Mepahist - mepaco
advertisement

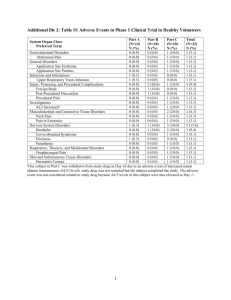
Mepahist COMPANY NAME ARAB COMPANY FOR PHARMACEUTICALS AND MEDICINAL PLANTS (MEPACO – MEDIFOOD)- Egypt TRADE NAME Mepahist GENERIC NAME Betahistine dihydrochloride PHARMACEUTICAL FORM Tablets COMPOSITION Each tablet contains Active ingredients: Betahistine dihydrochloride 16 mg Inactive ingredients: PVP K 90, Aerosil 200, Acdisol, Lactose monohydrate, Avicel pH 101 and Magnesium stearate. PHARMACOLOGY AND TOXICOLOGY Mepahist exerts a relaxant action on the papillary sphincters of the microcirculation of the inner ear and thereby increases the blood supply to the stria vascularis of the labyrinth. Extensive investigations into acute, subacute and chronic effects of betahistine in animals and widespread application of betahistine in humans have demonstrated the low toxicity and safety of this drug. CLINICAL EXPERIENCE The effect of betahistine has been widely investigated in controlled trials. Betahisitne has been shown to reduce both the frequency and the severity of the vertigo attacks, to have a favorable influence on tinnitus and to stop or even partially reserve the progressive loss of hearing, especially if treatment is started early. Although progress can usually be seen within a few days, the onset of improvement is sometimes very gradual and clearly noticeable only after several weeks of treatment. In both cases continued treatment is recommended as optimal results are usually obtained after a few months. Long term tolerance of betahistine is usually excellent. INDICATIONS Vertigo, tinnitus and hearing loss associated with Ménière's syndrome. DOSAGE AND ADMINISTRATION Adults (including the elderly): initially 16 mg three times daily taken preferably with meals. Maintenance doses are generally in the range 24-48 mg daily. Children: no dosage recommendations are made for children. CONTRAINDICATIONS Phaeochromocytoma. Hypersensitivity to the active substance or to any of the excipients. WARNINGS AND PRECAUTIONS Caution is advised in the treatment of patients with a history of peptic ulcer. Clinical intolerance to Mepahist in bronchial asthma patients has been shown in a relatively few patients. These patients need to be carefully monitored during the therapy. SIDE-EFFECTS The following undesirable effects have been experienced with the below indicated frequencies in betahistine-treated patients in placebocontrolled clinical trials [very common ( 1/10); common ( 1/100 to <1/10); uncommon ( 1/1,000 to <1/100); rare ( 1/10,000 to <1/1,000); very rare (<1/10,000)]. Gastrointestinal disorders Common: nausea and dyspepsia In addition to those events reported during clinical trials, the following undesirable effects have been reported spontaneously during postmarketing use and in scientific literature. A frequency cannot be estimated from the available data and is therefore classified as “not known”. Immune System disorders Hypersensitivity reactions, e.g. anaphylaxis have been reported. Gastrointestinal disorders Mild gastric complaints (e.g. vomiting, gastrointestinal pain, abdominal distension and bloating) have been observed. These can normally be dealt with by taking the dose during meals or by lowering the dose. Nervous System disorders Headache Skin and subcutaneous tissue disorders Cutaneous and subcutaneous hypersensitivity reactions have been reported, in particular angioneurotic oedema, urticaria, rash, and pruritus. DRUG INTERACTIONS No in-vivo interaction studies have been performed. Based on in-vitro data no in-vivo inhibition on Cytochrome P450 enzymes is expected. Although an antagonism between Mepahist and antihistamines could be expected on a theoretical basis, no such interactions have been reported. PREGNANCY AND LACTATION Pregnancy: There are no adequate data from the use of betahistine in pregnant women. Animal studies are insufficient with respect to effects on pregnancy, embryonal/foetal development, parturition and postnatal development. The potential risk for humans is unknown. Betahistine should not be used during pregnancy unless clearly necessary. Lactation: It is not known whether betahistine is excreted in human milk. There are no animal studies on the excretion of betahistine in milk. The importance of the drug to the mother should be weighed against the benefits of nursing and the potential risks for the child. EFFECTS ON ABILITY TO DRIVE AND USE MACHINES Betahistine is regarded to have no or negligible effects on the ability to drive and use machines as no effects potentially influencing this ability were found to be related to betahistine in clinical studies. PACKAGE AND STORAGE A carton box of 2 strips (PVC-Aluminum) each of 10 tablets and inner leaflet. Store in a dry place at temperature not exceeding 30° C.