Epithelial-Mesenchymal crosstalk in the regulation of renal proximal
advertisement

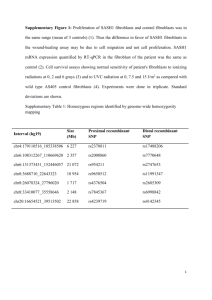
S8(T) EPITHELIAL-MESENCHYMAL CROSSTALK IN THE REGULATION OF RENAL PROXIMAL TUBULAR EPITHELIAL CELL FUNCTION Luo, D, Steadman, R, Phillips, A, Meran, S Institute of Nephrology, University Hospital of Wales, Cardiff BACKGROUND: The function of epithelial/specialised cells in many tissues is regulated by adjacent stroma. In cancer biology, during malignant transformation, epithelial cells undergo changes similar to the process of EMT in renal fibrosis. Recent reports suggest the important role of stromal fibroblasts in the initiation, as well as the progression of carcinomas. This specialised “carcinoma-associated stroma” is reported to have an abundance of myofibroblasts that express factors that support the survival, proliferation and malignant transformation of epithelial cells in a paracrine fashion. Analogous to tumorigenesis, fibrogenesis is associated with epithelial cell transdifferentiation and an abundance of myofibroblasts. However, the importance of epithelial-stromal crosstalk within the kidney and the potential role they play in progressive cortico-interstitial fibrosis has received little attention. AIM: To test the hypothesis that fibroblasts once transformed, or fibroblasts with the ability to undergo myofibroblastic transformation, differentially regulate proximal tubular cell phenotype when compared to fibroblasts that are resistant to this transformation. METHODS: We utilised an established library of phenotypically heterogenous fibroblasts which have been characterised with respect to their scarring (SF) (dermal) and non-scarring (NSF) (oral mucosal fibroblasts) potentials. We have set up a 3D co-culture system using type I collagen from rats tail to study the influence of these distinct fibroblast phenotypes on HK2 phenotype. The fibroblasts were grown within the gel, with the HK2 cells grown as monolayers on the surface of the gel. Transwell inserts were used to separate the two cell cultures for analysis. RESULTS: Fibroblasts grown within gels generated increased levels of TGF-1 compared to fibroblasts grown in 2D-culture. This increased TGF-1 resulted in increased alpha-smooth muscle actin expression, suggesting that fibroblasts adopt a myofibroblast phenotype within gels. SF phenotypes grown within gels also demonstrated more alpha-SMA expression compared to NSF phenotypes. Work was then directed towards the effect of co-culture of SF and NSF phenotypes on HK2 phenotype. Co-culture of HK2 cells with fibroblasts enhanced their E-cadherin expression as compared to HK2 culture alone. Furthermore, co-culture of HK2 with SF enhanced E-cadherin expression more than co-culture with NSF’s. Co-culture of HK2 cells with fibroblasts also enhanced HK2 proliferation, and SF phenotypes appeared to enhance this more than NSF’s. Differences in EDA-fibronectin and Hyaluronan Synthase-2 expression were also highlighted, with increased levels of both present in HK2/SF co-culture as compared with HK2/NSF co-culture and HK2 monoculture. Differences in IL-1 and EGF production were also identified, with HK2/fibroblast co-cultures (specifically HK2/SF) demonstrating increased levels. Use of the ALK5 inhibitor did not abrogate the changes in Ecadherin seen in HK2/fibroblast co-cultures. However, increased levels of miR192 and reduced ZEB-2 mRNA expression were identified in SF/HK2 cultures that may account for the differences in E-Cadherin expression seen. CONCLUSIONS: We have identified some interesting differences in HK2 phenotype, proliferation, cytokine production and matrix generation when these cells are co-cultured with fibroblasts, and when they are co-cultured with fibroblasts with scarring versus non-scarring phenotypes. We have also identified changes in miR192, HAS2 and EGF Receptor expression that may account for the changes in phenotype and proliferation seen.