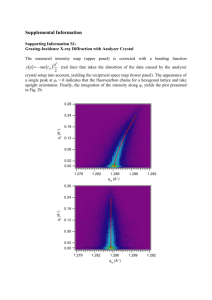
Full text
advertisement

Construction of Adjacent Quaternary and Tertiary Stereocenters via an Organocatalytic Transformation of Morita-Baylis-Hillman Carbonates OBoc CN COOMe + R COOEt R' O N N R CN * * COOEt COOMe R' OH Dirk Jan van Steenis Report Research project, master Molecular Design Synthesis and Catalysis (MDSC) What is organocatalysis? “Organocatalysis is the acceleration of a chemical transformation through addition of a sub-stoichiometric amount of an organic compound which does not contain a metal atom” - Peter I. Dalko and Lionel Moisan “Catalytic reactions mediated by small organic molecule in absence of metals or metal ions.” - Carlos F. Barbas, III “ A field of chemistry that pays my mortgage and has gotten me many free dinners.” - David W. C. Macmillan A part of this report has been submitted for publication: Dirk Jan V. C. van Steenis, Tommaso Marcelli, Martin Lutz, Anthony L. Spek, Jan H. van Maarseveen, Henk Hiemstra*. 2006. 2 Table of Contents Summary ___________________________________________________________ 4 List of abbreviations __________________________________________________ 5 Chapter 1; Introduction ________________________________________________ 7 1.1 Organocatalysis _________________________________________________________ 7 1.2 Cinchona Alkaloids ______________________________________________________ 9 1.2.1 Bi-functional cinchona alkaloids ............................................................................. 12 1.3 Morita-Baylis-Hillman (MBH) Reaction ___________________________________ 14 1.3.1 The Morita-Baylis-Hillman mechanism.................................................................. 15 1.3.2 Morita-Baylis-Hillman Adducts .............................................................................. 16 1.4 Dynamic Kinetic Asymmetric Transformation (DYKAT) of Morita-Baylis-Hillman Adducts _________________________________________________________________ 19 1.5 Functionalization of Morita-Baylis-Hillman Adducts via a Tandem SN2’-SN2’ Mechanism _______________________________________________________________ 21 1.6 All-carbon Quaternary Stereocenters ______________________________________ 25 Chapter 2; Allylic Nucleophilic Substitution of Morita-Baylis-Hillman Carbonates 27 2.1 Goal _________________________________________________________________ 27 2.2 Catalyst Synthesis ______________________________________________________ 29 2.3 Preliminary Experiments and Optimization ________________________________ 32 2.4 Results _______________________________________________________________ 35 2.4.1 2.4.2 2.4.3 Substrate scope ................................................................................................. 35 X-Ray analysis ................................................................................................. 38 Mechanistic considerations .............................................................................. 40 2.5 Conclusions __________________________________________________________ 41 Samenvatting _______________________________________________________ 43 Acknowledgements __________________________________________________ 44 Chapter 3 Experimental _______________________________________________ 45 References _________________________________________________________ 70 3 Summary Organocatalysis, catalytic transformations with small organic molecules, has found renewed interest in both academia and industry. Although it was already known for a century that organic compounds could catalyze asymmetric reactions, it is only since half a decade that the potential of organocatalysis is understood and is starting to be fully explored. In the treatment of diseases, one enantiomer of a medicine is usually more potent, and in the worst scenario, the opposite enantiomer can cause serious side effects or even death. Therefore, the demand from both pharmaceutical and chemical industry for new reliable asymmetric transformations of molecular skeletons is higher then ever. Nowadays, the construction of (highly) functionalized asymmetric skeletons still suffers from drawbacks, especially when there is a quaternary stereocenter involved in the target molecule. Organocatalysis gained popularity in a relatively short time span, because it has led to a large assortment of new asymmetric demanding transformations in the last five years. In some cases, organocatalysts meet the selectivity and efficiency levels of established metal catalyzed organic reactions. Since organocatalysis has a hidden potential, it could provide a solution for challenging alterations in the future. Moreover, organocatalysis can serve as a green alternative for transition metal catalyzed reactions. During this master research project in organocatalysis we found that the organocatalyst β-isocupreidine (β-ICPD) effectively catalyzes a one-step asymmetric transformation of Morita-Baylis-Hillman carbonates. This transformation led to new molecular assemblies with vicinal quaternary and tertiary stereocenters. We observed high chemo-, diastereo-, and enantioselectivities with this reaction catalyzed by β-isocupreidine, in addition the chemical yields of these transformations are excellent. This one-step organocatalytic allylic alkylation is the first, out of six reactions reported, in which the reaction mechanism can not be only explained in terms of a conjugate addition and thereby leading to adjacent quaternary and tertiary stereocenters. 4 List of abbreviations H2O: water CDCl3: deutero-chloroform CHCl3: chloroform DCM: dichloromethane CH2Cl2: dichloromethane EtOAc: ethyl acetate EI: electron impact Et2O: diethyl ether FAB: fast atom bombardment 1 proton1 nuclear magnetic resonance H NMR: 13 carbon13 nuclear magnetic resonance HRMS: high resolution mass spectroscopy J: coupling constant Hz: hertz IR: infra red FTIR: Fourier transform infra red UV: ultraviolet K2CO3: potassium carbonate MD3OD: deutero-methanol MeOH: methanol MgSO4: magnesium sulfate MHz: megahertz MS: mass spectrometer nm: nanometer PE: petroleum ether HCl: hydrochloric acid KOH: potassium hydroxide H3PO4: phosphoric acid N2: nitrogen gas DBU: 2,3,4,6,7,8-hexahydropyrrolo[1,2-a]pyrimidine DABCO: 1,4-diaza-bicyclo[2.2.2]octane DMAP: N,N-dimethylpyridin-4-amine C NMR: 5 CO2: carbon dioxide TLC: thin layer chromatography Boc2O: tert-butoxycarbonyl anhydride THF: tetrahydrofuran DMSO: dimethylsulfoxide ee: enantiomeric excess dr: diastereomeric ratio h: hours min: minutes Pd: palladium Boc: tert-butylcarbonate OAc: acetate BINAP: 2-(diphenylphosphino)-1-(2-(diphenylphosphino)naphthalen-1yl)naphthalene NH3: ammoniac UV: ultra violet AAA: asymmetric allylic alkylation DYKAT: dynamic kinetic asymmetric transformation KAT kinetic asymmetric transformation MBH Morita-Baylis-Hillman Å: Angström Eq.: equivalent KBr: potassium bromide Mp: melting point H: proton λ: Lambda HPLC: high performance liquid chromatography s: singlet d: doublet m: multiplet dd: double doublet t: triplet mmol: millimoles M: molarity 6 Chapter 1; Introduction 1.1 Organocatalysis Less then a decade ago it was generally accepted that highly efficient asymmetric transformations where only restricted to chiral organometallic complexes and enzymes. Indeed, the levels of regio- and enantioselectivity achieved nowadays by these catalyst systems is impressive. The number of organic reactions reported in the last decades catalyzed by these complexes has enormously contributed to our quality of life. The oil, chemical and pharmaceutical industry and even our economy, are extensively relying on these ways of influencing molecular properties. Nevertheless, the need for new and reliable asymmetric transformations is high. The amount of new chiral drugs introduced on the market is higher then ever and subsequently the demand for new efficient asymmetric transformations. Nature is chiral and does everything with tremendous (asymmetric) efficiency, moreover, in multiple reactions catalyzed by nature, there is no metal involved. Apparently nature does not always need metals for challenging transformations. Scientists nowadays use nature as an inspiration source and try to mimic these reactions in the lab. The concept of organocatalysis can be seen as a blackboard example of this. In recent years asymmetric organocatalysis, is regaining interest and can be considered as a rapidly expanding research field.1 In addition, organocatalysis has proven in the last years to be a valuable alternative in respect to the traditional asymmetric catalytic methods, a few examples are given below; Cheap catalyst sources with respect to organometallic catalysts No toxic transition metals in the product (catalyst leaching). Environmentally friendly Usually less demanding reaction conditions Evidence has been found that this metal free type of catalysis has played a important-role in the formation of essential key-building blocks for life. The natural amino acids, L-alanine and L-isovaline which can catalyze for instance certain Aldol reactions, have been found in an enantiomeric excess (ee) of 15 % on meteorites1b. Although the first asymmetric transformation with a small organic molecule was reported in 1912, by Bredig and Fiske2, it is no more then a few years ago that the potential of organocatalysis has been understood by the scientific community. List and Barbas3 reported in 2000 a breakthrough in organocatalysis 7 and “pulled the trigger”. They reported that simple proline catalyzes an aldol reaction in good enantiomeric excesses. Since then, organocatalysis is starting to get the attention it deserves and has become a mature concept in the field of (homogenous)catalysis and in some cases organocatalysts meet the selectivity and efficiency levels of established metal catalyzed organic reactions. During the ninety years of absence, only a few scientists understood the potential of metal free catalysis. In the sixties, Pracejus applied cinchona alkaloids in the asymmetric conversion of ketenes to (S)-methyl hydratropate4. The seventies brought an other milestone, Hajos and Parrish reported a L-proline catalyzed Robinson annulation in excellent enantioselectivities5. Moreover, Wiechert reported an organocatalyic aldol reactions in good enantioselectivities6. In the eighties, Wynberg and co-workers reported various 1,2 and 1,4 additions catalyzed by cinchona alkaloids7. Besides these notable reports, the field of organocatalysis has been remarkably overlooked. In respect to the traditional transition metal based catalysts, organocatalysts are often inexpensive, environmentally friendly and the reactions can usually be performed under aerobic reaction conditions. Different from enzymes, organocatalysts do not require buffer conditions, and therefore have less solubility problems and have a broad substrate scope. Moreover, organocatalysts can usually simply be separated from reaction mixtures without considerable catalyst leaching and recovered via extraction. An other interesting and attractive feature of organocatalysts is that they usually possess an excellent functional group tolerance. These characteristics can make them practically and interesting counterparts of traditional homogenous catalysts and enzymes, therefore it is justified to continue further exploration of organocatalysis. The vast majority of (asymmetric) organic reactions can be explained by a nucleophile and electrophile mechanism. Organocatalysts can be divided into B S several subclasses namely; Lewis bases, Lewis acids, Brønsted bases and Brønsted acids1d. The majority of the organocatalyst published to date works via an Lewis base mechanism (Fig. 1). S For simplicity reasons, only the Lewis base and the Brønsted B B P acid mechanism are taken into account in this report. The simplified Lewis base mechanism can be best described by the following steps; the Lewis base (B), for instance a cinchona P alkaloid, starts the catalytic cycle via a nucleophilic addition to, or deprotonation of, the substrate (S). The resulting chiral Fig 1 Simplified Lewis base catalytic cycle 8 intermediate undergoes a reaction and the newly formed product (P) is separated from the catalyst. The catalyst is now regenerated and available for a new catalytic cycle. Many organocatalytic reactions described in literature cannot simply be explained by a single Lewis base mechanism. Often there is a second mechanism involved, usually a Brønsted acid mechanism. This type of organocatalysis is called bi-functional organocatalysis. Cinchona bifunctional organocatalysis was first introduced by Wynberg and coworkers7d. They proposed that both a base and an acid can be crucial in activation and orientating the reaction partners, an elegant example being the asymmetric addition of aryl-thiols to conjugated cycloalkenones7c. In a bi-functional catalyzed reaction, the organocatalyst bears besides a Lewis base, typical a nitrogen or phosphorus atom, also a Brønsted acid functionality. These facts have been only recently understood and are only since the last years frequently applied, although it was already known in the eighties. Especially, activation and orientation through thio-urea hydrogen bonding is emerging8. Other widely applied Brønsted acids functionalities in organocatalysis are urea and alcohol functional groups. Similar to enzyme catalysis, there is now also hydrogen bonding involved in the transition state. This kind of hydrogen bonding usually makes the electrophile more electron deficient and therefore more prone to nucleophilic attack. Besides activation, the hydrogen bonding motive holds the electrophile also in a kind of a conformational lock (orientation). Via this conformational lock higher enantioselectivities can be obtained. Brønsted acid activation through thio-ureas has been applied in asymmetric reactions such as the Michael (conjugate) addition, (aza)-Henry, Strecker and Friedel-Craft alkylations9. 1.2 Cinchona Alkaloids For practical and economical considerations, an important prerequisite for an organocatalyst is that both enantiomers of the catalyst are readily available. Now, there is access to both enantiomers of a product with usually similar ranges of asymmetric induction. Cinchona alkaloids are a natural class of compounds that exhibit this unique feature and makes them highly attractive candidates for asymmetric catalysis 10. Quinine (QN) / Quinidine (QD), and Cinchonine (CN) / Cinchonidine (CD) (Fig. 2), are two pairs of so called pseudoenantiomers. These pseudoenatiomers are not enantiomers, because they are not mirror images. 9 3 3 4 N 1 OH OH 9 9 1 N 8 8 N 1 N 6´ 6´ R Quinine (QN) Cinchonidine (CD) 4 R R=OMe R=H Quinidine (QD) Cinchonine (CN) Fig 2 The four most natural occurring cinchoa alkaloids 10 They are called pseudoenantiomers because they are “almost enantiomeric pairs”. To date there are more then twenty different O cinchona alkaloid producing plants known, by far the most important is the “Cinchona pubescens”. To date, more then thirty HN N different kinds of cinchona alkaloids have been isolated and R characterised. The tree bark of the Cinchona pubescens can contain over 50 % of the main four cinchona alkaloids which Quinicine Cinchonicine can be easily gathered, via extraction. The cinchona alkaloids family forms a unique class of natural compounds and has found R=OMe R=H Fig 3 Cinchona alkaloids used by Pasteur widespread application, such as in the pharmaceutical, chemical and beverage industry and are produced on a multi-ton scale (aprox. 700 t/year) and therefore readily available.11 Already three-hundred years ago, quinine (QN) received attention because of its biological activity although the structure was not understood yet. Quinine (QN) was officially isolated and reported by Pelletier in 182012 In the past decades quinine (QN) has been extensively studied and used for the treatment of malaria. However, alternatives for the treatment of malaria have been developed due to side effects of quinine. Nowadays cinchona alkaloids are mainly used by the chemical and beverage industry. Quinine (QN) is used by the soft drink industry for the typical bitter flavour. The chemical compositions of cinchona alkaloids differ, but usually they bear two key characteristics; they all have an aromatic quinoline part and usually a basic quinuclidine moiety in the molecular skeleton (Fig.2). The pseudoenantiomers, quinine (QN) / quinidine (QD) have an additional functionality; they bear at the 6’-position of the aromatic quinoline part a methoxy-group. This functionality gives a handgrip for further functionalization. A milestone achieved by making use of the chiral properties of cinchona alkaloids, was the resolution of racemic tartaric acid. Pasteur, achieved the separation of the enantiomeric pair by using the cinchona alkaloid derivatives quinicine and cinchonicine (Fig. 3). The first asymmetric reaction catalyzed by a cinchona alkaloid was reported in 1912 by Bredig and Fiske2. The asymmetric addition of hydrogen cyanide to benzaldehyde was reported in an enantiomeric excess (ee) of 20 %. Further development of cinchona alkaloids in asymmetric catalysis was reported in 1960 by Pracejus in the asymmetric conversion of ketenes4. An other cinchona alkaloid pioneer, Wynberg, expanded the reactions catalyzed by cinchona alkaloids to various 1,2 and 1,4 additions. 7 In the last two decades cinchona alkaloids are recognized as a privileged class of chiral auxiliaries and have found a variety of applications in asymmetric synthesis13. 11 1.2.1 Bi-functional cinchona alkaloids In order to enlarge to scope of chemical transformations catalyzed by cinchona alkaloids, many efforts have been put into the modification of these natural compounds. In the past two decades modified cinchona alkaloids have led to excellent results in various research fields. In phase transfer catalysis (PTC), quaternary ammonium ions derived from cinchona alkaloids have played an important role. The quinuclidine nitrogen atom can perfectly undergo transformation into a quaternary ammonium ion and could serve as a phase transfer catalyst. Cinchona alkaloids have played a key role in asymmetric dihydroxylation reactions, 14 C9dimerized Cinchona alkaloids have successfully been applied as chiral ligands in the Sharpless asymmetric dihydroxylation (AD). To stress the importance of these ligands; various analogues of these dimers are now commercially available. Barry K. Sharpless received OR the Nobel prize in chemistry in 2001 for his N contribution to asymmetric synthesis. The cinchona 9 Lewis Base dihydroxylation ligands formed a substantial part of N his work. Other alterations, different than at the C9 position of the cinchona alkaloid, usually occur at the 6´ OH Brønsted acid C6’-position of quinine (QN) and quinidine (QD). When this position is changed from a methoxy into a Fig 4 Bi-functional organocatalyst Brønsted acid functionality, these organocatalysts become bi-functional (Fig. 4) as discussed in paragraph 1.1. These types of organocatalysts are often referred to as cupreines and cupreidines15. In 1999, Hatakeyama proved for the first time that when a C6’ methoxy is transformed into a C6’-OH Brønsted acid function, high enantioselectivities could be obtained via this moiety. Since then bi-functional cinchona organocatalysts are fully explored. In the past few years several bi-functional modified quini(di)ne catalysts have been published16 A broad spectrum of reactions are catalyzed with excellent (enantio)selectivities. The reactions published in the past few years have been performed with surprisingly simple and accessible organocatalysts. Besides unmodified cinchona alkaloids, three different kind of successful catalysts classes for the creation of chiral skeletons can be distinguished in literature namely; 1) Quinidine derived oxaza-twistanes (i.e. β-isocupreidine)11 2) C6’ hydroxyl and C9 alkylated cupreidine catalysts15 12 3) a C6 or C9 thiourea equipped quini(di)ne 16 Constrained oxaza-twistanes (Fig. 5), are a class of catalysts whose the synthesis 3 has been mainly explored by the group N of Hofmann. These compounds posses 9 9 special characteristics, due to the extra N constrained ring between C9 and C3, in β-isocupreidine, N OH 6´ respect to other cinchona alkaloids. Especially N O O (IUPAC 6´ OH Fig. 5 β-isocupreidine name; 3R, 8R, 9S-10, 11- Dihydro-3,9epoxy-6’-hydroxycinchonane), derived in one step from quinidine, has proven to be a excellent catalyst in various reactions. The β-isocupreidine quinuclidine nitrogen atom owes, due to the extra cycle, more basic and nucleophilic character. Moreover, β-isocupreidine has less conformations due to the extra ring. β-Isocupreidine has been successfully applied to multiple reactions such as the asymmetric Morita-Baylis-Hillman reaction17(see later) and the asymmetric aza-MBH reaction between the strongly activated Michael acceptor 1,1,1,3,3,3hexa-fluoroisopropyl acrylate and aryl-imines18. β-isocupreidine has also been applied in the construction of quaternary stereocenters51f. The enantio-complementary catalyst of βIsocupreidine has recently been synthesized19. Although the synthesis 3 involves 19 steps, now there is access to R2 4 both enantiomers of a product. A second N 9 superior catalyst class has been introduced by Deng and co-workers. C6’ 8 1 N 6´ sc = R1 catalysts, again easily accessible, gave BnCPD PHNCPD excellent levels of enantio- and C6´ ThQD C9 ThQD diastereo-discrimination in a broad H N CF3 S R1 hydroxyl and C9 alkylated cupreidine H N R1 = OH R1 = OH R1 = sc R 1= H R2 = Bn R2 = Phn R2 = Bn R2 = sc CF3 compilation of conjugate additions20. In this elegant collection of papers Fig 6 Some frequently used quinidine derrived organocatalysts published by Deng, it is demonstrated how various 1,3-dicarbonyl, 1,3-nitro-carbonyl and 1,3 cyano-carbonyl pronucleophiles can react with simple Michael-acceptors. These catalysts reported by Deng, can also 13 simultaneously construct adjacent quaternary and tertiary stereocenters, which is a tremendous challenge in synthetic organic chemistry. These reported asymmetric reactions elegantly show how via organocatalysis highly functionalised skeletons are accessible with relatively simple catalysts. (Fig. 6) The third expanding catalyst category is thiourea modified quinidines15. Two versions have been published recently, the C6´ 9e and C921 thiourea equipped quinidine. Although the catalyst synthesis requires additional steps compared to the catalysts of Deng, they have important advantages. Due to the electron deficient thio-urea moiety of the catalyst, high levels of enantiodiscrimination can be achieved like in a fundamental reaction like the Henry reaction. The organocatalytic Henry reaction, a reaction between an aldehyde and typical nitro-(m)ethane, has recently been elucidated in high enantioselectivities9e, 15. The catalyst of choice was a C6’ thiourea equipped quinidine with a the C9 carbon a benzyl protective group (fig 6,C6’ THQD). In these two papers dealing with cinchona catalyzed Henry reaction, it is verified that the C6’ thiourea moiety is a stronger hydrogen donor compared to the C6’ hydroxyl donor (BnCPD)22. 1.3 Morita-Baylis-Hillman (MBH) Reaction The Morita-Baylis-Hillman reaction was first O described in 1968 by Morita23; he observed a phosphine-derivative catalyzed addition of an R H + EWG Base OH R EWG aldehyde to an acrylate, yielding densely funtionalised β-hydroxy-α-methylene esters Fig 7 Some frequently used quinidine derrived organocatalysts (Fig. 7). He named the reaction the “Carbinol Addition”. In 1972 the reaction was reinvented and patented by two chemists at the Celanese Corporation in New York, Anthony Baylis and Melville E. D. Hillman24. The yield described by Morita was poor but Baylis and Hillman found that if 1,4-DiAzaBiCyclo[2.2.2]Octane (DABCO) was used instead of the phosphine derivative, the yield was roughly three times higher. After its discovery, the Morita-Baylis-Hillman reaction was almost “forgotten” for a decade, partly due to the slow reaction rate (typically days). However, the Morita-BaylisHillman is recognized now as an important carbon-carbon bond forming reaction like the aldol, Grignard, Friedel-Crafts, Heck and Suzuki reaction25. The MBH reaction has become a well-established reaction due to fact that it meets several important standards; the MBH reaction generates multiple functional groups and thereby it provides a simple and atom economic carbon-carbon bond forming process. Moreover, the MBH reaction encloses an 14 immense synthetic potential, almost every aldehyde and activated olefin undergoes the MBH reaction. Supporting evidence for this statement can be found with a simple search in literature. The amount of papers dealing with MBH chemistry is impressive. Up to now the MBH reaction emerged to an important classic “text book” organic reaction but still faces major challenges, illustrative features being the selective formation of the tertiary stereocenter26 and rate. Recently, Verkade and co-workers have made an impressive step forward27. A highly active two catalyst combination, relying on a proazaphosphatrane organocatalyst and titanium-tetra-chloride was reported. Although the products are obtained racemic and the catalysts are air-sensitive, the methodology presented tolerates a broad substrate scope. For more then a decade the selective formation of the tertiary stereocenter has become an enormous challenge. Selective formation of this stereocenter would provide a route to optically enriched β-hydroxy-α-methylene esters, ketones, nitriles etc. The first breakthrough in the asymmetric MBH reaction was reported in 1999 by Hatakeyama and coworkers17. They used β-isocupreidine (Fig. 5) to catalyze the asymmetric MBH reaction between aldehydes and the strongly activated Michael acceptor, 1,1,1,3,3,3-hexafluoroisopropyl acrylate. Manifold chiral catalysts have been tried28, including Brønsted acids and derivatized binaphthtyls. So far high enantioselectivities have not been reported with simple acceptors such as methyl acrylate and acrylonitrile. A recent trend in the asymmetric (aza)-MBH reaction is the use of catalysts combinations. Usually DABCO and a chiral thiourea are used29. 1.3.1 The Morita-Baylis-Hillman mechanism The Morita-Baylis-Hillman reaction involves a three component, nucleophilic amine or phosphine mediated addition of an aldehyde to an activated olefin. The commonly accepted mechanism involves in the first step 1) a reversible conjugate (Michael-type) addition of the nucleophilic catalyst to activated olefin (acrylate). The resulting zwitterionic enolate (1)(Fig. 8) can subsequently undergo two pathways; elimination of the catalyst or an aldol-like nucleophilic attack on the aldehyde 2) giving the second zwitterionic intermediate (2). Third, the second zwitterionic intermediate can undergo now again two reaction pathways. Reaction pathway 3) yielding the product, comprises an elimination and transfer of a proton. The other pathway is the elimination of benzaldehyde end thereby the intermediate is returning into the first zwitterionic species (1). Direct evidence for this reaction mechanism was never presented; it was only based on assumptions. Only direct evidence for the zwitterionic species (1) was presented by Drewes and co-workers30 it was assumed that the rate limiting step 15 O H O- 1) OR H R3+N A NR3 O Ph H - O 3) OR B OR 1 O Ph R3+N PhCHO 2) Ph O H R3+N O H - H O H E R A OR 2 Fig 8 Morita-Baylis-Hillman mechanism (RLS) of the MBH reaction is the elimination/transfer of a proton step 3) since a species like (1) could be isolated and analysed by X-ray crystallography. In 2004, the groups of Coelho and Eberlin31, did a fundamental study, by electrospray ionization (ESI), detecting the intermediates in the MBH reaction. They confirmed the generally assumed MBH reaction mechanism. In 2005 the group of Aggarwal and co-workers32, published a study towards the kinetics and mechanism of the MBH reaction in a-protic solvents. Interesting results have been reported. During the MBH reaction when an excess of starting materials are present, the RLS is the elimination/transfer of the proton in step (3). When significant amounts of products are present, the RLS is 2. This is due to hydrogen bond donor capabilities of the product (Fig. 8, intermediate A), the product can promote, via hydrogen bonding, elimination and hydrogen transfer of step 3. This effect can be described as autocatalysis. 1.3.2 Morita-Baylis-Hillman Adducts Nowadays the Morita-Baylis-Hillman chemistry can be divided into three major research domains namely; 1) Improvement of rate, yield and elucidation of the reaction mechanism 2) Selective formation of the tertiary stereocenter 3) Chemical development of a various transformations of MBH adducts 16 Key developments of first two domains have been shortly overviewed in paragraph 1.3 and 1.3.1, the third, transformations of MBH products, will be discussed shortly in the following Ph OH O Lactamization Baker´s Yeast R OH HO OH R C9H19 HO O N EWG O OH Epoxidation Dihydroxylation R EWG R = mnitroO phenyl Hydrogenation Aminohydroxylation OH OH COMe N H OH R EWG OH NHTs Fig 9. Transfromations of Morita-Baylis-Hillman alcohols text. So far, in the literature many papers appeared where MBH adducts are used to obtain compounds with certain molecular properties. Therefore various efficient transformations of MBH products have been reported and methodologies have been developed. MBH adducts33 have proven to be important intermediates in various applications, such as total synthesis. As discussed before the MBH reaction relies on an atom economic process, yielding three functionalities is close proximity. These three functionalities, an alcohol, an alkene and an electron withdrawing group (usually an ester), are essential key functional groups in organic chemistry today. In principle, these key functionalities are susceptible of stereo-, chemo- and enantioselective transformations and therefore MBH adducts posses an enormous synthetic potential all together. Upon modification of MBH alcohols/adducts various known organic compounds classes can be obtained like; (γ-butyro-) lactones, β-lactams, quinolines, indolizines, methylene-dioxanones, pyrrolidines, coumarins, naphthalenes and derivatives thereof. Moreover, MBH adducts undergo (perfectly) a tremendous scope of classic reactions such as: asymmetric dihydroxylation (AD), epoxidation, ring-closing metathesis, aldol condensations, aminohydroxylation, radical cyclizations, Heck, Friedel-Craft and hydrogenation. Many other reductions and photochemical reactions also have been reported 25. In figure (Fig. 9), a small selection of successfully applied alterations is presented. Especially, for the construction of highly functionalized molecular skeletons, like natural compounds, MBH adducts can be interesting building blocks. MBH adducts have been often used multifarious in total synthesis, examples being (-)-mycestericin E by Hatakeyama et 17 al.34 (Fig. 10), Pinnatoxin A by Kishi et al.35(Fig. 11) and Salinosporamide A by Corey et al.36(Fig12). OH COOR C6H14 O O OH COOH C6H14 H2N O OH Fig 10 (-)-Mycestericin E H H O TESO O O NHAlloc O O H O HO + O H O O H MsO CO2tBu HN H O O H CO2OH OTBS Fig 11 Pinnatoxin A O BMP COOMe N OBn O Me O BMP COOMe N OBn Me OH O OH O H N Me O Cl Fig 12 Salinosporamide A: 18 1.4 Dynamic kinetic Asymmetric transformation (DYKAT) of MoritaBaylis-Hillman adducts Since the creation of highly optically enriched β-hydroxy-α-methylene esters (and ketones, nitriles etc.) still remains a barrier till to date, Trost and co-workers have introduced in 2000 an alternative strategy, the so called “dynamic kinetic asymmetric transformation” (DYKAT) of Morita-Baylis-Hillman adducts37. This deracemization reaction procedure forms one out of the at least five enantiodiscrimination mechanisms in the famous palladium catalyzed asymmetric allylic alkylation (AAA)38. This strategy consists of a palladium catalyzed dynamic kinetic asymmetric transformation (DYKAT) of racemic Morita-Baylis-Hillman carbonates as depicted in figure 13. Dynamic kinetic asymmetric transformation refers to the conversion of chiral racemic substrates having the potential of being completely converted to one single enantiomer of the product. The R OMoc EWG + NuH Pd Nu R Nu EWG or R EWG DYKAT mechanism can be explained by the next illustrative Fig 13 Dynamic kinetic asymmetric transformation” (DYKAT) of MoritaBaylis-Hillman adducts example. In the first of step of a DYKAT reaction, the oxidative addition of a chiral palladium complex to the olefin occurs. Subsequently ionization of the leaving group happens and a diastereomeric complex is formed. Next, there are two possible options for the formation of a diastereomeric complex. Usually one diastereomeric complex is more favoured for nucleophilic attack (oxygen nucleophile), followed by decomplexation leading to the product. The second diastereomeric complex usually does not react or in much slower rates. In a DYKAT reaction inter conversion of this “slow reacting” diastereo-complex/π-allylintermediate to the “fast reacting” diastereo-complex π-allyl-intermediate happens and subsequently reacts. So the theoretically yield in a DYKAT reaction can be 100% compared to 50% yield in a kinetic asymmetric transformation (KAT)39. A KAT reaction, also known as a kinetic resolution, refers to the transformation of one single enantiomer of racemic starting material into the product. The remaining other enantiomer of a substrate does not react or does it in slower rates. Since the introduction, the DYKAT of MBH-adducts has been frequently used by Trost et al. to overcome difficult key steps in the total synthesis of natural and biological active compounds. A whole array of compounds has been synthesized via this 19 methodology, examples being the total synthesis of (+)-Hippospongic acid A40(Fig. 15), morphine, codeine38b (Fig. 14), and Furaquinocin’s A, B, and E41 (Fig. 16). OR O MeOOC O N R=H, Morphine R=CH3, Codeine Fig 14 R=H Morphine, R=CH3 Codeine O O Fig 15 (+)-Hippospongic acid A [sc] R1 R2 [sc]= MeO OH R3 O A= R1=OH, R2=CH3, R3=CH2OH B= R1=OH, R2= CH2OH, R3=CH3 E= [sc]= OH Fig 16 Furaquinocin’s A, B, and E 20 1.5 Functionalization of Morita-Baylis-Hillman adducts via a tandem SN2’-SN2’ mechanism As shown before, simple Morita-Baylis-Hillman adducts possess a huge synthetic potential25. As a result, in recent years a lot of papers have been published dealing with the (enantioselective) modification of the β-position, the alcohol functionality of MBH adducts. A number of strategies have been developed in the recent years focusing on two different targets. One is the expansion of synthetic methodologies in order to obtain various MBH adducts. The other is the development of a highly enantioselective route for obtaining MBHalcohols, because the direct and highly enantioselective Morita-Baylis-Hillman reaction between a simple aldehyde and an activated olefin has proven to be a daunting challenge. In both processes, usually the MBH-alcohol is transformed into a leaving group. First the MBH reaction is performed without a chiral catalyst. Once the product is obtained, the MBH alcohol is transformed in typically an acetate or carbonate moiety. Now these MBH-adducts are prone to a tandem SN2’-SN2’ mechanism. This mechanism, which is comparable with the DYKAT transformations of MBH adducts, includes several steps. The first step is usually an attack of a nucleophilic (chiral) nitrogen or phosporus atom on the vinylic moiety of the MBH adduct occurs, with subsequent ionization of the leaving group. Once the leaving group has deprotonated the pro-nucleophile, the intermediate (zwitterion) is prone to nucleophilic attack, finally leading to the product. In the last five years a lot of papers appeared making use of this mechanism. The mechanism can be described as an allylic substitution reaction or, when OAc Ph COOMe (DHQD)2PHAL THF/H2O Ph COOMe NR3 OAc OH NaHCO3 Ph COOMe Ph COOMe Fig 17 Organocatalytic allylic substitution SN2’-SN2’ mechanism carbon/nitrogen nucleophiles are used, an allylic alkylation or aminaton. In 2002, Kim and coworkers42 published an organocatalytic methodology which relies on this tandem SN2’-SN2’ mechanism (Fig. 17),. MBH-acetates (racemic) are transformed into MBH-alcohols with optical purities of 54-92% ee and in yields of 25-42%. The process involves a successive SN2’-SN2’ attack of a quinidine derived Sharpless asymmetric dihydroxylation ligand, (DHQD)2PHAL, to the vinyl moiety, followed by attack of a hydroxy anion, leading to the product. The first step of the process occurs via a kinetic resolution step, (DHQD)2PHAL 21 reacts faster with one enantiomer of the starting material. Since the O obtained enantioselectivities obtained can not be only explained by a kinetic resolution step, the author proposed that during the second step O Ph NHTs COOMe there is additional asymmetric induction. Supplementary, Kim and coworkers reported in 2002 similar reactions relying on again SN2’-SN2’ mechanism. MBH-acetates were reacted with a sub-stoichiometric Fig 18 N-p-toluenesulfonimide derived MBH adduct amount of DABCO and various pro-nucleophiles. When pronucleophiles, such as tosylamide, methyl-sulfonamide, phthalimide, benzotriazole were used, the allylic Ph NHTs COOMe 44 year reported by Orena and co-workers . In this NHTs a substitution (amination) products were obtained in moderate yields (40-91%)43. Similar work was in that COOMe Ph Fig 19 a) product b SN2’-SN2’ product b) Michael elegant paper is shown the sensitivity of the MBH-adducts towards bases. When N-ptoluenesulfonimide derived MBH where reacted with a sub-stoichiometric amount of DABCO in DCM, the allylic substitution products (Fig. 19, compound a) were obtained via the tandem SN2’-SN2’ mechanism. On the other hand when DABCO was replaced by DBU, the SN2’-SN2’ mechanism does not occur. Now, a SN2’-decarboxylation mechanism occurs leading to product b, as depicted in figure 19. The author proposed that the results can be explained by the higher basicity (less nucleophilic) of DBU compared to DABCO. DABCO favours a nucleophilic SN2’-attack to the double bond instead of deprotonation of the N-ptoluenesulfonimide. MBH adduct. Basavaiah co-workers45 reported a similar methodology in order to obtain enantiomerically enriched Morita-Baylis-Hillman adducts. Again the methodology relies on a allylic substitution tandem mechanism (Fig. 20). First the H COOMe Br + QD (NR3) OH DCM, rt, 24 h H COOMe + O - N R3 Br O OMe Fig. 20 Basavaiah’s chiral leaving group methodology organocatalyst (quinidine) replaces the bromo atom, resulting in a chiral-intermediate, similar of kind intermediate as reported by Kim. Subsequently, the chiral intermediate is prone to attack by the pro-nucleophile, ultimately leading to the product. Unfortunately, the results obtained by Basavaiah are rather poor with yields up to 47% and enantioselectivities up to 40 % ee. 22 In 2004 the groups of Krische and Lu independently reported the first metal free organocatalytic allylic alkylation relying on the tandem SN2’-SN2’ mechanism in excellent yields and regioselectivity’s. The group of Krische reported the highly regioselective formation of γ-butenolides46. Various γ-butenolides where created through a phosphine catalyzed substitution of MBH-acetates. The diastereo-selectivity ratios obtained in this reaction were excellent, up to 20:1. The regioselectivities were good, in general higher than 9:1. In addition, the reported yields were excellent, up to 94%. The group of Krische reported as well a phosphine catalyzed allylic amination and dynamic kinetic resolution and of MBH acetates47. A protocol for the amination of several MBH-adducts by 4,5-dichlorophthalimide and phthalimide is presented. Besides racemic aminations, there is as well a chiral example shown, commercially available Cl-OMe-BIPHEP promotes the amination of MBH acetates with phthalimide in 56% ee. Lu and co-workers48 have described a β-IC catalyzed nucleophilic O O O O OEt Beta-IC Toluene, rt, 12 h Nu O O OEt + OEt Nu Fig. 21 Organocatalytic allylic nucleophilic substitution of Morita-Baylis-Hillman carbonates substitution of tert-butylcarbonate by various carbon, anime, oxygen and phosphorus nucleophiles (Fig. 21). The tandem SN2-SN2’ nucleophilic substitutions occurs in high regioselectivities and excellent yields. However, in general the enantioselectivities obtained are moderate. This methodology shows a great tolerance towards various nucleophiles including; nitrogen, phosphorus, sulphur, oxygen and carbon. The proposed mechanism includes (Fig. 22), first the addition of the nucleophilic chiral amine to β-methylene functionality (vinyl moiety). After the organocatalyst is added to the α-methylene function, a cascade of steps occurs. First the activating group leaves the starting material, leading to CO 2 and tert-butoxide. Sub sequentially, deprotonation of the pronucleophile by the tert-butoxide anion leads to a SN2’ attack of the pronucleophile on the chiral intermediate. During the attack of the pronucleophile, the organocatalyst is displaced from the intermediate giving the product and the regenerated catalyst. Lu and co-workers found that β-isocupreiidine was the catalyst leading to the highest enantioselectivities and reactivity’s. In the report there is one asymmetric example shown with a 1,3-di-carbonyl carbon pro-nucleophile producing the 23 expected product in a moderate enantioselection (51% ee), high SN2-SN2’ ratio (chemoselectivity) and high yield. OBoc O Ph OH OMe OMe Ph O H 1a O OBoc N N Nu Ph * N N -ICPD A O -CO2 OMe 3a t-BuOH t-BuO OMe Ph O H O * N N C OMe O H Nu Nu (E) O NuH 2a Ph N N B Fig. 22 Mechanism of the organocatalytic allylic nucleophilic substitution of Morita-Baylis-Hillman carbonates 24 1.6 All-carbon Quaternary stereocenters Synthetic chemists nowadays can create almost every tertiary stereocenter with excellent levels of enantiocontrol and chemical yields. Various methodologies/series of tailor made ligands have been developed and are used commonly in organic synthesis. However, catalytic enantioselective C-C bond formation of all-carbon quaternary stereocenters, i.e. carbon stereocenters bearing four different carbon substituents, still represents a tremendous challenge for synthetic organic chemists49. Moreover, when a carbon stereocenter is situated near a vicinal tertiary or quaternary stereocenter, the construction of these features become even more problematic. The difficulty for the construction of these motives arises often from steric hindrance and a limited amount of reliable reactions. Frequently used quaternary C-C bond formation reactions are; cycloadditions like Diels-Alder, Pd-allylations reactions and Michael additions, also known as conjugate additions. Nevertheless, the assembly of quaternary stereocenters still remains a considerably underdeveloped research area even though they are common motives in natural and pharmaceutical compounds. To overcome these difficulties, additional research is needed in the future to have access to complex molecular skeletons in an economic and straightforward manner. An alternative strategy in order to obtain quaternary stereocenters can be completed by organocatalysis 1. As a matter of fact, in the last years a number of organocatalyzed formations of these assemblies have been reported. A variety of organocatalysts like proline and cinchona derivatives effectively catalyzes the formation of quaternary stereocenters.50 An outstanding example of these assemblies is the cinchona catalyzed Michael addition reported by Deng and co-workers in 200550c. The addition of various pro-chiral tri-substituted carbon nucleophiles to nitro-olefins was reported in high enantioselectivities (up to >99 % ee) and diastereoselectivities (up to >98:2 %) (Fig. 23). The organocatalysts of choice were simple cupreidine derived catalysts. To stress the challenge of simultaneous arrangements of vicinal quaternary and tertiary stereocenters; to date only six examples have been reported. Interestingly, five of them are organocatalytic51. 25 3 R2 4 N 9 R1 R3 R2 R4 + R4 NO2 Cat 1,2,3 R1 R2 8 1 N NO2 6´ R1 R3 1 BnCPD 2 PHNCPD 3 CPD R1 = OH R2 = Bn R1 = OH R2 = Phn R1 = OH R2 = OH Fig. 23 Organocatalytic Michael addition leading to vicinal quaternary and tertiary stereocenters 26 Chapter 2 Allylic Nucleophilic Substitution of Morita-BaylisHillman Carbonates 2.1 Goal Enantioselective organocatalysis has revealed a extraordinary number of new methodologies in only half a decade1Although organocatalysis only recently found renewed interest, the hidden potential of organocatalysis does not require further explanation by a simple look a the literature of the past years. Since there is still a vast request for new complex chiral skeletons and many problems chemists have to deal with, are still unsolved, organocatalysis can serve as a real alternative in organic chemistry in academia and industry. The palladium catalyzed asymmetric allylic alkylation Pd-catalyzed allylic substitution 38 (AAA) , mainly developed by R1 Trost and co-workers, is one of the most prominent frequently used transition metal [Pd] EWG Pd and LG asymmetric catalyzed R1 Nu EWG LG Nu R1 * EWG reactions. As mentioned before in paragraph 1.4, the AAA has a wide scope and allows access to an incredible asymmetric amount of skeletons. The AAA, the organocatalytic XR3 (X=P,N) R1 EWG XR3 Organocatalytic allylic substitution Fig. 23 Palladium versus organocatalytic allylic substitution dynamic kinetic asymmetric transformation (DYKAT) of Morita-Baylis-Hillman adducts, described by Lu et al.48, is the first procedure tolerating a variety of carbon, oxygen, nitrogen and phosporus nucleophiles in excellent regioselectivities. In fact, Lu and co-workers are the first who established an organocatalytic allylic alkylation with carbon nucleophiles, when dimethyl malonate was used a ee of 51% was obtained. This enantioselectivity is moderate, leaving room for improvement. During this master research project we wanted to contribute, and further explore the potent and exciting research area of organocatalysis. We became interested in the organocatalytic allylic nucleophilic substitution of MBH adducts for the reasons described before, and wanted to investigate the further potential of this reaction. Since Lu and co-workers have shown that 27 1,3-di-carbonyl entities easily can act as pro-nucleophiles, we were interested whereas via CC bond forming process also quartenary stereocenters could be constructed. We wanted to expand the organocatalytic allylic substitution by using chiral trisubstituted carbon nucleophiles yielding in one step vicinal quaternary and tertiary stereocenters (paragraph 1.6) assembled MBH-adducts. In literature chiral trisubstituted carbon nucleophiles, like ethylphenyl-cyanoacetate, are used frequently to construct all-carbon or heteroatom quaternary stereocenters52. MBH-adducts are, as discussed before in paragraph 1.3, valuable synthons in various syntheses making this procedure even more attractive. Moreover, we wanted to construct a β-isocupreidine catalyst with a C6’ thiourea motive. This thiourea motive, discussed in paragraph 1.2.1, is a much better hydrogen bond donor compared to a C6’ hydroxyl group and therefore it can lead to higher enantioselectivities in the organocatalytic allylic nucleophilic substitution with carbon nucleophiles. 28 2.2 Catalyst synthesis To confirm the results described by Lu et al., we first started the synthesis of the catalyst β- H OH OH N 9 N KBr-H3PO4 (1) N 1,2-hydrogen shift N 100 oC, 5 d 6´ OMe OMe OH O N N cyclization N N 61% (2) demethylation OMe OH Fig. 24 Mechanism of β-isocupreidine synthesis isocupreidine. To date, the most convenient and high yielding synthesis of β-isocupreidine (2) was reported by Hoffman11 and Hatakeyama et al.17 β-Isocupreidine is formed in one step from quinidine (1) under highly acidic conditions. This process comprises a acid-induced cyclo-isomerization via a carbocation mechanism (Fig. 24). First, the vinyl moiety is protonated by HBr, leading to a O secondary carbocation. This is followed by a 1,2-hydrogen N shift, because a tertiary carbocation is more stable then a secondary. Next, attack of the C9 hydroxyl group on the N tertiary carbocation takes place, leading to cyclization to an NH oxo-aza-twistane. In addition, in situ demethylation occurs, leading to a bi-functional catalyst. Similar work-up procedures S NH were used as described by Hatakeyama and co-workers, although for an analytically pure batch of β-isocupreidine, the F3C compound was subjected to re-crystallization and after Fig. 25 β-IC-Th (6) CF3 purification, the batch was once again subjected to column chromatography. The obtained yield was similar to what is described by Hatakeyama et al. (61% yield). This reaction suffers unfortunately from side-product formation, a plausible 29 option is likely to be cyclization via the secondary carbocation intermediate. Next, we started the synthesis of the β-isocupreidine catalyst, equipped with a strong Brøndsted acid thioureamoiety (Fig. 26). We envisaged to synthesize the target structure, starting from βisocupreidine via a similar strategy as reported by Hiemstra et al.15. The next envisioned steps included, in chronical order, a C6’OH triflation, a palladium catalyzed Buchwald amination of the triflate and subsequent hydrolysis. Once the synthesis of C6’-NH2 functionality was achieved, we envisioned to react the C6’NH2 with 3,5-bis-trifluorophenyl-isothiocyanate, yielding the target structure. We started our synthesis from pure β-isocupreidine and performed the triflation step of the C6’ hydroxyl group with N-phenyl-bis(trifluormethane sulfonimide) catalyzed by a catalytic amount (10 mol%) of DMAP. The reaction was stirred for 6h in DCM at reflux. Flash chromatography afforded (3) as a pure compound in 47% yield. Next, we subjected (3) to a palladium catalyzed Buchwald amination in THF. The reaction went smoothly overnight at reflux conditions and the crude reaction mixture was O O O N N Ph-N-(OTf)2, 1.1 eq. N N 6´ Pd(OAc)2, 10 mol% N 6´ DMAP 10 mol%, DCM, reflux 6 h OH N HN=CPh2,1.1 eq. (+/-) BINAP, 10 mol% Cesiumcarbonate 1.5 eq., THF, reflux 18 h O O S O CF3 6´ N Ph (3) 47% (2) Ph (4) 76% THF/H2O citric acid O O N 3,5-CF3PhNCS, 1.05 eq. N 6´ F3C N THF, 20 min. NH S N 6´ NH2 NH CF3 (6) 69% Fig. 26 Synthesis of a β-isocupreidine equipped with C6’ thiourea moiety (5) 81% subjected to flash chromatography. Compound (4) was obtained as pure compound in 76 % 30 yield. The C6´-NH2 was obtained via hydrolysis of the imine with citric acid. The reaction went without problems and compound (5) was obtained as a single product in 81% yield. The final step of the synthesis includes a nucleophilic addition of the free amine to the electrophile 3,5-bis-trifluoro-phenyl-isothiocyanate. The substrates were stirred in THF and were allowed to react for 20 min. During the reaction, the product precipitated from the reaction mixture as a white solid. Compound (6) was obtained as a single product in 69% yield. Meanwhile, the synthesis of the catalyst was reported by Deng et al.53 The results reported by Deng matched with our results and observations. In order to examine the bi-functional character of βisocupreidine (2), which was shown to be fundamental in the direct Baylis-Hillman reaction, we have decided the synthesize β-isoquinidine (2a). This organocatalyst has a identical molecular skeleton as (2) but now the bi-functional character is removed from the catalyst. The synthesis of the catalyst was accomplished by using sodium hydride and methyl iodide. O N N (2a) OMe Fig. 27 β-isoquinidine 31 2.3 Preliminary experiments and optimization In order to check the results reported by Lu; we first wanted to start a reaction with dimethyl malonate and Morita-Baylis-Hillman carbonate (7). Therefore we needed to synthesize the MBH carbonate. Since the synthesis of the MBH carbonate was not reported by Lu, we had planned to synthesize the MBH alcohol via an adapted procedure of Aggarwal et al56. and subsequently the Boc protection via procedure Trost et al.54 (Fig. 28) The procedure of Aggarwal represents a simple neat MBH reaction between benzaldehyde O 1) DABCO 50 mol% MeOH 0.75 eq., 4 d O H OEt OEt + OBoc O 2) (Boc)2O 1.1 eq. DMAP 5 mol% DCM, 1h. (7) 34 % Fig. 28 Synthesis of a Morita-Baylis-Hillman Carbonate and ethyl acrylate, catalyzed by 50 mol% DABCO. 0.75 eq. of methanol was added as an additive to the reaction mixture, because of the hydrogen bond donor capabilities to zwitterionic species 2 (paragraph 1.3.1) of methanol, resulting in rate enhancement. The MBH alcohol was obtained after flash-chromatography in a yield of 83% (6.4 g.). Next, the Boc protection was performed of the MBH alcohol with Boc-anhydride. Catalytic amount of DMAP (5 mol%) was used and the reaction was left stirring for 1h, the reaction mixture was purified by flash chromatography, yielding compound (7) as a colourless oil in 42% yield. The yield was somewhat disappointing, probably due to a short reaction time (concluded from O OBoc O COOEt + MeO C, 20 mol% O OMe O MeO OMe COOEt Tolueen0.05 M 12 h, rt 1 eq 1.2 eq (7) Fig. 29 Organocatalytic allylic alkylation with dimethylmalonate (8) other experiments) or by attack of DMAP on the product and consequent liberation of the Boc activating group. Subsequently, we repeated the experiment (Fig. 29) reported by Lu to confirm the level of enantioselection and yield. The reaction was performed at a 0.1 mmol scale of MBH carbonate and was left stirring overnight, a drop of the reaction mixture was 32 filtered to remove the catalysts, the filtrate was analyzed by chiral HPLC. And indeed, the results reported by Lu et al. were reproducible, the level of enantioselection was 53% and the chemoselectivity was 97%. Next, we examined the role of the acrylate function in the carbonate. We made a MBH carbonate like (7) but employed with a methyl COOEt ester instead of an ethyl ester electron withdrawing group. Carbonate (9) CN was synthesized in a similar way as (7), although higher in a higher yield, (10) the compound (9) was obtained in a yield of 67% over two steps. The catalysis experiment was repeated with carbonate (9), and HPLC analysis revealed that the level of enantiodiscrimination was changed slightly; we observed an ee of 63% instead of 51% in case of (7). We found that the enantioselection process shows a concentration effect. We found an optimum at 0.05 M, a similar concentration as used by Lu and co-workers. A decrease or increase in concentration of the reaction mixture gave lower levels of asymmetric induction. Employment of diisopropyl malonate as pronucleophile OBoc O gave slightly lower levels of asymmetric induction, 58% ee. OMe Subsequently, we investigated the Brønsted acid functionality of the catalyst in the organocatalytic allylic alkylation. The thiourea derived (9) β-isocupreidine catalyst (6) was tested in a catalysis experiment under similar conditions as βisocupreidine itself. The reaction went to completion within 24 h, and had a lower level of asymmetric induction, 41% ee. Employment of other solvents (increased polarity) resulted in lower levels of asymmetric induction and disordered product formation. These results where in comparison as described by Lu, toluene remains the solvent of choice. We decided to lower the catalyst loading of (6) to 10 mol%, leading to a decreased polarity of the reaction medium. We where delighted to observe a significant higher ee of 58%. This result supports the idea that the enantioselection process of the organocatalytic allylic alkylation is favoured by an apolar reaction medium. Hence, we lowered the reaction temperature. The reaction with 10 mol% of (6) at -20 ºC gave almost no asymmetric induction, 14 % ee. Interestingly, βisocupreidine (2), gave 72% ee., although the reaction rate was not practical and did not go to completion after almost a week. These findings may suggest a reversed temperature dependency of Brønsted acid activation. After we had confirmed the results reported by Lu et al. and examined the role of the thiourea Brønsted acid moiety, we started to investigate the chiral compound ethyl-phenyl-cyanoacetate (10) as pro-nucleophile in the organocatalytic allylic alkylation. Employment of this chiral nucleophile should theoretically result in four different compounds, two diastereoisomers with their corresponding enantiomers. These four compounds complicate analysis by chiral HPLC, a prerequisite for enantiomeric excess 33 determination being baseline separation. Therefore, we started first an experiment, by using 1 eq of carbonate (9) and 1 eq. of pro-nucleophile (10) catalyzed by the a-chiral catalyst DABCO (50 mol%). Fortunately, the reaction went to completion in less then 30 min. Removal of the catalyst by a short filtration over silica and analysis of the mixture by chiral HPLC revealed that the 4 different compounds were separable. Next we started a similar catalysis experiment as in the case for dimethyl malonate, but we replaced dimethyl malonate by (10). To our delight the reaction went to completion in 18h. (Fig. 30) Analysis of the crude reaction mixture revealed an enantiomeric excess of the main diastereoisomer of 68%. For the determination of the diastereomeric ratio we used 1H-NMR and we observed a ratio of 3:1. Lowering the reaction temperature to -20ºC, gave an improvement of both diastereo- and enantioselectivity, dr 4:1 and 84% ee. However the reaction rate was not practical roughly 30% conversion in 72h. Moreover, there was an other serious drawback; we could not separate the remaining amount of pro-nucleophile (10) from the product (11) by column OBoc + 1 eq (9) C, 20 mol% CN COOMe Ph COOEt 1.2 eq (10) Ph CN COOEt COOMe Tolueen0.05 M 12 h, rt (11) Fig. 30 Organocatalytic allyic alkylation with a pro-chiral cyano-acetate, leading to vicinal quaternary end tertiary stereocenters chromatography. Consequently, we started two similar experiments at -20ºC; one experiment (a) with one equivalent carbonate (9) and 5 equivalents pro-nucleophile (10) and one experiment (b) with reversed ratios of the starting substrates, 5:1 respectively. We were delighted to observe a strong rate enhancement in both cases, experiment (b) went to completion after 22h. The levels of diastereomeric and enantiomeric control were similar for this experiment compared to earlier experiments. There was an important additional advantage observed in experiment (b), we could isolate the product after column chromatography. For further experiments we decided to make a compromise between reaction rate, atom economy and stereo chemical considerations of the organocatalytic allylic alkylation. We fixed the amount of substrate for further experiments, two equivalents of carbonate (9) and one equivalent of pro-nucleophile (10). During the optimization of the organocatalytic allylic alkylation we found that complete removal of Boc-anhydride is essential for obtaining high enantiomeric excesses. This is most likely explainable in terms of Boc protection of the catalyst, catalyst poisoning. 34 2.4 Results 2.4.1 Substrate scope We decided to continue our examinations with the employment of different electrophile and pro-nucleophile reaction partners. Therefore we needed first to synthesize various MoritaBaylis-Hillman carbonates (as depicted in table one) via the procedures of Aggarwal and Trost. The synthesis of the MBH-substrates went without significant problems and generally in moderate to good yields (47-83%). The pro-nucleophiles were commercially avaiable. All the reactions were performed as discussed in the end of paragraph 2.3. namely; two equivalents of carbonate, one equivalent of pro-nucleophile, 20 mol% β-isocupreidine and at a concentration of 0.05 M. In general, what be can be concluded from table 1 is that compounds 11a-f were obtained in high yields and good enantioselectivities. A closer look at the table reveals the sensitivity of the pro-nucleophile with respect to the selected carbonate substrate, hindered substrates (10b and 10d, entries 2 and 4) required higher temperatures to obtain satisfying reaction rates; fortunately, the level of enantioselection was not negatively affected. A low enantiomeric excess and diastereomeric excess was observed for substrate 9d, possibly due to its high reactivity towards conjugate addition possibly, the electron withdrawing pnitro group interferes with the hydroxyl functionality of the catalyst. Table 1 Substrate scope EtOOC OBoc R1 R2 CN * COOMe + EtOOC CN R2 9a-f 10a-b R1 Ph (9a) 2-MeC6H4 (9b) 3-ClC6H4 (9c) 4-NO2C6H4 (9d) 2-naphtyl (9e) 3-BrC6H4 (9f) R2 Ph (10a) Me (10b) -ICPD (20 mol %) toluene R * COOMe 11a-f 35 Entry Product CN Ph T [ºC] t [h] Yield [%]b drc ee [%]d -20 48 94 4:1 83 0 96 95 4:1 79 -20 72 95 1.4:1 80 -20 48 95 1.1:1 16 20 24 66e 4:1 85 -20 72 95f 3:1 80 COOEt 1 COOMe 11a CN Ph COOEt 2 COOMe 11b CN Me COOEt COOMe 3 11c Cl CN Ph COOEt 4 COOMe 11d O2N Ph CN COOEt 5 COOMe 11e Ph CN COOEt COOMe 6 11f Br a Conditions: 0.6 mmol 9, 0.3 mmol 10, 0.06 mmol β-ICPD in 6 mL toluene. b Isolated yield (mixture of diastereomers). c Determined by 1H-NMR analysis of the crude reaction mixture. d ee of the major diastereoisomer determined by chiral HPLC. e Compound 11e was obtained as a pure diasteromer. f Reaction performed on 9.0 mmol of 9f An important prerequisite of an asymmetric methodology is the possibility to upgrade the optical purity, if the levels of enantioselection are not excellent. Since some adducts were 36 obtained as crystalline solids after chromatography, we run a reaction on a gram scale with the aim to upgrade the optical purity of the product via recrystallization. Hence, compound 9f (3.3 g, 9.0 mmol) was reacted with cyanoacetate 10a (0.78 mL, 4.5 mmol) to afford, after column chromatography, adduct 11f in 80% ee and 95% yield (entry 7). A single recrystallization from cyclohexane afforded compound 11f (59% yield, calculated on cyanoacetate 10a) with 98% ee as a pure diastereomer. In addition, a larger scale reaction between compound 9a (0.88 g, 6.0 mmol) and 10a (0.52 mL, 3.0 mmol) yielded adduct 11f after column chromatography in 86% ee and 95% yield. A single recrystallization from cyclohexane afforded compound 11f (63% yield, calculated on cyanoacetate 10a) with 99% ee as a pure diastereomer. Instead of employment of a prochiral trisubstituted cyanoacetate as pro-nucleophiles, we investigated the use of prochiral trisubstituted 1,3-dicarbonyl moieties. We decided to use ethyl 2-oxocyclohexanecarboxylate as reaction partner (Fig. 31). The mixture was allowed to react for 48 h at room temperature and the reaction was performed O O OBoc Ph COOMe OEt + COOEt O -ICPD (20 mol %) Ph COOMe * toluene, rt, 48 h. 12 13 Fig. 31 Organocatalytic allyic alkylation with 1-3 dicarbonyl moiety under similar conditions as used in table 1 (Fig. 31). Analysis of the crude reaction mixture revealed full conversion of the pronucleophile, yielding the postulated product. We were glad to observe an excellent diastereomeric ratio of >98:2 however the enantiomeric excess was moderate: 61% ee. Purification by column chromatography afforded 13 as a single diastereoisomer. Nevertheless, we were not able to determine the yield since the resulting compound 13 was not stable. The compound was prone to decomposition in CDCl3. Employment of ethyl 2-methyl-3-oxobutanoate as pro-nucleophile gave immediately disappointing results and we were not able to isolate a pure compound. 37 2.4.2 X-Ray analysis Since we could crystallize two products in pure form, we submitted the crystals for X-ray analysis. From these crystals we could assign unambiguously, the absolute and relative configuration of compound 11f. (Fig. 32) Both stereocenters have the R configuration. From compound 11a it was only possible to assign the relative configuration. The absolute configuration of compound 11a could not be reliably determined because of the lack of a heavy atom. The relative configuration of this Fig. 32 ORTEP drawing of product 11f Ph CN COOEt (R) (R) -ICPD (20 mol %) + 10a 9f toluene, 72h, -20 ºC (2 equiv.) (1 equiv.) OBoc (S) COOR COOMe + 11f Br 80% ee Br 9f 90% ee Fig. 33 Kinetic resolution effect of carbonate 9f compound was analogous to compound 11f, (Fig. During 34). column chromatography of the reaction mixture of compound 11f, we could recover the excess of the starting material used. Interestingly, HPLC analysis of the recovered starting material (9f) gave evidence for a kinetic resolution effect. Compound 9f was isolated in 90% ee. Recrystallization afforded Fig. 34 ORTEP drawing of product 11a 38 enantiopure crystals (47% yield based on 10a). X-Ray analysis revealed the absolute configuration, S (Fig. 35). From this result we can conclude that the catalyst has a preference for a MBH-carbonate bearing the R configuration. As suggested by Kim and coworkers,42 who observed a similar effect, this can be explained in terms of a kinetic resolution in the first step. This process, however, does not affect the enantioselectivity of the second conjugate addition generating the C-C bond. To confirm this statement we examined a catalytic experiment of 1 equivalent of 9a with 2 equivalents of 10a at room temperature (hence reversing the reagents ratio): after 24 h, 9a was completely converted to 11a with similar stereoselectivity (68% ee, 3:1 dr), showing that both enantiomers of the MBH carbonate undergo Michael addition although with different rates, followed by elimination of the leaving group leading to the same intermediate. Fig. 35 ORTEP drawing recorvered starting material 9f 39 2.4.3 Mechanistic considerations The importance of employment of bi-functional catalysts in organocatalysis, for obtaining high(er) enantioselectivities and reactivities, has been shown in numerous examples. As well in the asymmetric Morita-Baylis-Hillman reaction, the bi-functional character of βisocupreidine was shown to be fundamental. We believe that the bi-functional character of the catalyst is also essential in the organocatalytic allylic alkylation of MBH carbonates. In order to confirm this assumption we started a catalytic experiment with β-isoquinidine (2a) (β-IQD) as the catalyst. Indeed, the role of the C6’-OH of β-ICPD is crucial for obtaining high enantioselectivities. We observed a drastically lower enantiomeric excess of 30%. Furthermore, less than 5% conversion was observed after two weeks at room temperature, suggesting that the C6’-OH of β-ICPD is also crucial for activation of the system. Additional evidence was acquired via substrate 14, featuring a cyano group, a poorer hydrogen bond acceptor, instead of a methyl ester. We started a similar experiment as in case of the other allylic substitution experiments (table 1) and MBH electrophile 14 underwent the desired reaction (Fig. 36). As result, product 15 was obtained in high yield but no asymmetric induction was observed (Scheme 4). Ph OBoc COOMe a-IQD (20 mol %) Ph toluene, rt Ph 1a CN * COOEt COOMe O 3a <5% conv after 14 d N N Ph OBoc CN Ph a-ICPD (20 mol %) Ph toluene, -20 ºC, 48h 14 CN * COOEt CN 6' OR R=H -ICPD R=Me -IQD 15 95% yield, <2% ee Fig. 36 Effects of the structure of catalyst and substrate On the basis of all these observations, we propose a tentative hypothesis to rationalize our experimental results (Scheme 5). First, substrate 9a undergoes a conjugate addition to form adduct A (fig. 22, paragraph 1.5) . Subsequently, elimination of the OBoc group, leading to 40 the formation of CO2 and tert-butoxide anion provides Michael acceptor (B) (fig. 22, paragraph 1.5). We believe that this irreversible reaction step is responsible for the observed kinetic resolution. As shown in the experiment with β-isoquinidine, the presence of the C6’OH in the catalyst is required for activation of the ester prior to conjugate addition. As already proposed by Lu, the tert-butoxide anion deprotonates cyanoacetate 10a which, in turn, attacks the -unsaturated intermediate (β-ICPD and MBH carbonate). Probably, an intramolecular hydrogen bond between the phenolic OH and the ester moiety is responsible for the face shielding conferring stereoselectivity to the reaction; a DFT-minimized (Fig. 37) structure B (fig. 22) is supporting this hypothesis. Elimination of the catalyst from intermediate C liberates alkylated product 11a, closing the catalytic cycle. nucleophile Nu (R) Ph COOMe 1.97 Å Fig. 37 Minimized structure of the intermediate, β-ICPD and MBH carbonate (B3LYP/6-31G**//HF/321G*) 41 2.5 Conclusions In conclusion, we have developed new methodology that allows access to highly functionalized MBH adducts, featuring two vicinal stereocenters, one of them bearing four carbon substituents. These MBH-adducts can be created via a one step organocatalytic allylic alkylation of MBH carbonates and are obtained in excellent yields and chemo/enantio/ diastereo-selectivities. Moreover, this is the first, metal free, one-step creation of vicinal quaternary and tertiary stereocenters that is not completely explainable by a conjugate addition mechanism. The methodology could be extended to a broad scope of MBH aryl electrophiles, ranging from hindered to activated substrates. The role of the pro-nucleophiles was also investigated; compound 2a and 2b react with similar levels of asymmetric induction. However compound 2b was obtained in a lower diastereoselectivity, a plausible explanation being that steric-bulk is vital for obtaining high diastereoselectivities. Subsequently, we have shown for two examples that optically pure compound could be obtained, making this reaction practically executable. The crystals obtained for compounds 9f and 11f allowed assignment of the absolute configuration. For compound 11a only the diastereoselectivity could be determined. During our investigations we found evidence for the reaction mechanism. The reaction occurs most likely via two mechanisms, starting with a kinetic resolution followed by enantioselective attack of the chiral nucleophile. In addition, we showed that the carbonate that bears the R configuration, preferentially reacts with the catalyst. We also demonstrated the involvement of the OH-functionality of the catalyst in the catalytic cycle. The carbonyl moiety is vital to obtain highly enantiomerically enriched compounds. Future investigations could be aimed at broadening the scope of this transformation by employing alkyl instead of aryl electrophiles. Further design of catalysts that catalyze this reaction in higher regioselectivity’s and enantioselectivities and thereby improving the atom economy (less side-products). A detailed study towards the reaction mechanism could assist in the rational design of catalysts and lead to better understanding of this reaction. Additional research could be performed by investigating the importance of the electron wtihdrawing group of the carbonate substrate for the nucleophilic attack of the catalyst. If this reaction could be catalyzed with first an a-chiral base, for instance with quinuclidine, it would provide a immense step forwards in the metal free AAA. 42 Samenvatting Organokatalyse, katalyse met kleine organische moleculen, is in de afgelopen jaren opnieuw in de belangstelling gekomen van de academische wereld en de chemische industrie. Hoewel het al bijna een eeuw bekend is dat organische verbindingen asymmetrische reacties kunnen katalyseren, het is pas sinds enkele jaren dat dit onderzoeksgebied volledig wordt verkend. In de behandeling van ziektes is meestal èèn enantiomeer van een medicijn actiever, en kan in het slechtste geval, het andere enantiomeer tot ernstige bijwerkingen lijden of zelfs tot de dood. Daarom zijn de farmaceutische en chemische industrie continue opzoek naar nieuwe, efficiënte en betrouwbare asymmetrische reacties voor de constructie van moleculaire skeletten. De constructie van dergelijke asymmetrische, hoog gefunctionaliseerde moleculaire skeletten laat tegenwoordig veel te wensen over, helemaal wanneer er een quaternair stereocentrum in het molecuul is vertegenwoordigd. Organokatalyse kan hierin waardevolle nieuwe reacties bieden en kan daarnaast als een groen alternatief dienen voor bestaande metaal gekatalyseerde reacties. In dit verslag rapporteren wij een nieuwe asymmetrische èènstaps reactie van Morita-Baylis-Hillman adducten, die leiden tot moleculaire assemblages met een aangrenzend quaternair en tertiar stereocentrum. De uit quinidine, in èèn stap verkrijgbare organokatalysator β-isocupreidine, katalyseert effectief deze asymmetrische omzetting in goede chemo-, diastereo- en enantioselectiviteit. De opbrengst van deze reactie is uitstekend. Deze èèn-staps constructie van een aangrenzend quaternair en tertiar stereocentrum is een zeldzame reactie, tot op heden zijn er slechts zes bekend in de literatuur. Wat deze reactie uiterst uniek maakt is dat voor het eerst dit structuur niet via een conjugaatadditie worden gemaakt, maar via een organokatalytische alkylering. 43 Acknowledgements From April 2005- August 2006 I have performed my master research project in the research group of Prof. Dr. Henk Hiemstra, Synthetic Organic Chemistry, University of Amsterdam. I would like to thank first of all Henk and Jan for allowing me to perform my research project in the group. I would like to thank all the group members during my research period, thank you all for the great time. In particular, I would like to thank the “crazy” Italian Tommaso, for daily supervision, endless discussions, and talks about everything…… I appreciated it a lot that I could interfere in your PhD project about organocatalysis. Furthermore; Prof. Dr. Reek (Joost) for being the second corrector of my project, Han Peeters for recording the exact masses, Jan Geenevasen for daily assistance and for solving the problems with NMR/IR machine(s), Richard for fruitful discussions, Maxim for the “fun” on the lab and Jaap for exploiting the canteen in building D. Thanks a lot! At last but not least, I would like to thank my mother, my sister and Federica. 44 Chapter 3 Experimental General: All reagents were purchased from commercial suppliers (Aldrich and Acros Organics) and used without further purification, unless otherwise stated. Ethyl 2cyanopropanoate (2b) was obtained from TCI. Solvents where obtained from Biosolve. Heptane was obtained from Merck. Cyclohexane was obtained from Rathburn. Toluene was distilled from sodium and stored over 4Å molecular sieves prior to use. Column chromatography was performed using silica (0,035-0,070 mm pore diameter ca. 6 nm) obtained from Biosolve. NMR spectra (1H and 13 C) were measured in CDCl3 on a Bruker ARX 400 MHz spectrometer, CDCl3 was used as solvent. High-resolution mass spectra were recorded on a JEOL JMS-SX/SX 102 A tandem mass-spectrometer. Infrared spectra were -Isocupreidine (β-ICPD) and βisoquinidine (β-IQD) were prepared according to Hatakeyama et al.[55] β-Isocupreidine (β-ICPD) (2) Quinidine (1) (11.6 g, 35.8 mmol, 1 eq) was added to a solution of 85% O N N OH H3PO4 (175 ml) and KBr (42.8 g, 330 mmol, 10 eq.). The resulting mixture was stirred at 100 ºC for 5 days and finally cooled to 0 ºC. To the mixture was dropped carefully an ice cold solution of KOH (25 wt %, 700 ml), the PH was adjusted to 8 by using K2CO3 and extracted with CHCl3. The resulting orange organic layer was washed with brine (2 × 200 ml), dried over MgSO4 and concentrated in vacuo. Flash chromatography (MeOH-CHCl3 1-9) afforded (7.2 g, 23.2 mmol, 65%) as a off white solid. Spectroscopically pure compound was obtained by recrystallization from MeOH-H2O and an additional purification by column chromatography. β-ICPD was obtained as a light yellow solid. (7.3 g, 23.3 mmol, 65 %) 1H NMR (CDCl3, 400 MHz) δ 8.71 (d, 1H, J=4.4 Hz), 8.00 (s, 1H), 7.97 (d, 1H, J = 8.8 Hz) 7.57 (dd, 1H, J=4.5, 1 Hz), 7.25 (dd, 1H, J=9.0, 2.5 Hz), 6.00 (s, 1H), 3.68 (d, 1H, J=13.5 Hz), 3.46 (d, 1H, J=6.0 Hz), 3.19 (dd, 1H, J=13.0, 8.5 Hz), 3.09-3.02 (m, 1H), 2.77 (d, 1H, J=14.0 Hz), 2.24 (t, 1H, J=10 Hz), 1.93-1.84 (m, 1H), 1.81-1.64 (m, 4H), 1.30-1.25 (m, 1H), 1.05 (t, 3H, J=14.8 Hz). C NMR (CDCl3, 400 MHz) δ 156.8, 146.8, 143.3,141.5, 131.5, 127.1, 122.5, 119.0, 105.6, 13 76.9, 72.5, 56.6, 54.0, 46.4, 32.8, 27.4, 23.2, 23.0, 7.3. 45 β-Isocupreidine derived with trifluoromethylsulfonyl group (3) β-isocupreiidine (β-ICPD) (2) (5.0 g, 16.1 mmol) was added to a solution O N N O O S F O F F of N-phenyl-bis(trifluormethane sulfonimide) (6.3 g, 17.7 mmol, 1.1 eq) and 4-di-methylaminopyridine (DMAP) (0.2 g, 1.6 mmol, 10 mol %) in CH2Cl2 (35 ml). After being stirred for 5h at reflux temperature, CH2Cl2 was removed by rotary evaporation and the reaction mixture was dissolved in EtOAc and washed with sodiumhydrogencarbonate (5 × 75 ml). Flash chromatography (EtOAc/MeOH/NH3 25% in H2O), 92-6-2) afforded 3 as a yellow foam. (3.4 g, 7.6 mmol, 47 %) (IR (CHCl3) cm-1: 2968, 1730, 1675, 1598, 1511, 1457, 1426, 1376, 1141, 1098, 1053, 1007, 978, 932, 912, 887, 846, 829. 1H NMR (CDCl3, 400 MHz) δ 9.01 (d, 1H, J=4.4 Hz), 8.23 (d, 1H, J=9.6 Hz), 8.04 (d, 1H, J=2.8 Hz), 7.82 (dd, 1H, J=4.4 Hz, 0.8 Hz), 7.62 (dd, 1H, J=9.2 Hz, 2.4 Hz), 5.97 (s, 1H), 3.61 (d, 1H, J=13.6 Hz), 3.49 (d, 1H, J=6.0 Hz), 3.06 (dd, 2H, J=8.4, 4.0 Hz ), 2.74 (d, 1H , J=13.6 Hz), 2.19-2.17 (m, 1H), 1.771.66 (m, 4H), 1.60-1.54 (m, 1H), 1.34 (q, 1H, J=19.8 Hz, 12.8 Hz, 6.4 Hz), 1.04 (t, 3H, J=15.2 Hz). 13C NMR (CDCl3, 400 MHz) δ 151.5, 147.2, 147.0, 145.1, 133.3, 129.4, 126.1, 118.8 (q, 479 Hz), 115.5, 77.6, 73.1, 56.9, 54.7, 46.7, 33.0, 27.5, 24.2, 23.5, 7.4. HRMS (FAB, MH+) calcd. for C20H22N2O4F3S : 443.1252 observed; 443.1258. β-Isocupreidine derived with diphenylmethamine group (4) To a stirred solution of (3) (1.5 g, 3.39 mmol) in THF (15 ml) at 70 ºC was added stepwise Pd(OAc)2 (76 mg, 0.34 mmol, 10 mol %), BINAP O N N (0.32 g, 0.51 mmol, 1.5 eq. with respect to Pd(OAc)2) and cesiumcarbonate (1.65 g, 5.1 mmol, 1.5 eq. with respect to the starting N material). Finally, benzophenone imine (0.67 g, 3.7 mmol, 1.1 eq. with respect to the starting material) was added and the reaction mixture was left stirring for 24 h. The reaction mixture was allowed to cool down to room temperature, filtered through Celite pad using DCM as the eluens. The solvents where removed by rotary evaporation and the residue was subjected to column chromatography (EtOAc/MeOH/NH3 (25% in H2O), 92-6-2). The eluens was remopved by rotary evaporation yielding 4 as yellow foam.(1.25 g, 2.6 mmol, 76 %) IR (CHCl3) cm-1: 2967, 2884, 1675, 1612, 1596, 1574, 1499, 1446, 1377, 1317, 1291, 1244, 1140, 1099, 1053, 1006, 959, 909, 46 851. 1H NMR (CDCl3, 400 MHz) δ 8.76 (d, 1H, J=4.8 Hz), 7.94 (d, 1H, J=8.8 Hz), 7.78-7.76 (m, 2H), 7.65 (dd, 1H, J=4.4, 0.8 Hz), 7.51-7.47 (m, 1H), 7.43-7.39 (m, 2H), 7.25 (dd, 1H, J=8.8, 2.0 Hz), 7.21-7.14 (m, 5H), 7.05 (d, 1H, J=2 Hz), 5.73 (s, 1H), 3.43 (d, 1H, J=13.2 Hz), 2.99 (d, 1H, J=6 Hz), 2.96-2.94 (m, 1H), 2.61 (d, 1H, J=13.6 Hz), 2.08 (m, 1H), 1.641.58 (m, 4H), 1.47-1.46 (m, 1H), 1.25 (t, 2H, J=14.4 Hz), 1.11 (dd, 1H, J=12.4, 6.4 Hz), 1.00 (t, 3H, J=14.8 Hz). 13 C NMR (CDCl3, 400 MHz) δ 169.3, 149.8, 148.6,145.1, 144.0, 139.2, 135.8, 131.0, 130.7, 129.5, 129.5, 128.5, 128.2, 128.0, 125.8, 125.0, 119.1, 111.4, 77.2, 73.1, 56.3, 55.0, 46.8, 32.7, 27.3, 24.3, 23.7, 7.3. HRMS (FAB, MH+) calcd. for C32H32N3O : 474. 2545 observed; 474.2547. Amine (5) To a stirred solution of (4) (1.0 g, 2.21 mmol) in THF (5 mL) was added a O N N NH2 solution of citric acid (10 mL, 10% in H2O). The reaction mixture was left stirring for 24 h. Work-up procedures can be found in the manuscript published by Deng et al.53 Yellow foam. (0.55 g, 1.78 mmol, 81 %) IR (CHCl3) cm-1: 3400, 2966, 2884, 1632, 1593, 1516, 1473, 1356, 1281, 1253, 1178, 1142, 1099, 1053, 1006, 978, 909, 854, 827. 1 H NMR (CDCl3, 400 MHz) δ 8.66 (d, 1 H, J=4.4 Hz), 7.89 (d, 1H, 8.8 Hz), 7.61 (dd, 1H, J= .4, 0.8 Hz), 7.10 (dd, 1H, J=8.8, 2.4 Hz), 7.04 (d, 1H, J=2.4 Hz), 5.86 (s, 1H), 4.14-4.08 (m, 3H), 3.56 (d, 1H, J= 13.6 Hz), 3.50 (d, 1H, J= 6 Hz), 3.00 (dd, 2H, J=8.8, 4.0 Hz), 2.67 (d, 1H, 13.6 Hz), 2.13 (t, 1H, J=10.4 Hz), 1.791.74 (m, 1H), 1.67-1.62 (m, 3H), 1.53-1.49 (m, 1H), 1.02 (t, 3H, 14.8 Hz). 13 C NMR (CDCl3, 400 MHz) δ 146.4, 145.1, 142.9, 141.6, 131.5, 126.9, 120.9, 119.1, 102.4, 77.2, 73.1, 56.3, 54.7, 46.7, 32.9, 27.4, 24.2, 23.5, 7.4. HRMS (FAB, MH+) calcd. for C19H24N3O : 310.1919 observed; 310.1918. 47 bistrifluoro-thiourea (6) to a stirred solution of (5) (250 mg, 0.81 mmol) in THF (10 mL) was added 3,5-bis-trifluoromethyl-phenyl-isothiocyanate (155 μL, 0.85 mmol, O 1.05 eq.) The reaction mixture was left stirring for 20 minutes and N subsequently, the reaction mixure was cooled to 0 ºC. The white N precipitate was filtered and dried. The residue was subjected to column NH S NH F3C chromatography (EtOAc/MeOH/NH3 (25%in H2O)). Combined fractions CF3 yielding (6) as a white solid. (332 mg, 0.55 mmol, 69 %) IR (CHCl3) cm1 : 3093, 2971, 2885, 1621, 1595, 1550, 1508, 1473, 1384, 1326, 1301, 1279, 1180, 1141, 1099, 1052, 1008, 977, 886, 851. 1H NMR (CD3OD, 400 MHz) δ 8.84 (d, 1H, J=4.8 Hz), 8.26 (s, 2H), 8.06-7.94 (m, 3H), 7.84 (d, 1H, J=4.4 Hz), 7.70 (s, 1H), 3.56 (s, 1H), 5.95 (s, 1H), 4.90, (s, 1H), 3.52 (d, 1H, J=6.8Hz), 3.04-3.00 (m, 2H), 2.87-2.83 (m, 1H), 2.17 (s, 3H), 1.79-1.69 (m, 4H), 1.60-1.53 (m, 1H), 1.40-1.37 (m, 1H), 1.05 (t, 3H, J=7.2 Hz). C NMR (CDCl3, 400 MHz) δ. HRMS (FAB, MH+) calcd. for C28H27N4OF6S : 581.1810 13 observed; 581.1802. [α]D25 = 77.1 (c 1.0, CHCl3), Litrature value: [α]D25 = 76.7. Mp: 155-157 ˚C. Procedure for the preparation of the MBH-alcohols: Morita-Baylis-Hillman alcohols were prepared according to Aggarwal et al.56 To a round bottom flask charged with MeOH (0.75 eq.) was added the arylaldehyde (1 eq.) and methyl acrylate (1.2 eq.). To the solution was then added 1,4-diaza-bicyclo[2.2.2]octane (50 mol %) and the solution was stirred for 48-96 h. The crude reaction mixture was purified by column chromatography. (PE/Et2O or PE/EtOAc mixtures). methyl 2-(hydroxy(phenyl)methyl)acrylate 1 OH COOMe H-NMR (CDCl3, 400 MHz): δ 7.40-7.30 (m, 5H), 6.35 (t, J=1.6 Hz, 1H), 5.88 (t, J=2.4 Hz, 1H), 5.56 (s, 1H), 3.71 (s, 3H), 3.36 (s, 1H). C-NMR (CDCl3, 400 MHz): δ 166.7, 142.0, 141.3, 128.3, 127.7, 13 126.6, 125.9, 72.9, 51.9. ethyl 2-(hydroxy(phenyl)methyl)acrylate 48 1 OH COOEt H-NMR (CDCl3, 400 MHz): δ 7.41-7.34 (m, 4 H), 7.32-7.28 (m, 1H), 6.36 (t, J=0.8 Hz, 1H), 5.84 (t, J=1.2 Hz, 1H), 5.58 (s, 3H), 4.19 (q, J=21.6, 7.2 Hz, 2H), 3.18 (bs, 1H), 1.26 (t, J=14.4 Hz, 3 H). 2-(hydroxy(phenyl)methyl)acrylonitrile 1 OH CN H-NMR (CDCl3, 400 MHz): δ 7.45-7.36 (m, 5H), 6.12 (s, 1H), 6.04 (s, 1H), 5.29 (s, 1H), 2.70 (bs, 1H). 13 C-NMR: (CDCl3, 400 MHz) δ 139.2, 129.7, 128.7, 128.6, 126.4, 126.2, 116.9, 73.8. methyl 2-((3-chlorophenyl)(hydroxy)methyl)acrylate 1 OH COOMe H-NMR (CDCl3, 400 MHz): δ 7.38 (s, 1H), 7.28 (m, 3H), 6.37 (s, 1H), 5.87 (s, 1H), 5.52 (s, 1H), 3.74 (s, 3H), 3.33 (s, 1H). 13C-NMR (CDCl3, 400 MHz): δ 166.6, 143.4, 141.4, 134.4, 129.7, 128.0, 126.8, 126.7, 124.8, 72.7, 52.1. Cl methyl 2-(hydroxy(4-nitrophenyl)methyl)acrylate 1 OH COOMe H-NMR (CDCl3, 400 MHz): δ 8.18 (dd, J=6.8, 2.0 Hz, 2H), 7.56 (dd, J=7.2, 1.6 Hz, 2H), 6.38 (s, 1H), 5.88 (d, J=0.8 Hz, 1H), 5.62 (s, 1H), 3.73 (s, 3H), 3.33 (s, 1H). O2N 13 C-NMR (CDCl3, 400 MHz): δ 166.4, 148.7, 147.5, 141.0, 127.4, 127.2, 123.6, 72.6, 52.2. methyl 2-(hydroxy(naphthalen-5-yl)methyl)acrylate 1 OH COOMe H-NMR (CDCl3, 400 MHz): δ 8.03-8.01 (m, 1H), 7.90-7.87 (m, 1H), 7.83 (d, J=8.0 Hz, 1H), 7.66 (d, J=7.2 Hz, 1H), 7.52-7.48 (m, 3H), 6.40 (s, 1H), 6.37 (s, 1H), 5.59 (t, J=2.0, 1H), 3.80 (s, 3H), 3.11 (s, 1H). 13 C-NMR (CDCl3, 400 MHz): δ 167.4, 141.9, 136.4, 133.9, 130.9, 128.9, 128.7, 127.4, 126.3, 125.7, 125.5, 124.6, 123.9, 69.5, 52.2. methyl 2-((3-bromophenyl)(hydroxy)methyl)acrylate 1 OH COOMe H-NMR (CDCl3, 400 MHz): δ 7.56 (t, J=3.2 Hz, 1H), 7.45-7.42 (m, 1H), 7.34-7.32 (m, 1H), 7.26-7.22 (m, 1H), 6.39 (s, 1H), 5.87 (s, 1H), 5.54 (s, 1H), 3.76 (s, 3H), 3.15 (s, 1H). 13 C-NMR (CDCl3, 400 MHz): δ 166.6, 143.7, 141.4, 131.0, 130.1, 129.7, 126.9, 125.3, Br 49 122.7, 72.9, 52.2. methyl 2-(hydroxy(o-tolyl)methyl)acrylate 1 OH COOMe H-NMR (CDCl3, 400 MHz): δ 7.45-7.43 (m, 1H), 7.27-7.17 (m, 3H), 6.35 (s, 1H), 5.83 (s, 1H), 5.63 (s, 1H), 3.79 (s, 3H), 2.65 (bs, 1H), 2.35 (s, 3H) 13 C-NMR (CDCl3, 400 MHz): δ 167.2, 141.7, 138.8, 135.7, 130.5 127.9, 126.3, 126.3, 126.2, 69.3, 52.1, 19.1. General procedure for the preparation of MBH-carbonates (9a-f and 14) To a solution of the Morita-Baylis-Hillman alcohol (1 eq.) in DCM (0.6 M) was added Boc2O (1.05 eq.) and 4-dimethylaminopyridine (10 mol %). The solution was stirred for the indicated time and subsequently the solvent was removed by rotary evaporation. The reaction mixture was purified by column chromatography (PE/Et2O or PE/EtOAc). Finally, the Morita-BaylisHillman carbonate was left under high vacuum at 60 ºC to remove eventual traces of unreacted Boc2O. The purity of all Morita-Baylis-Hillman carbonates 9a-f was confirmed by 1 H-NMR. 2-(methoxycarbonyl)-1-phenylallyl tert-butyl carbonate (9a) Reaction time 90 min. Purified by column chromatography O O (PE/Et2O 2:1) White solid. (1.36 g, 4.6 mmol, 83%). FTIR O (CHCl3) cm-1: 3027, 2985, 1741, 1632, 1440, 1396, 1371, 1281, COOMe 1255, 1156, 1086, 962, 882 cm-1. 1H-NMR (CDCl3, 400 MHz): δ 7.42-7.31 (m, 5H), 6.49 (s, 1H), 6.42 (s, 1H), 5.93 (s, 1H), 3.73 (s, 3H), 1.48 (s, 9H). 13C-NMR (CDCl3, 400 MHz): δ HRMS (FAB, MH+) calcd. for C16H21O5 293.1389, observed 293.1399. 2-(methoxycarbonyl)-1-o-tolylallyl tert-butyl carbonate (9b) Reaction time 100 min. Purified by column chromatography O O (PE/Et2O 2:1). White solid (1.42 g, 4.64 mmol, 47%). FTIR O COOMe (CHCl3) cm-1: 3029, 2984, 1739, 1634, 1440, 1396, 1371, 1281, 1157, 1082, 1036, 962, 881, 818 cm-1. 1H-NMR (CDCl3, 400 50 MHz): δ 7.32-7.30 (m, 1H), 7.21-7.16 (m, 4H), 6.72 (s, 1H), 6.43 (s, 1H), 5.72 (s, 1H), 3.73 (s, 3H), 2.41 (s, 3H), 1.46 (s, 9H). 13C-NMR (CDCl3, 400 MHz): δ 165.6, 152.6, 139.1, 136.4, 135.5, 130.6, 128.4, 127.1, 127.0, 126.1, 82.5, 72.7, 52.0, 27.8, 19.1. HRMS (FAB, MH+) calcd. for C17H23O5 307.1545, observed 307.1551. 2-(methoxycarbonyl)-1-(3-chlorophenyl)allyl tert-butyl carbonate (9c) Reaction time 60 min. Purified by column chromatography O O (PE/Et2O 2:1). Light yellow solid. (4.40 g, 13.5 mmol, 68 %) O COOMe FTIR (CHCl3) cm-1: 3030, 2985, 1744, 1633, 1577, 1475, 1439, 1396, 1371, 1278, 1156, 1082, 1037, 964, 883, 849, 818 cm-1. 1HNMR (CDCl3, 400 MHz): δ 7.39 (s, 1H), 7.30-7.28 (m, 3H), 6.44- Cl 6.43 (m, 2H), 5.95 (s, 1H), 3.72 (s, 3H), 1.47 (s, 9H). 13 C-NMR (CDCl3, 400 MHz): δ 165.2, 152.2, 139.7, 139.2, 134.4, 129.8, 128.7, 127.7, 126.3, 126.0, 83.0, 75.0, 52.1, 27.8. HRMS (FAB, MH+) calcd. for C16H20O5Cl : 327.0999 observed; 327.0997. 2-(methoxycarbonyl)-1-(4-nitrophenyl)allyl tert-butyl carbonate (9d) Purification by column chromatography (PE/Et2O 2-1) colour O O less oil. (1.42 g, 4.21 mmol, 57 %) FTIR (CHCl3) cm-1: 3030, O 2985, 2955, 1745, 1633, 1609, 1526, 1495, 1441, 1396, 1371, COOMe 1351, 1279, 1258, 1155, 1088, 1038, 968, 890, 851 cm-1. 1HNMR (CDCl3, 400 MHz): δ 8.21 (d, J=6.8 Hz, 2H), 7.60 (d, O2N J=6.8 Hz, 2H), 6.53 (s, 1H), 6.47 (s, 1H), 6.03 (s, 1H), 3.73 (s, 3H), 1.47 (s, 9H). 13 C-NMR (CDCl3, 400 MHz): δ 165.0, 152.1, 147.8, 145.0, 138.6, 128.7, 126.9, 123.7, 83.4, 74.6, 52.2, 27.7. HRMS (FAB, MH+) calcd. for C16H20NO7 338.1240, observed 338.1237. 2-(methoxycarbonyl)-1-(naphthalen-5-yl)allyl tert-butyl carbonate (9e) Reaction time 60 min. Purified by column chromatography O O (PE/Et2O 2:1). Light yellow solid. (1.42 g, 4.1 mmol, 66 %) FTIR O (CHCl3) cm-1: 3022, 2985, 1740, 1635, 1513, 1439, 1396, 1371, COOMe 1278, 1256, 1156, 1088, 968, 886, 847 cm-1. 1H-NMR (CDCl3, 400 MHz): δ 7.40-7.14 (m, 7H), 6.49 (s, 1H), 6.39 (s, 1H), 5.91 (s, 1H), 3.69 (s, 3H), 1.45 (s, 9H). 13C-NMR (CDCl3, 400 MHz): 51 δ 165.8, 152.7, 139.2, 133.9, 133.3, 131.0, 129.3, 128.8, 128.1, 126.6, 125.9, 125.2, 123.5, 82.8, 72.5, 52.2, 27.8. HRMS (FAB, MH+) calcd. for C20H23O5 343.1545, observed 343.1542. 2-(methoxycarbonyl)-1-(3-bromophenyl)allyl tert-butyl carbonate (9f) Reaction time 60 min. Purified by column chromatography O O (PE/EtOAc 4:1). White solid. (6.3 g, 17.0 mmol, 61 %). O Enantiomerically- enriched compound obtained after kinetic COOMe resolution with 2a (0.78 g, 2.1 mmol, 47 %). FTIR (CHCl3) cm-1: 3028, 2985, 2954, 1743, 1633, 1573, 1475, 1440, 1396, 1371, Br 1278, 1255, 1194, 1156, 1085, 1037, 998, 964, 883, 847, 818 cm- . H-NMR (CDCl3, 400 MHz): δ 7.55 (t, 1H J=3.6 Hz), 7.47-7.44 (m, 1H), 7.37-7.35 (m, 1 1 1H), 7.25-7.21 (m, 1H), 6.45 (s, 2H), 5.97 (s, 1H), 3.74 (s, 3H), 1.49 (s, 9H). 13 C-NMR (CDCl3, 400 MHz): δ 165.2, 152.3, 139.9, 139.2, 131.7, 130.6, 130.1, 126.5, 126.3, 122.6, 83.1, 75.0, 52.2, 27.8. HRMS (FAB, MH+) calcd. for C16H20O5Br : 371.0494 observed; 371.0494. [α]D25 = +110.6 (c 0.99, CHCl3). Mp: 77 ˚C. HPLC Daicel Chiralcel AD, heptane/2-propanol (95:5), flow 1.0 mL/min, λ=254 nm, tr(major)=5.5 min, tr(minor)=6.0 min. tert-butyl 2-cyano-1-phenylallyl carbonate (14) Reaction time 90 min. Purification by column chromatography O (Pe/EtOAc 2.5:1). Light yellow oil. (2.2 g, 8.5 mmol, 64 %). FTIR O O (CHCl3) cm-1: 3028, 2985, 2231, 1745, 1456, 1396, 1371 1277, 1258, CN 1157, 1090, 1037, 953, 883, 848 cm-1. 1H-NMR (CDCl3, 400 MHz): δ 7.44-7.41 (m, 5H), 6.13 (s, 1H), 6.11 (d, J=0.8 Hz, 1H), 6.07 (d, J=1.2 Hz, 1H), 1.51 (s, 9H). C-NMR (CDCl3, 400 MHz): δ 152.1, 135.5, 131.7, 129.4, 129.0, 13 127.1, 123.3, 116.2, 83.6, 77.0, 27.8. HRMS (FAB, MH+) calcd. for C15H18O3N 260.1287, observed 260.1288. 2-(ethoxycarbonyl)-1-phenylallyl tert-butyl carbonate (7) Reaction time 90 min. Purified by column chromatography O O (PE/Et2O 2:1) White solid. (2.4 g, 7.8 mmol, 53%). FTIR (CHCl3) O COOEt 52 cm-1: 3019, 2977, 1742, 1522, 1423, 1046, 929, 876. 1H-NMR (CDCl3, 400 MHz): δ 7.437.30 (m, 5H), 6.50 (s, 1H), 6.43 (s, 1H), 5.91 (s, 1H), 4.21-4.15 (m, 2H), 1.48 (s, 9H), 1.25 (t, J=14.4 Hz, 3H). 13 C-NMR (CDCl3, 400 MHz): δ 164.9, 152.4, 139.9, 137.6, 128.5, 128.4, 127.7, 125.5, 82.5, 75.9, 60.9, 27.7, 14.0. HRMS (FAB, MH+) calcd. for C22H22O4N 364.1549, observed 364.1535. General procedure for the preparation of racemic 11a-f and 15: Racemic samples for HPLC analysis were prepared using MBH-carbonate (9a-f or 14, 0.2 mmol) and pronucleophile (10a-b, 0.2 mmol) in toluene (2 mL). 1,4-Diaza-bicyclo[2.2.2]octane (0.1 mmol) was added to the solution and stirring was continued for 30 min at room temperature. The reaction mixtures were filtered over silica (Et2O) and the solvent was removed by rotary evaporation. In order to obtain analytically pure samples, the reaction mixtures were subjected to flash chromatography to afford racemic 11a-f and 15. General procedure for the asymmetric allylic alkylation of MBH-carbonates (9a-f and 14): β-ICPD (20 mol %) was added to the total volume of toluene and the resulting suspension was heated and stirred until formation of a clear solution. Subsequently, the mixture was allowed to cool to the indicated temperature. MBH-carbonate (9a-f) was added to the reaction mixture and the mixture was stirred until complete dissolution (~5-10 min). Cyanoacetate (10a-b) was added in one portion and the mixture was kept at the indicated temperature for the described reaction time. The experiments were stopped by filtration of the reaction mixture over a short pad of silica gel and elution with Et2O. Solvents were removed by rotary evaporation and the crude mixture was purified by column chromatography. 1-ethyl-5-methyl-2-cyano-4-methylene-2,3-diphenylpentanedioate (11a) Ph CN COOEt Amounts used: 6.0 mmol (1.75 g) 2-(methoxycarbonyl)-1phenylallyl tert-butyl carbonate (9a COOMe - phenyl-cyano-acetate (10a), 6o mL toluene and 20 mol % β-ICPD (186.2 mg). Reaction mixture was stirred for 72 h, small scale experiment 48 h. Reaction temperature -20ºC. Purification by column chromatography (PE/Et2O 4:1). Enantiopure crystals of the major diastereomer were obtained by crystallizing the mixture of diastereoisomers from cyclohexane (2 mL). Transparent crystals (0.69 g, 1.9 mmol, 63%). FTIR (CHCl3) cm-1: 3029, 3011, 2954, 1742, 1718, 1631, 1497, 1450, 1234, 1196, 1154, 1094, 1034, 1005, 956, 858, 819 cm-1. 1H-NMR (CDCl3, 400 MHz): 7.76-7.40 (m, 2H), 7.59-7.57 (m, 2H), 7.44-7.32 (m, 6H), 6.39 (s, 1H), 6.06 (s, 1H), 5.47 (s, 1H), 4.1053 4.04 (m, 2H), 3.56 (s, 3H), 1.06 (t, J=14.2 Hz, 3H). 13 C-NMR (CDCl3, 400 MHz): δ 166.7, 166.6, 137.5, 136.6, 133.1, 129.7, 129.1, 128.6, 128.6, 128.2, 127.1, 118.0, 63.4, 59.3, 52.3, 49.9 13.7. HRMS (FAB, MH+) calcd. for C22H22O4N 364.1549, observed 364.1535. [α]D25 = 231.8 (c 1.01, CHCl3). Mp: 101˚C. HPLC Daicel Chiralcel OD-H, heptane/2-propanol (95:5), flow 1.0 mL/min, λ=254 nm, tr(major)=17.4 min, tr(minor)=20.5 min. 1-ethyl 5-methyl 2-cyano-4-methylene-2-phenyl-3-o-tolylpentanedioate (11b) Ph CN COOEt Amounts used: 0.6 mmol (183.8 mg) 2-(methoxycarbonyl)-1-otolylallyl tert-butyl carbonate, 0.3 mmol (52.1 ul) ethyl-phenyl- COOMe cyano-acetate (10a), 6 mL toluene and 20 mol % β-ICPD (18.6 mg). Reaction mixture was stirred for 96 h. Reaction temperature 0 ºC. Purification by column chromatography (PE/ Et2O 4:1). Light yellow solid (109 mg, 0.29 mmol, 95%). FTIR (CHCl3) cm-1: 3028, 3010, 2953, 2250, 1742, 1721, 1629, 1495, 1450, 1440, 1392, 1369, 1333, 1237, 1195, 1140, 1095, 1033, 995, 953, 909 cm-1. 1H-NMR (CDCl3, 400 MHz, major diastereoisomer): δ 8.00 (d, J=7.6 Hz, 1H), 7.78-7.75 (m, 2H), 7.44-7.41 (m, 3H), 7.23-7.19 (m, 3H), 6.42 (s, 1H), 5.91 (s, 1H), 5.42 (s, 1H), 4.15-4.03 (m, 2H), 3.36 (s, 3H), 2.40 (s, 3H), 1.06 (t, J =14.4 Hz, 3H). 13 C-NMR (CDCl3, 400 MHz): δ 166.7, 166.5, 138.2, 137.5, 137.0, 132.6, 131.2, 130.8, 129.2, 128.8, 127.5, 127.4, 126.4, 126.1, 118.3, 63.2, 59.2, 51.9, 47.6, 19.9, 13.6. HRMS (FAB, MH+) calcd. for C23H24O4N 378.1705, observed 378.1711. HPLC Daicel Chiralcel OD-H, heptane/2-propanol (98:2), flow 0.7 mL/min, λ=254 nm, tr(minor)=20.9 min, tr(major)=42.6 min. 1-ethyl 5-methyl 3-(3-chlorophenyl)-2-cyano-2-methyl-4-methylenepentanedioate (11c) CN Amounts used: 0.6 mmol (193.1 mg) 2-(methoxycarbonyl)-1-(3COOEt COOMe chlorophenyl) allyl- tert-butyl carbonate, 0.3 mmol (52.1 ul) ethyl 2cyanopropanoate (10b), 6 mL toluene and 20 mol % β-ICPD (18.6 mg). Reaction mixture was stirred for 72 h. Reaction temperature -20 ºC. Purification by column chromatography (Toluene/EtOAc 95:5). Cl Yellow oil. (97 mg, 0.29 mmol, 95%). FTIR (CHCl3) cm-1: 3028, 2989, 2955, 2247, 1741, 1631, 1596, 1574, 1477, 1442, 1381, 1284, 1245, 1198, 1170, 1125, 1084, 1015, 965, 909, 856 cm-1. 1H-NMR,(CDCl3, 400 MHz, major diastereoisomer): δ 7.417.26 (m, 4H), 6.71 (s, 1H), 6.43 (s, 1H), 4.66 (s, 1H), 4.13-4.07 (m, 2H), 3.78 (s, 3H), 1.08 (t, J=7.2 Hz, 3H). 13 C-NMR (CDCl3, 400 MHz) (major diastereoisomer) δ 168.4, 166.8, 139.6, 137.3, 134.5, 129.9, 129.2, 129.1, 128.2, 126.7, 119.2, 63.1, 52.7, 49.1, 48.9, 23.4, 13.7. HRMS (FAB MH+,) calcd. for C17H19O4N : 336.1003 observed; 336.1008. HPLC Daicel 54 Chiralcel OD-H, heptane/2-propanol (99.5-0.5), flow 0.9 mL/min, λ=254 nm, tr(major)=31.1 min, tr(minor)=20.3 min. 1-ethyl 5-methyl 2-cyano-4-methylene-3-(4-nitrophenyl)-2-phenylpentanedioate (11d) Ph CN Amounts used: 0.6 mmol (202.4 mg) 2-(methoxycarbonyl)-1COOEt (4-nitrophenyl) allyl tert-butyl carbonate 0.3 mmol (52.1 ul) COOMe ethyl-phenyl-cyano-acetate(10a) , 6 mL toluene and 20 mol % β-ICPD (18.6 mg). Reaction mixture was stirred for 48 h. O2N Reaction temperature -20 ºC. Purification by column chromatography (PE/Et2O 4:1). Light yellow solid. (116 mg, 0.29 mmol, 95 %) FTIR (CHCl3) cm-1: 3029, 2955, 1743, 1632, 1606, 1525, 1496, 1450, 1350, 1291, 1276, 1236, 1197, 1154, 1113, 1035, 1003, 961, 909, 858 cm-1. 1H-NMR (CDCl3, 400 MHz, major diastereoisomer): δ 8.24 (dd, J=7.2, 2.0 Hz, 2H), 7.78 (d, J=8.8 Hz, 2H), 7.71 (dd, J=8.0, 1.2 Hz, 2H), 7.47-7.41 (m, 3H), 6.46 (s, 1H), 6.08 (s, 1H), 5.60 (s, 1H), 4.14-4.08 (m, 2H), 3.58 (s, 3H), 1.10 (t, J=14.4 Hz, 3H). 1H-NMR (CDCl3, 400 MHz, minor diastereoisomer): δ 7.96 (d, J=8.8 Hz, 2H), 7.46-7.34 (m, 5H), 7.10 (d, J=8.8 Hz, 2H), 6.70 (s, 1H), 6.62 (s, 1H), 5.06 (s, 1H), 4.36-4.27 (m, 2H), 3.69 (s, 3H), 1.22 (t, J=14.0 Hz, 3H). 13 C-NMR (CDCl3, 400 MHz, mixture of diastereoisomers): δ 166.3, 166,3, 166.2, 165.9, 147.8, 147.5, 144.2, 142.3, 139.0, 136.5, 132.4, 132.3, 130.8, 130.6, 129.7, 129.6, 129.4, 129.3, 129.3, 126.9, 126.6, 126.1, 123.7, 123.0, 117.5, 117.1, 63.9, 63.9, 58.5, 58.1, 52.8, 52.6, 52.4, 49.4, 13.9, 13.7. HRMS (FAB, MH+) calcd. for C22H21O6N2 409.1400, observed 409.1409. HPLC Daicel Chiralcel AD, heptane/2-propanol (95:5), flow 1.0 mL/min, λ=254 nm, tr(major)=46.7 min, tr(minor)=14.8 min. 1-ethyl 5-methyl 2-cyano-4-methylene-3-(naphthalen-5-yl)-2-phenylpentanedioate (11e) Ph CN Amounts used: 0.6 mmol (205.4 mg)2-(methoxycarbonyl)-1COOEt COOMe (naphthalen-5-yl) allyl tert-butyl carbonate, 0.3 mmol (52.1 ul) ethyl-phenyl-cyano-acetate (10a), 6 mL toluene and 20 mol % βICPD (18.6 mg). Reaction mixture was stirred for 24 h. Reaction temperature 20 ºC. Purification by column chromatography (PE/Et2O 5:1 and Toluene/EtOAc 99.5:0.5). Light yellow solid. Compound obtained as a single diastereoisomer. (82.7 mg, 0.20 mmol, 66%) FTIR (CHCl3) cm-1: 3029, 3012, 1743, 1718, 1631, 1512, 1496, 1450, 1440, 55 1368, 1334, 1236, 1196, 1140, 1095, 1035, 993, 967, 857 cm-1. 1H-NMR (CDCl3, 400 MHz, major diastereoisomer) δ 8.26 (d, J=8.4 Hz, 1H), 8.18 (d, J=7.2 Hz, 1H), 7.89-7.83 (m, 4H), 7.57-7.45 (m, 6H), 6.39 (s, 1H), 6.06 (s, 1H), 5.89 (s, 1H), 4.08-3.98 (m, 2H), 3.40 (s, 3H), 0.97 (t, J=14.4, 3H). 13C-NMR (CDCl3, 400 MHz) δ 166.7, 166.4, 138.4, 135.4, 134.2, 132.4, 131.4, 131.1, 129.3, 128.9, 128.5, 127.5, 126.7, 126.7, 126.0, 124.9, 124.2, 123.7, 118.3, 63.3, 59.3, 52.0, 47.5, 13.5. HRMS (FAB, MH+) calcd. for C26H24O4N : 414.1705 observed; 414.1696. HPLC Daicel Chiralcel OD-H, heptane/2-propanol (95:5), flow 1.0 mL/min, λ=254 nm, tr(major)=37.9 min, tr(minor)=10.5 min. 1-ethyl 5-methyl 3-(3-bromophenyl)-2-cyano-4-methylene-2-phenylpentanedioate (11f) Ph CN Amounts used: 9.0 mmol (3.34 g) 2-(methoxycarbonyl)-1-(3COOEt bromophenyl)allyl tert-butyl carbonate, 4.5 mmol (780 ul) ethyl- COOMe phenyl-cyano-acetate (10a), 90 mL toluene and 20 mol% β-ICPD (279.3 mg). Reaction mixture was stirred for 72 h. Reaction temperature Br -20ºC. Purification by column chromatography (PE/Et2O 4:1). Enantiopure crystals where obtained by crystallizing the mixture of diasterioisomers from cyclohexane (2.5 mL). white/transparent crystals. (1.16 g, 2.65 mmol, 59 %) FTIR (CHCl3) cm-1: 3029, 3012, 2955, 1743, 1632, 1593, 1570, 1497, 1475, 1450, 1273, 1235, 1196, 1154, 1095, 1077, 1035, 999, 960, 857 cm-1. 1H-NMR (CDCl3, 400 MHz) δ 7.73 (m, 3H), 7.55-7.53 (m, 1H), 7.49-7.48 (m, 1H), 7.45-7.38 (m, 3H), 7.28-7.24 (m, 1H), 6.41 (s, 1H), 6.05 (s, 1H), 5.43 (s, 1H), 4.18-4.06 (m, 2H), 3.59 (s, 3H), 1.12 (t, J=14.4 Hz, 3H). 13C-NMR (CDCl3, 400 MHz): δ 166.6, 166.4, 139.0, 136.9, 132.9, 132.8, 131.4, 130.1, 129.2, 129.2, 129.2, 128.1, 127.0, 122.6, 117.7, 63.7, 59.0, 52.3, 49.4, 13.7. HRMS (FAB, MH+) calcd. for C22H21O4NBr 442.0654, observed 442.0642. [α]D25 = -164.0 (c 1.0, CHCl3). Mp: 108 ˚C. HPLC Daicel Chiralcel AD, heptane/2-propanol (95:5), flow 1.0 mL/min, λ=254 nm, tr(major)=9.7 min, tr(minor)=12.5 min. ethyl 2,4-dicyano-2,3-diphenylpent-4-enoate (15) Ph CN Amounts used: 0.6 mmol (155.6 mg) tert-butyl 2-cyano-1-phenylallyl COOEt CN carbonate, 0.3 mmol (52.1 ul) ethyl-phenyl-cyano-acetate, 6 mL toluene and 20 mol % β-ICPD (18.6 mg). Reaction mixture was stirred for 48 h. Reaction temperature -20 ºC. Purification by column chromatography (PE/Et2O 4:1). Mixture of diastereo-isomers. Yellow oil. (96 mg, 0.29 mmol, 95%) FTIR (CHCl3) cm-1: 3067, 3026, 3012, 2986, 2929, 2250, 1744, 1619, 1497, 56 1451, 1394, 1369, 1297, 1236, 1194, 1096, 1034, 1004, 946, 909, 859 cm-1. 1H-NMR (CDCl3, 400 MHz) (mixture of diastereoisomers) δ 7.82 (dd, J=8.4, 1.6 Hz, 2H, major), 7.67 (dd, J=8.0, 1.6 Hz, 2H, major), 7.53-7.04 (m, 16H), 6.26 (s, 1H, minor), 6.25 (s, 1H, minor), 5.93 (s, 1H, major), 5.75 (s, 1 H, major), 4.72 (s, 1H, major), 4.58 (s, 1H, minor), 4.41-4.26 (m, 2H, minor), 4.12-4.02 (m, 2H, major), 1.30 (t, J=7.2, 3H, minor), 1.05 (m, J=6.8 Hz, 3H, major). C-NMR (CDCl3, 400 MHz, mixture of diastereoisomers): δ 166.5, 165.9, 135.6, 13 133.1, 132.9, 132.1, 132.1, 130.9, 129.7, 129.6, 129.5, 129.3, 129.2, 129.0, 129.0, 128.6, 128.5, 126.9, 126.6, 122.9, 120.6, 117.6, 117.3, 117.0, 116.6, 64.1, 63.7, 58.4, 57.7, 55.4, 54.9, 13.8, 13.5. HRMS (FAB, MH+) calcd. for C21H19O2N2 331.1447, observed 331.1447. HPLC Daicel Chiralcel AD, heptane/2-propanol (9:1), flow 1.0 mL/min, λ=254 nm, tr(major)=21.6 min, tr(minor)=17.4 min. 3-ethyl 1,1-dimethyl 2-phenylbut-3-ene-1,1,3-tricarboxylate (8) O Prepared according procedure as described by Lu et al.,48 similar O MeO OMe COOEt results where obtained. FTIR (CHCl3) cm-1: 3030, 3006, 2955, 1735, 1630, 1495, 1437, 1263, 1196, 1153, 1034, 954, 816. 1H-NMR (CDCl3, 400 MHz): δ 7.28-7.20 (m, 5H), 6.33 (s, 1H), 5.75 (d, J=0.8 Hz, 1H), 4.72 (d, J=12.4 Hz, 1H), 4.21 (d, J=12.4 Hz, 1H), 4.16- 4.11(m, 2H), 3.75 (s, 3H), 3.49 (s, 3H), 1.23 (t, J=6.8 Hz, 3H). 13 C-NMR (CDCl3, 400 MHz): δ 168.1, 167.8, 166.3, 141.0, 138.5, 128.5, 128.4, 127.4, 124.6, 56.3, 52.9, 52.6, 52.1, 46.5. HRMS (FAB, MH+) calcd. for C17H21O6 321.1338, observed 321.1344. Ethyl 1-(2-(methoxycarbonyl)-1-phenylallyl)-2-oxocyclohexanecarboxylate (13) O Amounts used: 0.6 mmol ()2-(methoxycarbonyl)-1-phenylallyl tert- COOEt butyl carbonate, 0.3 mmol (52.1 ul) ethyl-phenyl-cyano-acetate, 6 COOMe mL toluene and 20 mol % β-IC (18.6 mg). Reaction mixture was stirred for 24 h. Reaction temperature 20 ºC. Purification by column chromatography (PE/Et2O 4-1 and Toluene/EtOAc 98-2). 1H NMR (CDCl3, 400 MHz)(mixture of diastereoismers)(major diastereoisomer) δ 7.48 (d, 2H, J=6.8 Hz), 7.36-7.28 (m, 3H), 6.38 (s, 2H), 6.12 (s, 1H), 4.23 (q, 2H, J=21.6, 14.4, 7.2 Hz), 3.73 (s, 3H), 2.31 (m, 2H), 2.25 (m, 2H), 1.63 (m, 2H), 1.50 (m, 2H), 1.32 (t, 3H, J=14.4, 7.2 Hz). HRMS (FAB, MH+) calcd. for C20H25O5 : 345.1702 observed; 345.1700. HPLC Daicel Chiralcel AD, heptane/2-propanol (98-2), Flow 1.0 ml/min, UV 254 nm. 57 1 H-NMR spectrum of compound 11a 58 13 C-NMR spectrum of compound 11a 59 Chiral HPLC trace of racemic 11a Chiral HPLC trace of compound 11a after chromatography 60 Chiral HPLC trace of compound 11a after recrystallization 61 1 H-NMR spectrum of compound 11f 62 13 C-NMR spectrum of compound 11f 63 Chiral HPLC trace of racemic 11f Chiral HPLC trace of compound 11f after chromatography 64 Chiral HPLC trace of compound 11f after recrystalization 65 Computational Methods Computations were carried out using the Spartan ’04 program.[57] The initial structure of intermediate B was computed using ab initio methods (HF/3-21G*) and the output was used as starting geometry for full optimization at the B3LYP/6-31G** level of theory. Intermediate B Charge = 1 Multiplicity = 1 Stoichiometry = C30H33N2O4 B3LYP/6-31G** Energy = -1573.2839293 Hartree ATOM 1 H 2 C 3 C 4 N 5 C 6 C 7 C 8 C 9 H 10 H 11 C 12 H 13 C 14 H 15 C 16 O 17 H 18 C 19 C 20 H 21 C 22 H 23 H 24 N 25 C 26 H 27 H 28 C 29 H 30 H 31 C 32 H Coordinates (Angstroms) X Y Z 3.587610 -2.798929 -0.209392 3.977707 -1.789207 -0.203604 3.194582 -0.739082 0.232108 5.848494 -0.346249 -0.688123 3.768810 0.568007 0.251311 5.299926 -1.542895 -0.643783 5.104745 0.705355 -0.251565 3.089753 1.719181 0.713392 5.914178 -2.375264 -0.981700 6.697684 2.080251 -0.684198 3.677277 2.969011 0.630869 2.105373 1.640033 1.153452 4.993238 3.110899 0.116984 5.428024 4.103720 0.074261 5.686226 2.003195 -0.300262 3.041796 4.102797 1.021429 2.132970 3.878144 1.287395 1.753261 -0.977105 0.669100 0.721448 -0.413689 -0.351565 0.889665 0.638850 -0.572101 0.781798 -1.354690 -1.561185 0.373685 -0.909079 -2.472027 1.829496 -1.583296 -1.757142 -0.738417 -0.530099 0.256744 -1.743453 -0.823710 -0.859653 -1.611594 -0.053469 -1.618105 -2.736404 -0.712829 -0.425626 -1.497031 -2.248957 -1.402279 -1.699280 -2.258862 -2.476851 -2.181829 -2.967630 -0.943016 -0.025449 -2.598661 -1.128681 0.284346 -3.494342 -1.669086 66 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 58 59 60 61 62 63 64 65 66 67 68 69 C C H H H O C H H C C H C O C H H H O C C C C C C H H H H H C H H C H H H 0.191053 -0.737503 -0.350845 -1.769122 1.602048 1.528584 -1.190797 -0.363427 -2.017572 -1.593975 -2.830233 -2.891482 -0.564031 -0.779038 0.092377 1.137708 -0.064072 -0.203498 0.363232 -4.105859 -6.604006 -4.957461 -4.552390 -5.791863 -6.184903 -4.646006 -3.965543 -6.133641 -6.821479 -7.568192 -0.032335 -1.022589 -0.070008 1.035693 0.794391 2.019822 1.098324 -2.780020 -1.777860 -1.533590 -2.118543 -0.514754 -2.391962 0.669389 0.898267 0.276389 1.919946 2.213097 3.153882 3.006070 3.939053 5.099176 4.796798 5.666953 5.686674 3.033751 1.488454 0.194995 1.566730 0.783010 0.147777 0.909095 2.141850 0.787051 -0.369839 0.969959 -0.303159 -4.202553 -4.536296 -4.161566 -5.215067 -6.199928 -4.925667 -5.318959 0.417367 1.124726 2.114655 1.225234 1.656507 0.768683 1.109496 1.782354 1.696479 0.359021 -0.121539 -0.663687 0.330203 -0.594240 -0.578344 -0.650626 0.341290 -1.445054 1.131368 -0.054508 -0.060273 -1.173759 1.080740 1.076080 -1.184372 -2.041390 1.993706 1.966871 -2.061088 -0.059288 0.936512 0.601467 2.031984 0.510480 0.918493 0.884521 -0.577167 67 X-ray crystal structure determinations X-ray intensities were measured on a Nonius KappaCCD diffractometer with rotating anode and graphite monochromator ( = 0.71073 Å) at a temperature of 150(2) K up to a resolution of (sin /)max = 0.65 Å-1. The structures of 9f and 11f were solved with Direct Methods (SIR2004[58] for 9f and SHELXS-97[59] for 11f). The initial coordinates for 3a were taken from the isostructural 3f. Full-matrix least-squares refinement was performed with SHELXL97[60]. Non-hydrogen atoms were refined freely with anisotropic displacement parameters. Hydrogen atoms were located in the difference Fourier map (9f and 11f) or introduced in geometrically optimized positions. All hydrogen atoms were refined with a riding model. In 11a the ethyl group was refined with a disorder model. Structure drawings, geometry calculations and checking for higher symmetry were performed with the PLATON[61] program. Further crystallographic details are given in Table S1. 68 Table S1: Crystallographic details for 9f, 11f, and 11a 9f 11f 11a Formula C16H19BrO5 C22H20BrNO4 C22H21NO4 Fw 371.22 442.30 363.40 Crystal colour colourless colourless colourless Crystal size [mm3] 0.51 x 0.30 x 0.24 0.27 x 0.12 x 0.03 0.30 x 0.30 x 0.21 Crystal system monoclinic monoclinic monoclinic Space group P21 (no. 4) P21 (no. 4) P21 (no. 4) a [Å] 8.9808(1) 7.6675(1) 7.4898(1) b [Å] 10.6947(1) 15.6520(3) 15.5364(2) c [Å] 9.1042(1) 8.9590(2) 8.6839(1) [°] 110.7788(4) 109.8294(7) 107.0520(5) V [Å3] 817.555(15) 1011.43(3) 966.08(2) Z 2 2 2 dcalc [g/cm3] 1.508 1.452 1.249 [mm-1] 2.534 2.059 0.086 Abs. correction method multi-scan [62] multi-scan[62] multi-scan[62] Abs. correction range 0.40-0.54 0.72-0.94 0.94-0.98 reflections (measured/unique) 24600/3744 23036/4628 24677/2308 Rint 0.0421 0.0465 0.0343 Parameters / restraints 203 / 1 255 / 1 257 / 3 R1/wR2 [I>2(I)] 0.0169/0.0412 0.0347/0.0640 0.0311/0.0782 R1/wR2 [all refl.] 0.0173/0.0414 0.0486/0.0683 0.0342/0.0805 S 1.075 1.031 1.031 Flack x parameter[63] 0.008(4) -0.014(6) [*] Friedel pair coverage 99.9% 98.9% [*] min/max [e/Å3] -0.30/0.18 -0.34/0.25 -0.16/0.19 [*] The absolute structure of 11a could not be determined reliably. Therefore Friedel pairs were merged prior to the final refinement. 69 References 1 a) Dalko, P. I; Moisan, L. Angew. Chem. Int. Ed. 2001, 40, 3726, b) Dalko, P. I; Moisan, L. Angew. Chem. Int. Ed. 2004, 43, 5138, c) Seayad, J; List, B. Org. Biomol. Chem. 2005, 3, 719, d) Houk, K. N; List, B. Acc. Chem. Res. 2004, 37, 487. 2 Bredig, G; Fiske, P. S. Biochem. Z. 1912, 46, 7. 3 List, B; Lerner, R. A; Barbas ΙΙΙ, C. F. J. Am. Chem. Soc. 2000, 122, 2395. a) Pracejus, H. Justus Liebigs Ann. Chem. 1960, 634, 9, b) Pracejus, H. Justus Liebigs Ann. Chem. 1960, 634, 4 23. 5 Hajos, Z. G.; Parrish, D. R. J. Org. Chem. 1974, 39, 1615 6 Eder, U; Sauer, G; Wiechert, R. Angew. Chem. Int. Ed. Engl. 1971, 10, 496. 7 a) Hermann, K; Wynberg, H. Helv. Chim. Act. 1977, 60, 2208, b) Hermann, K; Wynberg, H. J. Org. Chem. 1979, 44, 1979, c) Hiemstra, H; Wynberg, H. J. Am. Chem. Soc. 1981, 103, 417, d) Smaardijk, A. A; Wynberg, H. J. Org. Chem. 1987, 52, 135. 8 a) Schreiner, P. R. Chem. Soc. Rev. 2003, 32, 289, b) Pihko, P. M. Angew. Chem. Int. Ed. 2004, 43, 2062 c) Akiyama, T; Itoh, J; Fuchibe, K. Adv. Synth. Catal. 2006, 348, 999. 9 a) Sigman, M. S; Vachal, P; Jacobsen, E. N. Angew. Chem. Int. Ed. 2000, 39, 1279, b) Hoashi, Y; Okino, T; Takemoto, Y. Angew. Chem. Int. Ed. 2005, 44, 4032, c) Herrera, R. P; Sgarzani, V; Bernardi, L; Ricci, A. Angew. Chem. Int. Ed. 2005, 44, 6576, d) Okino, T; Hoashi, Y; Takemoto, Y. J. Am. Chem. Soc .2003, 125, 12672 e) Marcelli, T; Maarseveen, J. H; Hiemstra, H. Angew. Chem. Int. Ed. 2006, 45, 929. 10 a) Tian, S. K; Chen, Y; Hang, J; Tang, L; McDaid, P; Deng, L. Acc. Chem. Res. 2004, 37, 621, b) Kacprzak, K; Gawronski, J. Synthesis, 2001, 961, c) France, S; Guerin, D. J; Miller, S. J; Lectka, T. Chem. Rev. 2003, 103, 2985. 11 Hoffmann, H. M. R; Frackenpohl, J. Eur. J. Org. Chem. 2004, 4293, 12 Pelletier, J; Caventon, J. B. Ann. Chim. Phys. 1820, 14, 69. 13 Yoon, T. P; Jacobsen, E. N. Science, 2003, 299, 1691. 14 Kolb, H. C; VanNieuwenhze, M. S; Sharpless, B. K. Chem. Rev. 1994, 94, 2483. 15 Marcelli, T; Maarseveen, J. H; Hiemstra, H. Angew. Chem. Int. Ed. 2006, Accepted. 16 McCooey, S. H; Connon, S. J. Angew. Chem. Int. Ed. 2005, 44, 6367. 17 Iwabuchi, Y; Nakatani, M; Yokoyama, N; Hatakeyama, S. J. Am. Chem. Soc. 1999, 121, 10219. 18 Kawahara, S; Nakano, A; Esumi, T; Iwabuchi, Y; Hatakeyama, S. Org. Lett. 2003, 5, 3103. 19 Nakano, A; Ushiyama, M; Iwabuchi, Y; Hatakeyama, S. Adv. Synth. Catal. 2005, 347, 1790. 20 A representative selection being: a) Li, H; Song, J; Liu, X; Deng, L. J. Am. Chem. Soc. 2005, 127, 8948, b) Wang, Y; Liu, X; Deng, L. J. Am. Chem. Soc. 2006, 126, 3928. 21 Vakulya, B; Varga, S; Csampai, A; Soos, T. Org. Lett. 2005, 7, 1967. 22 Marcelli, T; van der Haas, R. N. S; van Maarseveen, J. H; Hiemstra, H. Synlett, 2005, 2817. 23 Morita, K; Suzuki, Z; Hirose, H. Bull. Chem. Soc. Jpn. 1968, 41, 2815. 24 Baylis, A. B; Hillman, M. E. D. German Patent 2155113, Chem. Abstr., 1972, 77, 3417q. 25 a) Basavaiah, D; Rao, A, J; Satyanarayana. Chem. Rev. 2003, 103, 811, b) Basavaiah, D; Rao, P. D; Hyma, R. S. Tetrahedron, 1996, 52, 8001. 70 26 Langer, P. Angew. Chem. Int. Ed. 2000, 39, 3049. 27 You, J; Xu, J; Verkade, J. G. Angew. Chem. Int. Ed. 2003, 42, 5054. 28 a) McDougal, N. T; Schauss, S. E. J. Am. Chem. Soc. 2003, 125, 12094, b) Wang, J; Li, H; Yu, X; Zu, L; Wang, W. Org. Lett. 2005, 7, 4293. 29 a) Sohtome, Y; Tanatani, A; Hashimoto, Y; Hagasawa, K. Tetrahedron Lett. 2004, 45, 5589, b) Raheem, I. T; Jacobsen, E. N. Adv. Synth. Catal. 2005, 347, 1701, c) Aroyan, C. E; Vasbinder, M .M; Miller, S. J. Org. Lett. 2005, 7, 3849, d) Berkessel, A; Roland, K; Neudörfl, J. Org. Lett. 2006, 8, 2699. 30 Drewes, S. E; Njamela, O. L; Emslie, N, D; Ramesar, N; Field, J. S. Synth. Commun. 1993, 23, 2807. 31 Santos, L. S; Pavam, C. H; Almeida, W. P; Coelho, F; Eberlin, M. N. Angew. Chem. Int. Ed. 2004, 43, 4330. 32 Aggarwal, V. K; Fulford, S. Y; Lloyd-Jones, G. C. Angew. Chem. Int. Ed. 2005, 44, 1706. 33 K. Y. Lee, S. Gowrisankar, J. N. Kim, Bull. Korean Chem. Soc. 2005, 26, 1481-1490. 34 Iwabuchi, Y; Furukawa, M; Esumi, T; Hatakeyama, S. Chem. Comm. 2001, 2030. 35 McCauley, J, A; Nagasawa, K; Lander, P. A; Mischke, S. G; Semones, M. A; Kishi, Y. J. Am. Chem. Soc. 1998, 120, 7647. 36 Reddy, L. J; Fournier, J. F; Reddy, B.V. S; Corey; E. J. Org. Lett. 2005, 7, 2699. 37 Trost, B. M; Tsui, H. C; Toste, D. F. J. Am. Chem. Soc. 2000, 122, 3534. 38 a) Trost, B. M; Crawley, M. L. Chem Rev. 2003, 103, 2921, b) Trost, B. M. J. Org. Chem. 2004, 69, 5813, c) Trost, B. M. Chem. Pharm. Bull. 2002, 50, 1. 39 Trost, B. M; Patterson, D. E; Hembre, E. J. Chem. Eur. J. 2001, 7, 3768. 40 Trost, B. M; Machacek, M. R; Tsui, H. C. J. Am. Chem. Soc. 2005, 127, 7014. 41 a) Trost, B. M; Thiel, O. R; Tsui, H. C. J. Am. Chem. Soc. 2002, 124, 11616, b) Trost, B. M; Thiel, O. R; Tsui, H. C. J. Am. Chem. Soc. 2003, 125, 13155. 42 Kim, J. N; Lee, H. J; Gong, J. H. Tetrahedron Lett. 2002, 43, 9141. 43 a) Gong, J. H; Kim, H. R; Ryu, E. K; Kim, J, N. Bull. Korean. Chem. Soc. 2002, 23, 789, b) Kim, J. N; Lee, H. J; Lee, K, Y; Gong, J. H. Syn. Lett. 2002, 173. 44 Ciclosi, M; Fava, C; Galeazzi, R; Orena, M; Sepulveda-Arques, J. Tetrahedron Lett. 2002, 43, 2199. 45 Basavaiah, D; Kumaragurubaran, N; Sharada, D. S; Reddy, R. M. Tetrahedron. 2001, 57, 8167. 46 Cho, C. W; Krische, M. J. Angew. Chem. Int. Ed. 2004, 43, 6689. 47 Cho, C. W; Kong, J. R; Krische, M. J. Org. Lett. 2004, 6, 1337. 48 Du, Y; Han, X, Lu, X. Tetrehedron Lett. 2004, 45, 4967. 49 a) Christoffers, J; Baro, A. Adv. Synth. Catal. 2005, 347, 1473, b) Christoffers, J; Baro, A. Angew. Chem. Int. Ed. 2003, 42, 1688, c) Christoffers, J; Mann, A. Angew. Chem. Int. Ed. 2001, 40, 4591, d) Peterson, E. A; Overmann, L .E. Proc. Natl. Acad. Sci. USA, 2004, 101, 11943, e) Trost, B. M; Jiang, C. Synthesis. 2006, 3, 369. 50 a) Knudsen, K. R; Jørgenson, K. A. Org. Biomol. Chem. 2005, 3, 1362, b) Li, H; Song, J; Liu, X; Deng, L. J. Am. Chem. Soc. 2005, 127, 8948, c), Chowdari, N. S; Barbas ΙΙΙ, C. F. Org. Lett. 2005, 7, 867, d) Li, H; Wang, Y; Tang, L; Deng, L. J. Am. Chem. Soc. 2004, 126, 9906, e) Liu, T. Y; Long, J; Li, B. J; Jiang, L; Li, R; Wu, Y; Ding, L. S; Chen, Y. C. Org. Biomol. Chem. 2006, 4, 2097, f) Saaby, S; Bella, M; Jørgenson, K. A. J. Am. Chem. Soc. 2004, 126, 8120. 51 a) Wu, F; Hong, R; Khan, J; Liu, X; Deng, L. Angew. Chem. Int. Ed. 2006, 45, 4301-4305, b) Lu, F; Li, H; Hong, R; Deng, L.Angew. Chem. Int. Ed. 2006, 45, 947-950, c) Li, H; Wang, Y; Tang, L; Wu, F; Liu, X; Guo, 71 C; Foxman, B. M; Deng, L. Angew. Chem. Int. Ed. 2005, 44, 105-108 d) Austin, J. F; Kim, S.-G; Sinz, C. J; Xiao, W.-J; MacMillan, D. W. C. Proc. Natl. Acad. Sci. USA 2004, 101, 5482; e) Taylor, M. S; Jacobsen, E. N. J. Am. Chem. Soc. 2003, 125, 11204. 52 Liu, X; Li, H; Deng, L. Org. Lett. 2005, 7, 167. 53 Song, J; Wang, Y; Deng, L. J. Am. Chem. Soc. 2006, 128, 6048. 54 55 Barry M. Trost, Neil G. Andersen, J. Am. Chem. Soc. 2002, 124, 14320-14321. Y. Iwabuchi, M. Nakatani, N. Yokoyama, S. Hatakeyama, J. Am. Chem. Soc. 1999, 121, 10219-10220. 56 V. K. Aggarwal, I. Emme, S. Y. Fulford, J. Org. Chem. 2003, 68, 692-700. 57 Spartan ’04 build 123 (31 Jan 2005); B.J. Deppmeier, A.J. Driessen, T.S. Hehre, W.J. Hehre, J.A. Johnson, P.E. Klunzinger, J.M. Leonard, I.N. Pham, W.J. Pietro, Jianguo Yu, J. Kong, C.A. White, A.I. Krylov, C.D. Sherrill, R.D. Adamson, T.R. Furlani, M.S. Lee, A.M. Lee, S.R. Gwaltney, T.R. Adams, C. Ochsenfeld, A.T.B. Gilbert, G.S. Kedziora, V.A. Rassolov, D. R. Maurice, N. Nair, Y. Shao, N.A. Besley, P.E. Maslen, J.P. Dombroski, H. Dachsel, W.M. Zhang, P.P. Korambath, J. Baker, E.F. C. Byrd, T. Van Voorhis, M. Oumi, S. Hirata, C.P. Hsu, N. Ishikawa, J. Florian, A. Warshel, B.G. Johnson, P.M.W. Gill, M. HeadGordon, J.A. Pople; Wavefunction Inc., Irvine CA (USA). 58 M.C. Burla, R. Caliandro, M. Camalli, B. Carrozzini, G.L. Cascarano, L. De Caro, C. Giacovazzo, G. Polidori, R. Spagna (2005). J. Appl. Cryst. 38, 381-188. 59 G. M. Sheldrick (1997). SHELXS-97. Program for crystal structure solution. University of Göttingen, Germany. 60 G. M. Sheldrick (1997). SHELXL-97. Program for crystal structure refinement. University of Göttingen, Germany 61 62 A. L. Spek, J. Appl. Cryst., 2003, 36, 7-13. G. M. Sheldrick (1999). SADABS: Area-Detector Absorption Correction, v2.10, Universität Göttingen, Germany. 63 H. D. Flack (1983). Acta Cryst. A39, 876-881. 72