Plans For Monday, January 12, 2015
advertisement

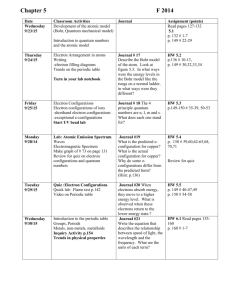
Plans for Monday, January 12, 2015 By: ORHS Pre-AP Chemistry Chemistry Learning Standards: [Integrated] Use the Periodic Table to identify and explain the properties of chemical families, including alkali metals, alkaline earth metals, halogens, noble gases, and transition metals (CCRS VII C-1)[5B] [Integrated] Use the Periodic Table to identify and explain periodic trends, including atomic and ionic radii, electronegativity, and ionization energy. (CCRS VII C-2)[5C] [Integrated] [Concept Recommendation]Group and Period Trends [Integrated] Differentiating Environment [Integrated] Flexible Grouping OBJECTIVE TSW learn how trends occur in the periodic table in a regular order and use this information to predict atomic and ionic radii, electronegativity, and ionization energy AGENDA 1. Notes over periodic trends 2. Trends Straw lab 3. Trends homework homework in notes packet ASSESSMENT None today HOMEWORK Periodic trends graphing homework DIFFERENTIATED INSTRUCTION lab Plans for Tuesday, January 13, 2015 By: ORHS Pre-AP Chemistry Chemistry Even day, same as Monday Plans for Wednesday, January 14, 2015 By: ORHS Pre-AP Chemistry Chemistry Learning Standards: [Integrated] [Concept Recommendations]Energy, Wavelength and Frequency relationshipsCalculate Energy, Frequency and WavelengthOrbital notation, electron dot diagrams, electron configuration, and noble gas configuration. [Integrated] Express the arrangement of electrons in atoms through electron configurations and Lewis valence electron dot structures. (CCRS VII B-1)[6E] [Integrated] List the four quantum numbers and state what each number indicates about the electron. (PRE-AP) [Integrated] Use the Periodic Table to identify and explain the properties of chemical families, including alkali metals, alkaline earth metals, halogens, noble gases, and transition metals (CCRS VII C-1)[5B] [Integrated] Use the Periodic Table to identify and explain periodic trends, including atomic and ionic radii, electronegativity, and ionization energy. (CCRS VII C-2)[5C] [Integrated] [Concept Recommendation]Group and Period Trends OBJECTIVE TSW Demonstrate ability to predict trends on the periodic table based on location, apply electron and noble gas configurations, use quantum numbers to describe electrons in atoms AGENDA 1. Quiz over trends, electron configuration, and quantum numbers 2. Graphing activity - trends 3. Mini Test review ASSESSMENT Quiz over unit topics HOMEWORK Finish graphing activity and test review DIFFERENTIATED INSTRUCTION Plans for Thursday, January 15, 2015 By: ORHS Pre-AP Chemistry Chemistry Even day, same as Wednesday Plans for Friday, January 16, 2015 By: ORHS Pre-AP Chemistry Chemistry Learning Standards: [Integrated] [Concept Recommendations]Energy, Wavelength and Frequency relationshipsCalculate Energy, Frequency and WavelengthOrbital notation, electron dot diagrams, electron configuration, and noble gas configuration. [Integrated] Express the arrangement of electrons in atoms through electron configurations and Lewis valence electron dot structures. (CCRS VII B-1)[6E] [Integrated] List the four quantum numbers and state what each number indicates about the electron. (PRE-AP) [Integrated] Use the Periodic Table to identify and explain periodic trends, including atomic and ionic radii, electronegativity, and ionization energy. (CCRS VII C-2)[5C] [Integrated] Use the Periodic Table to identify and explain the properties of chemical families, including alkali metals, alkaline earth metals, halogens, noble gases, and transition metals (CCRS VII C-1)[5B] [Integrated] [Concept Recommendation]Group and Period Trends OBJECTIVE TSW demonstrate mastery of periodic trends, electron configurations, and quantum numbers AGENDA 1. Take up review and answer questions 2. Mini Test Electron Configuration, Quantum Numbers, and Periodic Trends 3. Covalent nomenclature ASSESSMENT Unit Mini Test HOMEWORK Finish covalent nomenclature DIFFERENTIATED INSTRUCTION