Chemistry in the Wind
advertisement

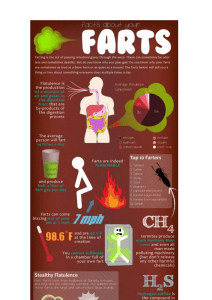
Chemistry in the Wind Questions from “Flatus Chemistry”, by Claudia Vanderborght (ChemMatters, Feb. 2003) 1. During WWII, what problem faced pilots flying at high altitude that led to extreme discomfort? 2. How did Boyle’s Law explain the problem? 3. How did the Air Force remedy or at least mitigate the problem? 4. Why would a medieval Europe embrace whistle buttocks? 5. Why did Dr. Michael Levitt get involved in odoriferous studies? 6. What seems to cause excess gas? 7. What is lactose? 8. What is the composition of tubal aroma? 9. What role do bacteria play in the production of human “dos vientos”? 10. Why would stools float? 11. Write the equation in the production of methane. 12. What’s the environmental impact of flatulating cows and termites? 13. What was NASA’s concern regarding “breaking wind”? 14. Explain the circumstances by which a patient literally exploded on the operating table. 15. Would you ever hire out your nose for “wind studies”? 16. T/F Men naturally produce more liters of flatus than women. 17. What gives flatus its characteristic odor? 18. Write the equation for the production of ”rotten egg gas.” 19. T/F 300 ppm of H2S would be quite deadly. 20. Name some foods that would make us quite the hit at King Arthur’s Buffet. 21. What is the advantage of producing H2S? Chemistry in the Wind KEY Questions from “Flatus Chemistry”, by Claudia Vanderborght (ChemMatters, Feb. 2003) 1. During WWII, what problem faced pilots flying at high altitude that led to extreme discomfort? Gas volume increase leading to intestinal cramps 2. How did Boyle’s Law explain the problem? Volume increases as pressure decreases and temperature is held constant 3. How did the Air Force remedy or at least mitigate the problem? Stop feeding pilots dried beans & peas, cabbage veggies, and beer & carbonated drinks 4. Why would a medieval Europe embrace whistle buttocks? Excessive flatus was a “sign of manly strength and a source of pride” 5. Why did Dr. Michael Levitt get involved in odoriferous studies? He had a physically and socially miserable patient with excessive gas 6. What seems to cause excess gas? Vegetable intake, certain bacteria species in digestive tract, not enough lactase enzyme 7. What is lactose? Milk sugar that is digested by the enzyme lactase 8. What is the composition of tubal aroma? 99% odorless N2, O2, H2, CO2, and CH4; 1% smelly gases 9. What role do bacteria play in the production of human “dos vientos”? Some bacteria consume H2 and produce CH4 and other gases 10. Why would stools float? Lower volume of gas – possibly expelled into stool instead of the air, making stool less dense 11. Write the equation in the production of methane. 4H2 + CO2 CH4 + 2H2O 12. What’s the environmental impact of flatulating cows and termites? Contributes to global warming because CH4 is 25 times better at trapping heat than CO2 13. What was NASA’s concern regarding “breaking wind”? That the methane could explode within the sealed aircraft 14. Explain the circumstances by which a patient literally exploded on the operating table. A surgeon touched an electrode to the patient’s colon, igniting the contained gases 15. Would you ever hire out your nose for “wind studies”? Answers will vary – but personally… you couldn’t pay me enough to do that! 16. T/F Men naturally produce more liters of flatus than women. FALSE – men and women produce the same amount and “quality” of gas 17. What gives flatus its characteristic odor? Mostly hydrogen sulfide (H2S), and some methanethiol (CH3SH) & dimethyl sulfide (CH3SCH3) 18. Write the equation for the production of ”rotten egg gas.” 4H2 + SO42- + 2H+ H2S + 4H2O 19. T/F 300 ppm of H2S would be quite deadly. TRUE – It would literally kill you 20. Name some foods that would make us quite the hit at King Arthur’s Buffet. Onions, eggs, broccoli, beer and some beans 21. What is the advantage of producing H2S? Less volume is produced (4 moles of H2 are consumed to make one mole of H2S)