Physical and Chemical Properties of Common Elements
advertisement

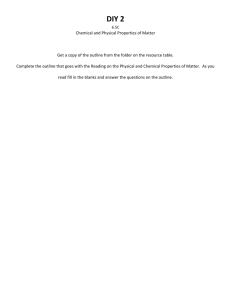
*In your Journal *LAB: Physical and Chemical Properties of Common Elements *FRAMING THE INVESTIGATION Introduction: An understanding of chemistry requires an understanding of the physical and chemical characteristics of substances. The physical properties of a substance are those properties that can be observed and measured without changing the composition of the substance. Examples of physical properties include mass, volume, length, odor, color, density, solubility, electrical conductivity, boiling point, and melting point. For example, a colorless, odorless liquid that has a boiling point of 100 oC and a melting point of 0 o C is probably water. When substances are examined without changing their identities, possibly changing their condition, we have produced a physical change. Melting ice or cutting a piece of paper, for example, does not change the identity of the water or paper, respectively. The chemical properties of a substance are those properties that can be observed when the substance is undergoing a change that results in a new composition, or identity, of the substances involved. Flammability and reactivity with other substances are examples of chemical properties. The fact that calcium carbonate decomposes on heating into calcium oxide and carbon dioxide is an example of a chemical property. Paper burning, however, would be a chemical change, as the paper becomes new and different substances: ash, water vapor and other gases. In this experiment you will examine a number of elements and describe some physical and chemical properties of each. *Problem: Will you be able to determine the difference between physical and chemical properties and physical and chemical changes? *Hypothesis: DESIGNING THE INVESTIGATION: Apparatus/Materials: magnet computer file 0.1m HCl Aluminum copper lead sulfur 5 (18 * 150 mm) test tubes LabPro Interface w/electrical probes LoggerPro software tapwater Iron Calcium turnings Mg ribbon charcoal (carbon) Procedure: 1. Read the directions to this lab. Obtain the listed materials, goggles and an apron. 2. *Build a data table that will include sample items in rows and the parameters in the columns. 3. *Examine a piece of each of the samples. Record the color, luster, and odor of each element. 4. *Attempt to scratch each in turn using a hardness kit. Compare & record the relative hardness of the elements. 5. *Hold one piece of each sample of the elements over the edge of the lab bench. Hold each firmly in place and push down on the end that protrudes. Compare & record the relative flexibility of each element. 6. *Bring a magnet into contact with each substance to see if it is magnetic. Record if it is or not. 7. Connect the electrical probes to the LabPro interface, plug in the computer cable and start up the LoggerPro computer program. You should see a screen ready to collect data. Do a “FILE”, “OPEN” command, then go into the “Physical Science with Computers” folder and find “Experiment 33”. Extend the ends of the probes and touch the voltage probe to a battery and record the value on the computer screen. 8. *Touch one end of the probe to the battery and place each element, one at a time at the other end of the battery, then touch the element with the other end of the probe, making a complete circuit. Record the value of the electrical conductivity of each of the elements in your table. 9. *If possible, place one small piece of each into the separate test tubes. To each test tube, add about 5 ml of water. Leave untouched for at least 5 minutes. Observe and record any reaction to water. If no change, record “No Reaction”. If the item is too large for the test tube, place in a small beaker and pour 5 ml of water over it and let it sit. 10. *Pour out the water but keep the items in place. Add 5ml of dilute HCl, being careful not to pour out Add the acid very carefully to the test tubes. Observe and record any reaction to HCl. If no change record “No Reaction”. Carefully clean out the test tubes by rinsing the acid out in the sink. Place the small pieces of your elements back in the trays if they are intact. Otherwise, place them in the trash, not the sink, please!! *COLLECTING AND PRESENTING DATA (Record observations in your journal followed by your data table) *ANALYZING AND INTERPRETING THE DATA: 1. Which of the properties observed are physical properties and which are chemical properties? What is your evidence? 2. Which elements reacted with the acid? Was this reactivity a physical or a chemical property? How do you know? Why must we add HCl to Ca carefully? 3. Do any of the elements conduct electricity? Which ones? Do these elements have any other similar properties? What are they? 4. What experimental errors could be present in this lab? What limitations could be possible? 5. *Look up and record in a separate table the melting and boiling points for the items studied. How do the melting points and boiling points of these substances compare to room temperature? Would it be easy to measure them in a school laboratory? Explain.