force it in a loop structure around the enzyme. Figure 3.3: Overall Structure of AMY1 in Complex with Thio-DP4. Calcium ions, spheres. The three upper calcium ions are those found both in AMY1 and AMY2 (Ca500, Ca501, and Ca502). The fourth Ca503 is located closer to the center, where it occupies the active site. Thio-DP4 substrate analog fragments are shown as surface representations. To the left, the starch granule binding surface site on domain A is shown with the two tryptophans residues (Trp278 and Trp279). In domain C (bottom part of the figure), an entire thio-DP4 molecule is curved around Tyr380 at the level of the sugar tongs. 11 Chapter 4 Materials and Methods Method Scheme Expression from P. pastoris in shake flask Purification Ultra filtration Affinity chromatography Protein characterisation kcat Km :AmyloseDP17, AmyloseDP440 and Cl-pGNP7 Kd: Starch granule binding 12 4.1 Expression of foreign genes in Pichia pastoris The insertion of gene in the host was not a part of my project, but I will tell the background for how it is done. The methylotrophic yeast Pichia pastoris is the yeast specie selected for its advantage over S. cerevisiae. Two attributes are critical in its selection: the existence of fermentation methods and presence of methanol regulated promoters [6]. The growth medium is a defined mixture of salts, trace elements, biotin and carbon. They are basic and cheap sources. Biochemical studies revealed that methanol metabolism required the induction of set of metabolic pathway enzymes. The most interesting is alcohol oxidase (AOX), the first enzyme in the methanol utilization pathway. AOX is undetectable in cells cultured on carbon sources like glycerol, but consists 30% of total soluble proteins in methanol-grown cells. Under control of an AOX promoter, foreign genes could be maintained in an expression-off mode on a non methanolic carbon source to minimize selection for non-expressing mutant strains during cell growth, then efficiently switched on by shifting to methanol. This expression system will be utilized to produce -amylase. The goal of the expression effort is the secretion of the foreign protein. Secretion requires the presence of a signal sequence to target the protein into the secretory pathway. The secretion signal sequence from the Saccharomyces cerevisiae factor prepro peptide has been used most successfully. The principle of insertion of a foreign gene in the host Pichia pastoris is shown at figure 4.1. The gene is inserted in the vector under control of AOX promoter. The vector contains also a wild type gene for histidine. The host’s histidine gene is modified bysite directed mutagenesis, so the host can not produce histidine and is therefore His−. If the insertion is succeeded the host will be His+. The inserted gene will only be expressed when there is methanol in the culture. 13 Figure 4.1: Vector insertion of foreign gene in Pichia pastoris where histidine gene provides the selection control For transferring the amylase gene to the host genome the plasmid pPICZA [7] is used. At figure 4.2 the construction of the plasmid is seen. 14 pUC origin ApaLI (4179) AOX1 promoter ApaLI (623) CYC1 transcription terminator ApaLI (3509) 5' AOX1 primer HindIII (873) Zeo(R) SmaI (3254) EcoRI (944) pP I C ZA A M Y 1 delta9 novel AvaI (3252) NcoI (949) 4593 bp XmaI (3252) AvaI (3242) NcoI (3162) EM7 promoter AMY1d9 AvaI (1616) TEF1 promoter BamHI (2678) 3' AOX1 primer 6xHis AvaI (2246) c-myc epitope Figure 4.2: Construction of amylase gene (AMY19) in combination with AOX promoter in the plasmid pPICZA. 4.2 Expression and purification of enzyme variants 4.2.1 Shake flask cultivation The expression of recombinant proteins like amylase in Pichia pastoris requires the preparation of several different media [8]. Recipes of these media start with preparation of stock solutions (Appendix 6). For the growth and selection of P. pastoris YPD agar plates are used. Agar plates are done by 1 L of Yeast Extract Peptone Dextrose Medium (YPD) which is made by dissolving 10 g yeast extract, 20 g of peptone and 20 g agar in 900 ml H2O. This solution is autoclaved and then 100 ml of 10X dextrose is added. This medium is then immediately transferred to agar plates and cooled at room temperature. Plates are stored at +4C. Species of 3 mutants (Trp278Ala, Trp279Ala and Trp278Ala/Trp279Ala) and wt of P. pastoris from -80C were streaked out on YPD plates and grown at 30C for 48 hours for best cultivation. Streaks were taken from the plates after growth and inoculated in MD medium. Minimal Dextrose Medium (MD) is composed of YNB, biotin and dextrose. The wt is histidine 15 mutant, because the insertion of wt of AMY1 gene into the host did not went well. Therefore it can not grow in MD media hence BMGY media is preferred for wt. Cultures are inoculated in 3 ml media for 24 hours at 30C at 190 rpm. MD is prepared by making an 20 ml stock solution where 2 ml 10X YNB, 40 µl 500X biotin and 2 ml 10X dextrose are mixed in autoclaved H2O with total volume up to 16 ml. The composition of BMGY is given below. The culture from MD medium was next transferred to BMGY to get higher cell densities. 1 ml of MD medium of every type is transferred to BMGY medium. To get an idea of the growth profile of the metabolism of P. pastoris a pilot scale experiment with small volumes is done. The culture was transferred to 25 ml fresh BMGY medium in a 250 ml flask and grown at 30C for 24 hours at 190 rpm. The composition of 25 BMGY is 0,25 g yeast extract and 0,5 g peptone autoclaved in 17,5 ml H2O and then mixed with 2,5 ml 10X YNB, 2,5 ml 10X glycerol, 2,5 ml 1 M potassium phosphate buffer pH 6 and 50 µl 500X biotin. After 24 hours 5 ml of the cultures are transferred to 200 ml BMGY in 1 L flask and grown at 30C with 190 rpm for 24 hours. After 24 hours a sample from the culture was taken to check the OD600nm. The OD600nm should be around 1 when BMGY culture resuspended in 100 ml BMMY (as BMGY except for 0.5 % methanol replacing glycerol). The volume of BMGY culture, when resuspended in BMMY gives 1, was then taken and the medium was replaced by 100 ml BMMY in 1 L flask for induction of the recombinant protein for 24 hours at 30C. After growth the supernatant was isolated by centrifugation the culture (9000 g, 15 min,+4C), while the pellet was kept for another induction. During growth there were taken samples to measure OD600nm and the sample was centrifuged so the supernatant could be used for measuring activity of -amylase by Insoluble Blue Starch assay. The pellet was again resuspended in fresh 100 ml BMMY for another induction up to 48 hours. Samples were taken at various times for activity and OD600nm measuring. Insoluble Blue Starch activity was measured on Insoluble Blue Starch suspended (6.25 mg/ml) in 20 mM sodium acetate buffer, 5 mM CaCl2, 5 mg/ml BSA. The reaction was initiated by enzyme addition (around 10 µl) to the suspension (800 µL) at 37C and stopped after 15 min by addition of 0.5 M NaOH (200 µL). After centrifugation (4 min, 13 000 g) supernatants were transferred to a micro-titer plate (300 µL). A620 values (Ceres UV900 HDI micro-plate reader, Biotek Instruments,Inc., UK) in 16 the range 0.8-1.2 were used to calculate activity . One unit was defined as the amount of enzyme that during 15 min reaction resulted in an increase in A620 of 1 in the supernatant of the stopped reaction mixture. This formula to get the units of -amylase per ml supernatant: Units/ ml= ((A620 − Ablank)/ (5 volume sample)) dilution factor 1000 Units per ml can be converted to mg amylase per ml because we know the value of 2900 U/mg for the purified AMY1 wt [9]. According to the data for OD and activity versus time, the large volume production of recombinant protein started. First all variants were grown on MD and BMGY for wt as in pilot scale then transferred to 25 ml BMGY media after 1 day, after growth for one day again, this culture was transferred to fresh 0.8 L BMGY media in 3 L flask and incubated for 24 hours at 30C and shaken at 145 rpm. After end of culture period the OD600nm was recorded and volume of BMGY was resuspended in 0.4 L BMMY media inoculated.The inoculated BMMY should have an OD600 = 1. The culture was grown at 30C at 195 rpm for 24 hours. After 24 hours the cultures were centrifuged at 9000 rpm and 4C for 15 min. The supernatant was kept, while the pellet was induced for another 48 hours in BMMY. Like before the cultures were centrifuged, the supernatants were combined with the previous supernatants and added 0.02% (w/v) sodium azide for germination protection. The activity, OD280 and volume were recorded (See APPENDIX 1). The pellets were discarded. The OD280 measures the aromatic residues of tryptophan and tyrosine, thus representing the total content of protein in the sample. By dividing OD280nm with extinction coefficient of amylase the content of amylase in a culture can be estimated. The extinction coefficient of -amylase for OD280nm is 2.4 for 1 mg/ml [10], so the concentration of -amylase can be found from OD280nm measurements. 4.2.2 Purification After production of variants of amylase, they were purified from the culture supernatant. First the supernatants were filtered through a 0.45 µm membrane to remove larger particles. The first step in purification was ultra filtration (fig. 4.3). Using a membrane of 17 cut-off at 10.000 kDa. The sample was applied and collected in the retentate, while the permeate was collected to check for content of -amylase. All purification steps were carried out at 4C. The second step in purification is affinity chromatography. First, the sample Figure 4.3: Overview of the principle for ultra filtration solution must be conditioned for to binding the enzyme to the gel material. This was done by adding 5% (w/v) ammonium sulfate (AMS). The samples were purified on cyclodextrin-Sepharose (ÄKTA explorer automated chromatograph). Affinity chromatography is a unique separation technique since it enables purification of almost any biomolecule on the basis of its biological function or chemical structure. It is a type of adsorption chromatography in which the molecule to be purified is specifically and reversibly adsorbed by a complementary binding substance (ligand) immobilized on an insoluble support (matrix). The dissociation constant (Kd) for the ligand binding substance complex should ideally be in the range 10-4 to 10-8 M to avoid too weak or too strong interactions [11]. It is also important that the immobilized ligand retains its specific binding affinity for the substance of interest (S) and the methods are available for selectively desorbing the bound substance in an active form, after washing away unbound material. Purification is often of the order of several thousand fold and recoveries of 18 active material are generally very high [11]. After preparation of the ligand matrix complex, affinity medium, the substance can be purified as follows. Affinity medium is equilibrated in binding buffer. The absorbance will be stable baseline (see chromatograms at Appendix 2). Sample is applied under conditions that favor the binding of the target molecule to the ligand. This will result in increased absorbance. Target molecules bind specifically but reversibly to the ligand and unbound material washes through the column. Washing will result in decrease of absorbance to stable baseline. Target protein is recovered by changing conditions to favor elution of the bound molecules. If any target protein is recovered we will se a peak in absorbance. Elution is performed non specifically by changing the pH, ionic strength, polarity or specifically by a competing protein. Target protein is collected in a purified, concentrated form and the affinity medium is re-equilibrated with binding buffer. For practical work first buffers were prepared: The equilibration buffer is Buffer A, (20 mM sodium acetate, 25 mM CaCl2, pH 5.5, the washing buffer is Buffer B (20 mM sodium acetate, 25 mM CaCl2, 200 mM NaCl, pH 5.5). The elution buffer is Buffer C (8 mg/ml -cyclodextrin in 20 mM sodium acetate buffer, 25 mM CaCl2, pH 5.5). All buffers are filtered and degassed to avoid dirt and air in the chromatography. Around 5 ml CNBr activated Sepharose coupled with -cyclodextrin is packed in a column. Sepharose is a bead-formed agarose gel. The hydroxyl groups on the sugar residues can be used for covalent attachment of a ligand. CNBr activated Sepharose enables ligands containing amino groups to be immobilized. The column pressure limit was set to 0.30 Mega Pascal and UV absorbance was done 280 nm. This column is equilibrated with buffer A (4 column volumes) and the sample was applied to the column at flow rate at 1 ml/min, followed by washing (4 column volumes Buffer B) to get stable baseline. The bound protein was then eluted with buffer C and the first peak appearing was collected in 12 ml tubes as 0.5 ml fractions and added 0.02% (w/v) sodium azide. All purification steps were carried out at 4C and SDS-Page was made for each purification step and stained by colloidal coomassie blue and silver (Appendix 7 for detailed method description). After purification -cyclodextrin was removed by dialysis. Bag tubes with molecular cut off at 6000 – 8000 kDa were used. Variants of proteins in dialysis tubes were dialyzed 19 against (1 mM MES, 25 mM CaCl2 and pH 6.8) under gentle stirring for 3 days with buffer changed every day. At fourth day the buffer was changed to 10 mM MES, 25 mM CaCl2 and pH 6.8. 4.3 Characterization of proteins Enzyme (E) catalyzed substrate (S) conversion reactions can be best described by the following reaction scheme [12]: k ES 1 k2 k1 ES E P ( 4.1) The scheme shows that the reaction velocity will be proportional to the concentration of the enzyme substrate ES complex as v = k2[ES]. To put these schemes in to mathematical framework we use Henri-Menten-Michaelis approach, which assumes a rapid equilibrium is established between (E + S) and the complex ES followed by slower conversion of ES back to free enzymes and substrate, thus: k2 k-1. The free enzyme Ef combines with S to form ES. The equilibrium dissociation constant for this complex is: Ks =([E]f [S])/ [ES] (4.2) where [E]f = [E] − [ES] (4.3) This can be arranged to: [ES] =(E][S])/(Ks + [S]) (4.4) A single chemical step defined by the first order rate constant k2 results in product formation. More likely there will be a series of chemical reactions following ES complex formation. These steps are described by the first order rate constant kcat: 20 Ks E S ES kcat E P(4.5) and the rate of product formation is given by the first order equation: v = kcat[ES] (4.6) Combining (4.4) and (4.6) we get: v = (kcat[E][S])/( Ks + [S]) (4.7) Equation 4.7 describes the reaction velocity as a hyperbolic function of [S] with a maximum value of kcat[E] at infinite [S]. This value is the maximum reaction velocity or Vmax: Vmax = kcat[E] (4.8) Combining this definition with equation 4.7 gives: v =(Vmax[S])/( Ks + [S]) (4.9) If the enzyme concentration is known the value of kcat can be calculated by dividing the experimentally determined Vmax by [E]. The units of kcat are reciprocal time in s−1. It provides a lower limit on the first order rate constant of the slowest step following substrate binding that leads to product release. Km is the substrate concentration that provides a reaction velocity that is half of the maximal velocity obtained under saturating substrate conditions or it represents the substrate concentration at which half of the enzyme active sites in the sample are filled or saturated by substrate molecules in the steady state. It is usually measured in concentrations in mM or mg/ml. The catalytic efficiency of an enzyme is best defined by the ratio of the kinetic constants: kcat/ Km. The ratio has units of a second order rate constant and is generally used to compare the efficiencies of enzyme variants. The kinetic constants Vmax and kcat are 21 determined graphically with initial velocity measurements obtained at varying substrate concentrations. The first way of graphing the data is plotting velocity as a function of [S]. For this purpose an excel calculation sheet is used to determine Km and Kcat from amylose DP440 and DP17 assays (Appendix 3). The absorbency for each initial time and substrate concentration is inserted in the sheet. Also a standard curve is determined and inserted in the sheet. The standard curve ensures that we have the corresponding absorbency at 540nm for a given concentration of the product. The increase in absorbency in the standard curve can be given as linear increase by determining the linear coefficients of plotting absorbency as function of known product concentration. The standard curves slope is used to determine product formation in the enzyme mix at various times. The initial determined OD540 for each substrate concentration at various times are inserted, then the slope of OD540/min can be determined. Dividing the slope with the slope of standard curve gives us the velocity in mM/min. Plotting reciprocal value of velocity as function of reciprocal value of concentration gives a linear line, which gives the possibility to determine Km and Vmax, equation 4.11.This plott is called Lineweawer Burk plot and was used before implementation of curve fitting programs. When enzyme kinetic data is generated it can be linearized by using the method of Linewear and Burk: v =(Vmax[S])/( Km + [S]) V max (4.10) Km 1 S If reciprocal of this equation is taken we get: 1 Km 1 1 (4.11) v V max Km V max We see that 4.11 is an equation for a straight line with slope of Km/Vmax and y intercept of 1/ Vmax, when the reciprocal of the velocity is plotted as function of the reciprocal of [S]. Plotting velocity as a function of concentration gives us a hyperbolic curve a so called Michaelis Menten plot. Usually curve fitting programs are used to generate line through the data. The program Curve Expert is used and the function y = (ax)/ (b+x), called growth saturation mode, is applied to curve fitting. The outcome of curve fitting is 22 the coefficients of the curve, where the coefficient a divided with enzyme concentration is Kcat and b corresponds to Km. Catalytic parameters were determined toward amylose DP440 (average degree of polymerization 440). This is done by first making amylose stock solution. 100 mg amylose is stirred for 30 min with 1.6 ml dimethylsulfoxide (DMSO). To this solution H2O is added up to 12 ml and then 3.5 ml concentrated buffer (100 mM sodiumacetate pH 5.5) and 18 µl 5% BSA (Bovine Serum Albumin) and 45 µl 2 M CaCl2. Finally H2O is added up to 18 ml. Also buffer with DMSO and buffer without DMSO were made.Buffer with DMSO were done by adding 62.5 µl 2 M CaCl2, 25 µl 5% BSA and 2.22 ml DMSO to 5 ml concentrated buffer and finally adding H2O to 25 ml. Buffer without DMSO is made by adding 100 µl 2M CaCl2 and 40 µl 5% BSA to 8 ml concentrated buffer and finally H2O up to 40 ml. After these solutions are made the series of substrate solutions are prepared (tab. 4.1) Then a standard series is prepared from a maltose stock containing 0.265 mM of maltose monohydrate (tab. 4.2) From amylose substrate solution 990 µl is taken and every solution is incubated at 37C for 5 min. On precise time 110 µl of enzyme solution (containing 8 nM variant of AMY1) is added to substrates. Then 200 µl is taken out at exactly 2.5 min up to 12.5 min and added to 500 µl reducing sugar assay reagents and 300 µl H2O, called STOP solution. The stop solution is made by two reducing sugar assay reagents. Solution A is made by dissolving 27.4 g Na2CO3 and 12.0 g NaHCO3 in 400 ml H2O then 971 mg disodium 2.2-bicinchionate and added H2O to 500 ml. 23 Table 4.1: Amylose substrate solution No Mg/ml Amylose stock (ml) Buffer with DMSO (ml) Buffer without DMSO 1 2.5 3 0 3 2 2.0 2.4 0.6 3 3 1.5 1.8 1.2 3 4 1.0 1.2 1.8 3 5 0.75 0.9 2.1 3 6 0.5 0.6 2.4 3 7 0.4 0.48 2.52 3 8 0.3 0.36 2.64 3 9 0.2 0.24 2.76 3 10 0.1 0.12 2.88 3 Table 4.2: Maltose substrate solution No Maltose mM Maltose stock (µl) µl H2O 1 0 0 2000 2 0.0158 120 1880 3 0.0211 160 1840 4 0.0317 240 1760 5 0.0528 400 1600 6 0.106 800 1200 This solution must be protected from light, Solution B is made by 620 mg CuSO4 5 H2O and 630 mg L-serine dissolved in 500 ml H2O. This solution is kept at 4C in the dark. Solution A and B are mixed in equal amounts before use. From maltose stock solution 500 µl aliquots are transferred into Eppendorf vials containing 500 µl reducing sugar assay reagents. All samples are as quickly as possible heated at 80C for 30 min, then quickly cooled and centrifuged at 14000 rpm for 5 min. 300 µl aliquots are transferred to a micro titer plate and the absorbency is read at 540 nm in an ELISA reader. 24 It is also possible to determine catalytically parameters toward amylose substrates with shorter degree of polymerization. It gives us the opportunity to compare the efficiency of variants of -amylase towards shorter and longer amyloses. In this case amylose DP17 (average degree of polymerization 17) reducing sugar assay is used. The substrate is made by dissolving 111.1 mg DP17 amylose (Amylose Ex-1, Hayashibara Bichemical Lab) in 10 ml buffer. The buffer contains 20 mM NaOAc, 5 mM CaCl2 pH 5.5. This solution is then heated in microwave for 30 second with effect of around 500 W to avoid boiling. Then 10 µl 5% BSA is added. From this stock the following concentrations are made by dilution in the mentioned buffer (Tab. 4.3). Then a standard series of maltose is made. Table 4.3: Amylose DP 17 substrate conc. Final No Concentration (%) 1 1.0 2 0.7 3 0.5 4 0.3 5 0.2 6 0.1 7 0.08 8 0.05 9 0.03 10 0.01 A stock of maltose at 200 µg/ml is diluted with the mentioned buffer to get these concentrations (Tab. 4.4). Every standard concentration is mixed with 500 µl reducing sugar assay reagents as used for DP 440. From amylose substrate solution 900 µl is taken and every solution is incubated at 37C for 5 min. 25 Table 4.4. Maltose conc. for amylose DP 17. No Conc. in µM 1 55.5 2 41.62 3 27.75 4 20.8 5 13.87 6 6.93 7 0 On precise time 100 µl enzyme solution (containing 20-50 nM variant of AMY1) is added to the substrates. Then 10 / 50 µl samples are taken out to Eppendorf tubes with 490 / 450 µl plus 500 µl reducing sugar assay reagents with exactly 2.5 min intervals up to 12.5 min (10 µl for conc. 0.2-1 % and 50 µl for conc. 0.01-0.1%). Both amylose and maltose samples are heated for 30 min at 80C, then quickly cooled and centrifuged at 14000 rpm for 5 min. 300 µl aliquots are transferred to a micro titer plate and the absorbency is read at 540 nm in an ELISA reader. Another substrate of determining kcat and Km is pNPG7. The hydrolytic activity of the amylase for p-nitrophenyl maltoheptaoside (pNPG7) was determined for the variants of enzymes using amylase assay kit. The parameters kcat and Km were calculated from the initial rates at the 0.25 - 10 mM substrate concentration range. Buffers and enzymes are made in concentrations so there is enough for kinetic determination in triplicate. First a 50 ml kit buffer is prepared by mixing 25 ml 0.1 M phosphate buffer with 0.1864 g KCl and 500 l 2% NaN3 and H2O up to 50 ml. Then “buffer F12” is made by adding 15 l 5% BSA into 15 ml kit buffer. Than two enzyme solutions are made. 15 mg glucosidase (over 70 Units/ml) in 1.5 mg “F12 buffer”, and 0.89 mg -glucosidase (20-40 Units/ml) in 700 l “buffer F12”. Then a of Cl-pNPG7 series is made, as shown at table 4.5. 26 Tabel 4.5. Cl-PNG7 substrate solution concentration series. No mM Cl-PNG7 -gluc. -gluc. Buffer F12 1 0.25 5 mM 36l 175 72 360 2 0.5 5 mM 72l 175 72 324 3 1 5 mM 144l 175 72 252 4 2 5 mM 288l 175 72 108 5 3 50 mM 43.2 l 175 72 353 6 5 50 mM 72 l 175 72 324 7 8 50 mM 115.2 l 175 72 280 8 10 50 mM 144 l 175 72 252 Then a standard solution concentration of Cl-pNP is made. 1.7 mg is transferred to 1 ml F12 buffer to get a concentration at 10 mM, this solution is diluted 20 times to get a concentration of 0.5 mM. The following concentrations are made, see tabel 4.6 Table 4.6. Standard curve for Cl-pNPG7 l Cl-pNP 0,5 mM l buffer F12 Final conc. mM 0 200 0 40 160 0.1 80 120 0.2 120 80 0.3 160 40 0.4 200 0 0.5 These standard solutions are directly transferred (100 l) to microtiter plate. Each substrate mixture (90 l) is transferred to a microtiter plate where they are pre incubated for 2 min at 30 C. To this mixture 10 l enzyme solutions are added. Enzymes are diluted in F12 buffer to get concentrations in 30- 50 mM. The p-nitrophenol release is determined spectroscopically at 405 nm with 7 readings from 2 to 8 minutes. To determine Km and Kcat an excel calculation sheet is used (Appendix 4). First the linearity of OD405 as function of concentration of Cl-pNP in mM is plotted. The slope (OD/mM) is 27 determined and used for transform the inserted enzyme substrate mix OD values from OD/min to the velocity in mM/min. Plotting velocity as a function of concentration gives us a hyperbolic curve a so called Michaelis Menten plot. Usually curve fitting programs are used to generate line through the data. The program Curve Expert is used and the function y = (ax)/ (b+x), called growth saturation mode, is applied to curve fitting. The outcome of curve fitting is the coefficients of the curve, where the coefficient a divided with enzyme concentration is Kcat and b corresponds to Km. Now a background for protein - ligand binding equilibrium is given for determination of the equilibrium dissociation constant Kd. Enzymes catalyze the transformation of the substrate to product. To ensure this enzyme and substrate must form binary complex with each other. The macromolecular partner is called the receptor (R) and the smaller molecular partner in this binding is called the ligand (L). The total concentrations of ligand and receptor can be described as: [R] = [RL] + [R]FREE (4.12) [L] = [RL] + [L]FREE (4.13) The position of the equilibrium between these partners is most commonly quantified in terms of the dissociation constant, Kd, for the binary complex at equilibrium [13]. Kd = ([R]free[L]free )/ [RL] (4.14) The tighter the ligand binds, the lower the value of the dissociation constant. The dissociation constant can be related to the Gibbs energy of binding for the receptor – ligand complex as follows: Gbinding = RTln(Kd) (4.15) The equilibrium constant can also be approached by determination of kinetic rates. The second order rate constant for complex association can be defined as kon and the first order rate constant for complex dissociation as koff.: 28 koff R L Kon RL (4.16) The equilibrium dissociation constant for the complex is thus given by the ration of koff to kon: Kd = koff / kon (4.17). At equilibrium the concentrations of the RL is complex constant. Hence the rates of complex association and dissociation are equal. d RL k on R f L f (4.18) dt d RL k off RL (4.19) dt combining eq. 4.18 and eq. 4.19 gives kon[R]f[L]f = koff [RL] (4.20) or (kon /koff ) [R]f[L]f = [RL] Knowing that kon/koff is equivalent to the equilibrium constant Ka thus Ka = 1/ Kd : Ka [R]f[L]f = [RL] (4.21) We prefer to work with an equation in terms of total added ligand and receptor that are easier determined than concentrations of free ligand or receptor. If we apply eq. 4.11 on eq. 4.21 and dividing both sides by 1+([RL]/[R]f we obtain: 29 R f R (4.22) RL 1 R f The fraction [RL]/[R] is referred to as the fractional occupancy of the receptor and is often represented by the symbol B (for bound receptor). Using Ka=[RL]/[R]f[L]f on eq.4.22 we obtain: RL K a L f R 1 K a L f (4.23) or, RL R L f K d L f (4.24) Under most conditions the concentration of receptor is less than that of the ligand. Therefore the formation of complex does not significantly diminish the concentration of free ligand thus: [L]f [L] (4.25) Eq. 4.24 can be rewritten as follows RL RL RK (4.26) K d L 1 d L Eq. 4.26 describes a hyperbola that is typical of saturable binding in biochemical situations. The equation is known as the Langmuir isotherm equation. Plots of [RL] as a function of total ligand concentrations are referred to as binding isotherms. By applying non-linear least squares fit to the data estimates of Kd can be obtained. For determination of the equilibrium dissociation constant the method of Starch Binding assay is used. In this assay the absorbency of free unbound -amylase is read. The free 30 concentration of the enzyme will fall if it binds to starch and therefore we will se a diminish in the absorbency. For this purpose the absorbency of enzyme mixed with various concentrations of starch is read. First a buffer DB is made of 20 mM sodium acetate, 5 mM CaCl2 0.005 % BSA at pH 5.5. Suspensions of barley starch granules are prepared. First a Mother 1 suspension is made by dissolving (after two washes) 800 mg barley starch granules in 10 ml DB buffer giving 80 mg/ml. Some of this suspension is diluted to 40 mg/ml and called Mother 2. Again some of Mother 2 is diluted to 4 mg/ml called Mother 3. Enzymes to be investigated are diluted in DB buffer to 100 nM in a total volume of 1.5 ml. Then following (table 4.7) mixtures are made at 4C. Table 4.7. Enzyme solution for starch binding analysis. No A B C D E F G H I J K L M N Starch 40 30 20 10 5 3 2 1.5 1 0.7 0.4 0.2 0.1 0 Enzm. 100 100 100 100 100 100 100 100 100 100 100 100 100 100 DB 400 525 400 650 775 825 400 525 650 725 800 850 875 900 500 375 500 250 125 75 500 375 250 175 100 50 25 0 mg/ml buffer Moth1 Moth2 Moth3 The enzyme solutions are incubated at 4C for 30 minutes by shaking to ensure binding reaction. After time the tubes are centrifuged at 13.000 rpm for 5 minutes at 4C. 750 L of the supernatant is transferred to new tubes on ice. To measure the activity a substrate solution of Insoluble Blue Starch with 10 mg/ml in DB buffer is prepared. For each tube at tabel 4.7 tubes with 500 l of substrate solution is prepared. Both the substrate and enzyme solution are placed at 37 C in a heating block for 15 min. After time 300 L enzyme solutions are added to the substrate solutions and mixed carefully. After exactly 15 minutes 200 L 0.5 M NaOH solution is added and mixed carefully to stop the reaction. Centrifuged for 4 min at 13.000 rpm and transferred to wells of a micro titer with volumes of 300 L and the absorbency is measured by ELISA reader at 620 nm. To determine the KD and number of binding site an excel calculation sheet is used 31 (Appendix5). OD620 for every enzyme substrate concentration is inserted in the sheet. The bound receptor to the ligand can be found by extracting the value of OD620 at zero concentration of starch, where all enzyme is free called E0, from the initial substrate concentrations OD620. These values can be transformed to fractional occupancy B by dividing with the E0. Plotting B as function of ligand concentration will give a hyperbole and applying the curve fitting with saturable growth model (y = a/(1+(b/x))) will give data coefficients where the coefficient a corresponds to Kd and b corresponds to the number of binding sites. 32 Chapter 5 Results and Discussion 5.1 Expression To get an idea of the production of -amylase from Pichia pastoris a pilot scale production was performed in shake flask. There were removed samples for activity measurements towards Insoluble Blue Starch, the data was outlined vs. time, see table 5.1: Table 5.1: Activity vs. time Mutant Time (h) Activity Units /ml W278A 0 0 W279A 0 0 W278A 24 24,34 W279A 24 42,64 W278A 48 1,50 W279A 48 2,74 From table 5.1 it is seen that maximum is reached about 24 hours and minimum level is reached around 48 hours. It was from here decided that the first induction with methanol should last between 24 hours up to 30 hours and the second induction should be around 40 hours and not longer than 48 hours. Then the first induction at large scale volumes were done for W278A and W279A. The result is shown on curve 5.2. After that a second induction at large scale was done. The result is shown on curve 5.3. 33 Curve 5.2 Activity vs time first induction 30 Units/ml 25 20 W278A 15 W279A 10 5 0 3 23 25 27 29 Time (h) Curve 5.3 Activity vs time second induction 50 Units/ml 40 30 W278A 20 W379A 10 0 1 20 22 24 43 45 47 Time in hours I have only measured the progress of activity values for the mutants W279A and W278A. From the curves it is observed that the concentration of -amylase peaks at around 24 hours for both variants than stabilize for the coming hours. Therefore for wt and the double mutant the first induction should be stopped after 24 hours to obtain maximum concentration. For wt and the double mutation variant I have only measured the final values found to 1.45 and 20.66 Units/ml respectively (Appendix 1). 34 5.2 Purification Table 5.4: Purification overview for Trp278Ala Fraction A280 Total activity in Units Recovery in (%) Specific activity Units/A280 Medium 15584.8 15225.6 Ultrafiltration 4035.0 12312.0 80.9 3.1 Affinity Chromat. 7.8 6547.8 53.2 836.5 Dialyze 4.46 4512 29.6 1012.4 Blue Starch activity U/mg 1.0 2450 For detailed background of purifacation overviews see Appendix 1. From table 5.4 the steps for expression and purification of the mutant W278A is seen. After ultra filtration 20% of the protein is lost. It is common to tolerate loss around 20% per purification step. After affinity chromatography around 27% is lost while the specific activity increases. The reason is that OD280 gets very lower because the only protein in the sample is amylase and affinity chromatography is thus a very efficient purification step. OD280 was very high in medium and ultra filtration due to the high content of various proteins peptides. After dialyzation around 23% is lost and that is much because dialyzation is not really a purification step. It is a method to remove bound -cyclodextrin from the amylase. So perhaps the period of dialysis should be shortened from 4 days to 2 days and using higher concentration of MES. The value of activity for Insoluble Blue Starch in U/mg is obtained by multiplying specific activity with 2.4, which is the extinction coefficient for -amylase. 35 Table 5.5: Purification overview for Trp279Ala Fraction A280 Specific activity Blue Starch Units Units/A280 activity U/mg 1.6 Total activity in Recovery in (%) Medium 15648.2 24396.0 Ultra filtration 4727.5 19247.9 78.9 4.1 Affinity Chromat. 7.7 8894.3 46.2 1157.4 Dialyze 7.0 6870.5 28.2 978 2330 Mutant W278A and W279A were expressed and purified simultaneously and gave the same patterns and profile for purification steps, though we get a little higher concentration of W279A mutant. Yields were approximately 2 and 3 mg for W278A and W279A respectively. Table 5.6: Purification overview for Trp278Ala/Trp279Ala Fraction A280 Total activity in Units Recovery in (%) Specific activity Units/A280 Medium 20256.0 16528.0 Ultra filtration 4818.0 12526.8 75.8 2.6 Affinity Chromat. 14.4 11440.0 69.2 739.9 Dialyze 3.3 3048.8 18.4 935.3 Blue Starch activity U/mg 0.8 2250 The double mutant and the wt were run simultaneously. But we do not see a similar pattern for these two proteins as found for W278A and W279A. The double mutant has the highest content of -amylase in the medium. The reason can be that it was my second run and therefore I became better to do the practical work in expression by shake flask. We see a loss of 25% after ultra filtration. It is a little more than for the other two mutants but is steady in the sensible range. But the loss in affinity chromatograph is only 15 % and is less than for the other mutants. The reason can again be that I got used to the practical methods after using time and trials with affinity chromatography. The reason why the loss in dialysis is so high is that I lost half the content of the dialysis tube. 36 Table 5.7: Purification overview for wt Fraction A280 Total activity in Units Recovery in (%) Specific activity Units/A280 Medium 3330 2602,8 Ultra filtration 1665 1998 76.8 1.3 Affinity Chromat. 1.6 1433 55.1 870.4 Dialyze 1.1 896.8 34.5 817.2 Blue Starch activity U/mg 0,8 1960 The amount of -amylase wt is very low. Though I have expressed wt three times, the content is not comparable with the other variants. The reason is as mentioned in Materials and Method section that the molecular insertion of -amylase gene at the host’s genome did not succeed well and therefore we get these low content. The molecular insertion must be redone. But the final content after purification is enough for the kinetic analysis. Yields were approximately 1.5 and under 0.5 mg for W278A/W279A and wt respectively. 37 Figure 5.1: Coomassie colloidal blue gel for W279A In figure 5.1 is seen that there appears several bands for medium, permeate and flow through from affinity chromatography, while there is single band for the retentate and elute fraction from affinity chromatography. This shows the capability of purification steps to isolate -amylase. 38 Figure 5.2: Silver stained gel From figure 5.2 it is seen for every variant the protein situation in medium (m), after ultra filtration (u) and affinity chromatography (a). For (m) we see several proteins in form of silver colouring all the column, but the band for -amylase is distinguished. The situation for (u) is the same but the band for -amylase gets clearer. For (a) there is only the amylase. It shows that (a) is a very efficient purification step. 5.3. Characterization For the variants of -amylase kinetic parameters were determined to see the effect of the mutations. First the kcat and Km values are determined by DP440 assay. It gives us the opportunity to see if the mutations at the surface binding sites affect the catalytic rate of the enzyme and whether the Km values changes. 39 Tabel.5.7. Kinetic paramaeters determined for amylose DP440 Ref -1 kcat (s ) [14] Km(mM) 1.trial W278A 162 W279A 183 212 Wt W278A/ 167 W279A Ref [14] kcat/Km Ref[14] 2.trial 229 47.38 156 19.09 1 nd 190 1.trial 0.28 0.27 0.25 2.trial 0.25 0.02 0.20 0.2 nd 0.435 1.trial 577 678 845 2.trial 923 768 177 0.10 0.15 0.04 1722 1188 7.07 437 This is seen that wt has the highest kcat value of 212 (s-1) which is around 20% higher than the reference value. The kcat for W278A is up to 229 (s-1) but the standard deviation is too high. The real value must be assumed to lie close to the W279A around 160 (s-1). The double mutant has the lowest kcat. It can be concluded that the binding site mutations do not effect the catalytic rate but it is expected that mutations effect Km so mutations have higher values for Km than the wt. For the wt we got half of the value for Km of the reference and the single mutants have values close to the wt. It can then be concluded that single mutations at surface binding site do not effect the Km, but the double mutant has even lower Km value than the wt. Dublicate values for Km values for each enzyme variant are close and therefore the standard deviation is also low. There must be something wrong the value of Km for double mutant. If I did the experimentals wrongly then I should not get close results for duplicates. If the concentration of enzyme I used was too high or too low I should not get a linear increase in absorbency with time or with increasing substrate concentration, but my excel sheets and micro titer plate show that I do. Also the concentration of enzyme has influence on kcat and not on Km. To compare the results from amylose DP440 we use amylose DP17 to see if I get similar results. 1 No data 40 Tabel.5.8. Kinetic rates determined for amylose DP17. Ref kcat (s-1) [14] Km (mg/ml) 1.trial 2.trial 1.trial 2.trial Ref[14] Ref [14] kcat/Km 1.trial 2.trial W278A 175 201 18.38 0.62 0.82 0.14 282 246 W279A 182 207 17.68 0.73 1.20 0.34 251 172 wt 227 211 11.31 0.71 0.52 0.13 0.57 320 404 W278A/ W279A 187 166 14.85 0.68 0.59 0.06 274 280 165 289.5 The value of kcat is highest for the wt while the others are not far from the wt value. The wt kcat value is 35% higher than the reference value, but the standard deviation is not so high. The wt’s Km value lies close to the reference, but the single mutants Km values are a little higher than the wt’s. It is an expected result. The double determinations are close to each other except for the W279A, which presumably to has a value close to the W278A. The double mutant has again lower value of Km than wt. The problem why I got this unexpected results can be that the assays for amylose DP440 and DP17 are very time consuming, skill and concentration requiring experiments, and more trials are needed before one gets reproducible results. But the project was time limited so the assays were not repeated. The Cl-pPNG7 assay is much easier than the two other assays. Tabel.5.9. Kinetic parameters determined for Cl-pNPG7. kcat (s-1) Ref Km (mg/ml) [14] 1.trial W278A W279A wt W278A/ W279A 2.trial 1.trial 39 39 37 41 1.02 38 1.21 38 0.49 46 42 2.61 40 2.trial 0.55 0.85 0.21 0.50 0.56 0.04 0.76 0.80 0.03 0.86 Ref [14] kcat/Km 1.trial 0.758 0.87 0.01 The double determinations of kcat for wt are very close to each other and to the reference value, around 5% lower. But the mutants have a little higher kcat values and but it is not significant higher. The Km value of wt perfectly matched with the reference and the duplicates are close to each other. The single mutants have lower km values than the wt with a decrease around 25%. An expected result is that the double mutant has higher Km 41 Ref [14] 2.trial 72 48 78 67 50 48 52.8 53 48 value than the wt, but the difference is not significant. The pattern that the double mutant has higher values of Km than the wt indicates an uncertainty in that if the double mutant really is mutated at the right sites. Therefore there must be done an amino sequence of this variant to verify the mutation site. Tabel 5.10 Specific activity for Insoluble Blue starch Variant U/mg W278A 2450 W2789A 2330 W278A/W279A 2250 Wt 1960 Reference for wt [9] 2900 From table 5.10 is seen that value wt of specific activity for Insoluble Blue starch is approximately 30% lower than reference value. All mutants have higher activity than wt, where W278A has the highest, approximately 25% more of wt. The mutants are therefore not effected negatively in activity for Insoluble Blue Starch. Tabel. 5.11 Dissociation constant determined by starch binding. Variant Kd (mg/ml) 2.672 W278A 3.136 W279A 0.702 Wt 0.706 W278A/W279A 2.0 Reference for W279A [10] 0.20 Reference for wt [10] Binding sites (n) 1 1 1 1 From table 5.11 is seen that the value of Kd for wt is almost 3.5 higher than the reference value. The double mutant has also the same Kd value as wt and it confirms that I have not worked with a real double mutant. Thus amino sequence of the double mutant is needed. The other single mutants have higher Kd values than wt, hence their binding to starch is not so tight as for wt. This is an expected result. Also the Kd of W279A is comparable with the reference value. It can be concluded that single mutations at the surface binding site do not influence the values of kcat and Km but influence the dissociation constant Kd. An amino acid sequence of every variant of -amylase was performed by lab technician at BNG but the data was not comparable (data not shown) due to the contamination with other proteins possibly skin proteins from my hands. 42 CHAPTER 6 Conclusion The cloning construction and insertion of the wt must be redone, so it can be expressed in required concentration for kinetic determination work. Another reason why it must be redone is that the expression of wt requires three runs of expression whereas the mutants require only one run, so wt can not be ran simultaneously. The use of shake flask method for expression of variants of -amylase results in enough amounts for determination of kinetic parameters. If one needs larger amount, a fermentor should be used. The purification protocol is good and gives -amylase in highly pure state. Especially the affinity chromatography is an extreme efficient way of recovering the target protein. The loss after dialysis is too high. The period of dialysis must be shortened. The single mutants have not significantly different values of kcat and Km from wt determined towards substrates amylose DP17 and DP440. The method of Cl-pNPG7 gave the most comparable with reference value for the wt, maybe because this short substrate is little affected by the surface site. There was a major difference in Kd between the single mutants and the wt though the reference value of wt was higher than the reference value. It can be concluded that single mutations at surface binding site did not influence Km and kcat, but that both single mutations at surface binding site changing tryptophan at position 278 and tryptophan at position 279 increased the Kd value by making it higher meaning that the mutants bind to starch less tighter than wt. The double mutant gave the same results as the wt throughout the assays used. So probably that the double mutant was not mutated at the planned positions. This missmutation could not be confirmed with amino acid sequencing due to contamination with foreign proteins. 43 Bibliography [1] L. Stryer, Biochemistry, 4. Edition, Chapter 18. [2] D.C Baulcombr, R.A. Martiensen, A.M. Huttley, C.M. Lazarus, Hormonal and development control of gene expression in wheat, JSTOR. 314, p 441-451, (1986) [3] M.T. Jensen, T.E. Gottschalk, B. Svensson, Differences in conformational stability of barley alpha-amylase Isozyme 1 and 2. Role of charged groups and Isozyme 2 specific salt-bridges, Cereal Sci. 38, p289-300,(2003) [4] P.K Nielsen, B.C. Brønsager, C.R Berland, B.W.Sigurskjold, B. Svensson, Kinetics and Energetics of the binding between barley -amylase/subtilisin Inhibitor and Barley -Amylase 2 Analyzed by Surface Plasmon Resonance and Isothermal Titration Calorimetry, Biochemistry. 42, pp 1478-1487, (2002). [5] X. Robert, R. Naser, T. E. Gottschalk, F. Ratajcak, H. Driguez, B. Svensson, N. Aghajari, The structure of barley -amylase Isozyme 1 reveals a novel role of domain c in substrate recognition and binding, Strucure.11 973-984, (2003) [6] J. M .Cregg, T. S. Vedvick, W. C. Raschenke, Recent advances in the expression of foreign genes in Pitchia pastoris, Biotechnology. 11, p 905-909,(1993) [7] Easy select Pichia Selection Kit, Catalog no. K1740-01, Invitrogen. [8] D. R. Higgins, Pichia Protocols, Methods in Molecular Biology, Chapter 9 Humana Press , Totowa New Jersey 44 [9] H. Mori, K. S. B.Jensen, T. E. Gottschalk, M. S. Motawia, I. Damager, B. L. Møller, B. Svensson, Modulation of activity and substrate binding modes by mutation of single and double subsites 11/12 and 25/26 of barley -amylase, Eur. J. Biochem. 268, p6545–6558, (2001). [10] M. Søgård, A. Kadziola, R. Haser, B. Svensson, Site directed mutagenesis og histidine 93, aspartic acid 180 glutamic acid 205, histidine 290 aspartic acid 291 at the active site and tryptophan 279 at the raw starch binding site in barley -amylase, The Journal of Biological Chemistry. 268, pp 22480-22484, (1993). [11] Pharmacia, Affinity Chromatography Principles and Methods, Chapter 2. [12] R. E. Copeland, Enzymes 2.ed., Chapter 5, WILEY [13] R. E. Copeland, Enzymes 2. ed., Chapter 4, WILEY [14] S. Bozonnet, K. Fukuda, B Kramhøft, B svensson, Mutational and gene shufling analysis of specifity of barley alpha amylase , Biocatalysis Biotransformation 21, pp 209-214 (2003). [15] Candiano et al., Electrophoresis 2004. 25, 1327-1333 45 Chapter 7 Appendix Appendix 1 Expression and purification overview 1. Induction 2. Induction Volume Affinity Chrom. U total 340 17.6 5984 W279A 320 28 8960 W278A 320 28.88 9241.60 W279A 340 45.4 15436 W278A / W279A 800 20.66 16528 1800 1.45 2602.8 Retentate W278A 150 82.08 12312 Filtrate W278A 510 5.10 2601 Retentate W279A 155 124.18 19247.9 Filtrate W279A 500 9.96 4980 Retentate W278A / W279A 330 37.96 12526.8 Filtrate W278A / W279A 720 1.78 1281.6 Retentate wt 370 5.4 1998 1650 0 0. Pool 1 6.5 894 5811 Pool 2 3.5 210.5 736.75 1 82.08 82.08 Flow Through 140 14.76 2066.4 Pool 1 5.5 1466 8063 Pool 2 2.5 332.5 831.25 Residue 6.5 124.18 807.17 Wt Ultrafiltration Units/ml W278A Filtrate wt W278A Residue W279A 46 Flow W278A / W279A 150 Waste Dialysis 11 1040 11440 Flow Through 320 1.72 550.4 Pool 6.1 235 1433.5 W278A 6.9 654 4512.6 W279A 9.1 755 6870.5 W278A / W279A 3.7 824 3048.8 Wt 5.9 152 896.8 A280 1. Induction 2. Induction Ultrafiltration 1611 0 Pool Wt 10.74 Dilution A280/ml U/A280 Total A280 W278A 2.02 10 20.20 37.80 6868 W279A 2.24 10 22.37 50.37 7158.4 W278A 2.72 10 27.24 1.06 8716.8 W279A 2.50 10 24.97 1.82 8489.8 W278A / W279A 2.53 10 25.32 0.82 20256 Wt 0.24 10 1.85 Retentate W278A 0.27 100 26.90 3.05 4035 Filtrate W278A 1.90 10 19.02 0.27 9700.2 Retentate W279A 0.31 100 30.50 4.07 4727.5 Filtrate W279A 2.67 10 26.73 0.37 13365 Retentate W278A / W279A 1.46 10 14.60 2.60 4818 Filtrate W278A / W279A 1.42 10 14.15 0.13 10188 Retentate wt 0.15 10 4.50 1.20 1665 Filtrate wt 0.22 10 2.24 0 3696 Pool 1 1.05 1 1.05 853.87 6.8 Pool 2 0.29 1 0.29 720.89 1 Residue 0.27 100 26.90 3.05 26.9 Flow Through 1.60 10 15.97 0.92 2235.8 3330 Affinity Chrom. W278A 47 W279A Pool 1 1.2 1 1.2 1226.78 6.57 Pool 2 0.45 1 0.45 747.19 1.11 Residue 0.31 100 30.50 4.07 198.25 Flow 1.77 10 17.73 0.61 2659.50 Waste W278A / W279A Pool Flow Through Wt Pool Dialysis 1.31 1.31 793.89 14.41 1.42 10 14.2 0.12 4544 0.4 1 0.27 870.37 1.65 W278A 0.39 1 0.65 6985.45 4.46 W279A 0.33 1 0.77 8899.61 7.03 W278A / W279A 0.65 1 0.88 3460.61 3.26 Wt 0.08 1 0.19 4821.51 1.10 48 Appendix 2 Affinity Chromatogram 49 Ahmet W278 W279 80405001:10_UV1_280nm Ahmet W278 W279 80405001:10_Conc Ahmet W278 W279 80405001:10_Fractions Ahmet W278 W279 80405001:10_P960_Flow Ahmet W278 W279 80405001:10_Cond Ahmet W278 W279 80405001:10_pH Ahmet W278 W279 80405001:10_Inject Ahmet W278 W279 80405001:10_Cond% Ahmet W278 W279 80405001:10_Flow Ahmet W278 W279 80405001:10_Logbook ml/min 0.8 0.6 0.4 0.2 0.0 F3 270.0 280.0 Waste A3 A5 A7 A9 A11 A14 B1 B3 B5 B7 B9 B11 B14 C1 C3 290.0 300.0 310.0 50 320.0 330.0 340.0 Waste ml Appendix 3 DP440 DP440 Date: 07-05-05 cells which have to be filled with data Enzyme batch: W278A cells which have to be transferred to next sheet [enz] in assay(µg/ml): 1,33 nmol/l Maltose conc (µg/ml) conc (mM) 0 0,16 0,161 0,1605 0 0,0000 6 0,556 0,555 0,5555 0,395 0,0167 8 0,654 0,659 0,6565 0,496 0,0222 12 1 1 0,9305 0,77 0,0333 20 1,45 1,46 1,455 1,2945 0,0555 40 2,648 2,648 2,648 2,4875 0,1110 Standard curve 3 y = 22,427x + 0,015 2,5 OD 540 nm = Maltose R2 = 0,9995 2 1,5 1 0,5 0 0,0000 0,0200 0,0400 0,0600 0,0800 0,1000 0,1200 conc (mM) slope 22,4273093 UOD/mM Time Dilution factor: conc 2,5 2,5 2,5 2 2,5 1,5 2,5 5 OD 540nm 0,737 OD 540nm 0,98 0,739 0,738 0,704 0,706 0,705 0,877 0,875 0,62 0,628 0,624 0,849 0,842 0,987 7,5 OD 540nm 1,254 1,256 0,876 1,175 0,8455 1,062 0,9835 10 12,5 1,255 OD 540nm 1,461 1,456 1,4585 OD 540nm 1,789 1,792 1,179 1,177 1,444 1,453 1,4485 1,731 1,061 1,0615 1,312 1,305 1,3085 1,598 UDO/min 1,7905 0,1032 1,733 1,732 0,10506 1,599 1,5985 0,09648 2,5 1 0,578 0,579 0,5785 0,725 0,721 0,723 0,979 0,978 0,9785 1,155 1,157 1,156 1,413 1,412 1,4125 0,08404 2,5 0,75 0,528 0,538 0,533 0,718 0,716 0,717 0,948 0,945 0,9465 1,144 1,142 1,143 1,4 1,404 1,402 0,08656 2,5 0,5 0,521 0,531 0,526 0,708 0,706 0,707 0,942 0,941 0,9415 1,139 1,138 1,1385 1,159 1,152 2,5 0,4 0,492 0,486 0,489 0,657 0,655 0,656 0,831 0,826 0,8285 1,026 1,024 1,025 1,026 1,018 2,5 0,3 0,496 0,491 0,4935 0,644 0,646 0,645 0,781 0,783 0,782 1,027 1,022 1,027 1,021 1,024 0,05266857 2,5 0,2 0,477 0,472 0,4745 0,617 0,624 0,6205 0,729 0,726 0,7275 0,96 0,963 0,96 0,954 0,957 0,04751429 2,5 0,1 0,436 0,446 0,441 0,491 0,495 0,493 0,548 0,545 0,5465 0,592 0,594 0,592 0,593 2 1,8 2,5 1,6 500 0,012 450 1,4 0,01 0,4 0,3 1/velocity 0,5 1 velocity 0,75 0,008 0,006 0,2 0,8 0,1 0,6 0,02038 350 1 1,2 0,07122 y = 35,59x + 55,775 400 2 1,5 OD 540 nm 0,014 0,593 0,08288 0,004 300 250 200 150 100 0,002 50 0,4 0 0,2 0 0 0,5 1 1,5 2 2,5 3 0 2 4 6 concentration (mg/ml) 0 0 2,5 5 7,5 10 12,5 15 1/conc time (min) 51 8 10 12 11/27/2003 DP 440 W278A 1.33 nmol/l std slope 0.0445885 22.42731 mg/mL vol microL OD/min mM/min sec-1 v v/s s s/v 1/s 1/v 2.5 200 0.1032 0.0115 144.16 144.16 57.66 2.5 0.01734 0.40 0.00694 Use curve expert to fit the data Data : mM/min = f(mg/ml) 2 200 0.10506 0.0117 146.76 146.76 73.38 2 0.01363 0.50 0.00681 1.5 200 0.09648 0.0108 134.77 134.77 89.85 1.5 0.01113 0.67 0.00742 1 200 0.08404 0.0094 117.39 117.39 117.39 1 0.00852 1.00 0.00852 0 .0 1 0.75 200 0.08656 0.0096 120.91 120.91 161.22 0.75 0.00620 1.33 0.00827 0 .0 1 0.5 200 0.08288 0.0092 115.77 115.77 231.55 0.5 0.00432 2.00 0.00864 0 .0 0 0.4 200 0.07122 0.0079 99.49 99.49 248.71 0.4 0.00402 2.50 0.01005 200 0.052668571 0.0059 73.57 73.57 245.24 0.3 0.00408 3.33 0.01359 0 .0 0 0.3 0.2 200 0.047514286 0.0053 66.37 66.37 331.86 0.2 0.00301 5.00 0.01507 0 .0 0 0.1 200 0.02038 0.0023 28.47 0 .0 0 Saturation Growth-Rate Model: y=ax/(b+x) Curve fit #2-8 Coefficient Data: a= 0.01295125 b= 0.28140147 slope -3.07 slope 0.0062 slope 0.0019 b 520.74 b 0.0018 b 0.0060 R2= 0.8508 R2= 0.9942 R2= 0.9412 kcat 162 kcat 170 kcat 163 kcat 166.1 Km 0.281 Km 0.326 Km 0.288 Km 0.3 564.4 kcat/Km 538 kcat/Km 577 kcat/Km Eadie-Hofstee 521 kcat/Km Hanes-Woolf Lineweaver-Burk Different methods to linearize the data: help to delete obviously wrong experimental points 52 Y Axis (units) S = 0.00021450 r = 0.98796502 0 0 .0 0.0 0.5 0.9 1.4 1.8 2.3 2.7 X Axis (units) Copy/paste info from curve expert to cell A16 Appendix 4 Cl-pNPG7 Cl-pNP mM Cl-pNP 0 0,1 0,2 0,3 0,4 0,5 UOD UOD slope standard = 0 0 0,414 0,823 1,205 1,575 2,055 4,04 UOD/mM 2,5 2 y = 4,04x + 0,002 R2 = 0,9989 1,5 1 0,5 0 0 0,1 0,2 0,3 0,4 0,5 53 0,6 02-08-02 Cl-pNPG7 cells which have to be filled with data AMY 1 3 nmol/l OD/min mM/min 0,25 3,512 0,8693 0,5 12,143 3,0057 1 23 5,6842 2 25,381 3 26,286 5 std slope 0,24752475 sec-1 v/s s s/v 1/s 1/v 4829,48 4829,48 19317,93 0,25 0,00005 4,00 0,00021 16698,29 16698,29 33396,59 0,5 0,00003 2,00 0,00006 31578,66 31578,66 31578,66 1 0,00003 1,00 0,00003 6,2824 34902,37 34902,37 17451,18 2 0,00006 0,50 0,00003 6,5064 36146,86 36146,86 12048,95 3 0,00008 0,33 0,00003 28,905 7,1547 39748,35 39748,35 7949,67 5 0,00013 0,20 0,00003 8 24,226 5,9965 33314,08 33314,08 4164,26 8 0,00024 0,13 0,00003 10 22,714 5,6223 31234,87 31234,87 3123,49 10 0,00032 0,10 0,00003 Saturation Growth-Rate Model: y=ax/(b+x) Curve fit #2-8 slope Coefficient Data: b a= b= 4,04 v 7,11578800 R2= 0,544525560 ko 39532 ko -0,46 slope 29128,97 b Data : mM/min = f(mM) 0,0000 slope 0,0000 0,0000 b 0,0000 0,2119 R2= 0,9646 R2= 0,8907 63986 ko 34581 ko 94759,0 Km 0,545 Km 2,197 Km ko/Km 72599 ko/Km 29129 ko/Km Eadie-Hofstee Use curve expert to fit the data: Growth Models, Saturation Growth Rate 0,343 Km 100674,8 ko/Km Hanes-Woolf Copy/paste info from curve expert to cell A14 4,1 23087 S = 0 .0 0 0 5 7 8 2 0 r = 0 .9 7 9 0 7 8 9 2 Lineweaver-Burk Values to report 1 0.0 Different methods to linearize the data: help to delete obviously wrong experimental points 1 0.0 Y Axis (units) mM 1 0.0 1 0.0 0 0.0 0 0.0 54 0 0.0 0.0 0.5 0.9 1.4 X Axis (units) 1.8 2.3 2.7 Appendix 5 Starch Binding Starch binding assay: 27-04-05 cells to be filled Enzyme: AMY1-W278A to be transferred to other worksheets concentration: dilution before assay: Results: blind: A B C D E F G H I J K L M N 0,07 [starch] 40 30 20 10 5 3 2 1,5 1 0,7 0,4 0,2 0,1 0 0,07 Abs. 0,525 0,554 0,594 0,621 0,734 0,812 1,084 1,112 1,212 1,294 1,345 1,412 1,422 1,453 0,079 0,527 0,594 0,612 0,613 0,754 0,842 1,112 1,122 1,234 1,304 1,356 1,402 1,433 1 0,079 0 0,525 0,554 0,594 0,621 0,734 0,812 1,084 1,112 1,212 1,294 1,345 1,412 1,422 1,453 Mean Abs. 0,527 0,526 0,594 0,574 0,612 0,603 0,613 0,617 0,754 0,744 0,842 0,827 1,112 1,098 1,122 1,117 1,234 1,223 1,304 1,299 1,356 1,3505 1,402 1,407 1,433 1,4275 1 1,457 Efree Ebound = abs. Etot-Efree 0,526 0,931 0,574 0,883 0,603 0,854 0,617 0,84 0,744 0,713 0,827 0,63 1,098 0,359 1,117 0,34 1,223 0,234 1,299 0,158 1,3505 0,1065 1,407 0,05 1,4275 0,0295 1,457 0 B 0,638984 0,60604 0,586136 0,576527 0,489362 0,432395 0,246397 0,233356 0,160604 0,108442 0,073095 0,034317 0,020247 0 B/L 0,015975 0,020201 0,029307 0,057653 0,097872 0,144132 0,123198 0,155571 0,160604 0,154917 0,182739 0,171585 0,202471 1/B 1,564984 1,650057 1,706089 1,734524 2,043478 2,312698 4,058496 4,285294 6,226496 9,221519 13,68075 29,14 49,38983 1/L 0,025 0,033333 0,05 0,1 0,2 0,333333 0,5 0,666667 1 1,428571 2,5 5 10 L/B 62,59936 49,5017 34,12178 17,34524 10,21739 6,938095 8,116992 6,427941 6,226496 6,455063 5,4723 5,828 4,938983 L 40 30 20 10 5 3 2 1,5 1 0,7 0,4 0,2 0,1 1,6 1,4 B 0,7 0,7 1,2 0,6 1 0,5 0,8 0,4 0,6 0,3 0,6 0,5 0,4 0,3 0,2 0,2 0,4 0,1 0,1 0,2 55 0 0,1 0 0 0 0 10 20 30 40 50 5 10 15 20 25 30 35 40 45 1 10 100 Summary: 27-04-05 Use curve expert to fit the data: Growth Models, Saturation Growth Rate w278a B Scatchard Hanes-Wolf 1/L 1/B B/L B L L/B 40 30 20 10 5 3 2 0,638678596 0,61871989 0,59119064 0,572608396 0,494838266 0,441156228 0,25395733 0,025 0,033333333 0,05 0,1 0,2 0,333333333 0,5 1,565732759 1,616240267 1,691501746 1,746394231 2,020862309 2,266770671 3,937669377 0,015966965 0,020623996 0,029559532 0,05726084 0,098967653 0,147052076 0,126978665 0,638678596 0,61871989 0,59119064 0,572608396 0,494838266 0,441156228 0,25395733 40 30 20 10 5 3 2 62,62931034 48,48720801 33,83003492 17,46394231 10,10431154 6,800312012 7,875338753 1,5 1 0,7 0,4 0,2 0,1 0,234686855 0,16586373 0,109428768 0,074328975 0,028217481 0,021335169 0,666666667 1 1,428571429 2,5 5 10 4,260997067 6,029045643 9,13836478 13,4537037 35,43902439 46,87096774 0,156457903 0,16586373 0,156326812 0,185822436 0,141087405 0,213351686 0,234686855 0,16586373 0,109428768 0,074328975 0,028217481 0,021335169 1,5 1 0,7 0,4 0,2 0,1 6,391495601 6,029045643 6,396855346 5,381481481 7,087804878 4,687096774 0 Kd/n = 1/n= 4,940730149 -Kd= 1,703616997 1/n= -3,296061182 1/n= 0,710738213 Kd/n= 1,451348932 4,450430329 R2= 0,958888467 R2= 0,824010538 R2= 0,996280881 n= Kd= 0,586986395 Kd= 2,900141379 n= 3,296061182 n= 1,406987808 Kd= Data : B = f(starch) S r Y A x is (u n i ts ) Langmuir [starch] User-Defined Model: y=a/(1+(b/x)) 0,689014184 Coefficient Data: 3,066409621 a = 0,692941 =n b= 2,6572012 =Kd Different methods to linearize the data: help to delete obviously wrong experimental points 56 0. 7 0 0. 5 8 0. 4 7 0. 3 5 0. 2 3 0. 1 2 0. 0 0 0.0 7.3 14.7 22.0 X Axis (units) 29.3 = = 0 . 0 3 4 8 1 7 7 5 0 . 9 9 0 2 5 0 9 8 36.7 44.0 Appendix 6 Stock Solutions for Media Preparation 10X YNB (13.4% YNB): For making 1 L of YNB, 134 g of YNB (Yeast nitrogen base with ammonium sulfate without amino acids) is dissolved in 1 L of H2O and filtered for sterilization through 0.45 µm membrane and stored at +4C 500X Biotin (0,02% Biotin): 20 mg of biotin is dissolved in 100 ml H2O, filtered for sterilization and stored at +4 C. 10X Dextrose (20% Dextrose): 200g of glucose is dissolved in 1 L H2O, autoclaved and stored at room temperature. 10X Methanol (5% Methanol) 50 ml of methanol is mixed in 950 ml H2O, filter sterilized and stored at +4C 10X Glycerol (10% Glycerol): 100 ml glycerol is mixed with 900 ml H2O, filter sterilized and stored at +4C 1 M potassium phosphate buffer, pH 6.0 : 132 ml of 1 M K2HPO4 with 868 ml 1 M KH2PO4. The pH is adjusted with KOH to pH=6.0. This solution is autoclaved and stored at room temperature. 57 Appendix 7 Coomassie blue and silver staining Colloidal coomassie blue stain [15]: Samples of variant enzymes from the medium and purification steps were loaded on gradient gels (Nupage Novex Bis Tris gels). The samples were mixed together with 7µl SDS Sample Buffer (3X) for coloring and 2.5µl DTT (dithiotreitol) for reducing sulphite bindings. The total volume of sample should be 20 µl, at table 7.1 a list of volumes of various enzymes is showed, rest volume is filled with H2O.The gel is fastened to its chamber and the chamber is filled with running buffer which is prepared by mixing 50 ml 20X Nupage MES (2-morpholinethansulfosyre) with 950 ml H2O. Table 7.1: Sample Volume for Staining Sample Volume in µl Medium 5 Permeate 10 Retentate 3 Flow through 10 Pool 5 (100X) Marker 6 Run conditions are done by setting the voltage to be constant at 200 V and choosing a run time at 35 min. After time the gel is fixed in fixer solution for 30 min. The fixer solution consist of 30% ethanol and 2% phosphoric acid. Then the gel is washed twice with wash solution which is 2% phosphoric acid. At last the gel is incubated over night in Blue silver stain. This stain can be made to 500 ml by first adding 50 ml 85% phosphoric acid to 50 ml H2O, then adding 50 g ammonium sulfate and heat stirring until complete dissolve 58 and then adding 600 mg Coomassie Blue G-250. Adding H2O to 400 ml and methanol to 500 ml completes the stain. The stain is kept from light. After time the gel is washed with deionized H2O and a digital photography is taken. Silver staining: Samples of variant enzymes from the medium and purification steps were loaded on gradient gels (Nupage Noves Bis tris Gels) as coomassie blue. The gel is fastened to its chamber and applied as in coomassie blue stain. The gel is the soaked in fixing solution for 30 min. The fixing solution is made of fresh 40% methanol, 5%formaldehyd. The gel is then washed twice with H2O for 5 min and then soaked in 0, 02% Na2S2O3 for 1 min and washed for 20 sec. The gel is then soaked in 0, 1% silver nitrate for 1 min and then soaked in fresh developer solution until bands appear. The developer solution is made of 0,3% Na2S2O3and 0,05% formaldehyde and 0,0004% Na2S2O3. Process was stopped by adding 2.3M citric acid and shaken for 10 min. The gel was washed and dried and a digital photography is taken. 59