LSU-ch02
advertisement

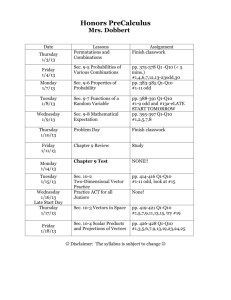
Chapter 2 Acids and Bases Multiple Choice 1. Identify the Brønsted-Lowry acids in the following reactions. (Sec. 2.3) CH3OH + I C2H5OH + CH3OH2 II III NaH V a) b) c) d) HCl Cl + IV C2H5ONa H2 + VII VI VIII I, III,VI, VII II, VI I, IV, V, VIII II, III, V, VIII 2. Identify the conjugate bases in the following reactions. (Sec. 2.3) CH3NH2 + H3CCO2H CH3NH3 + I C2H5OH + C2H5ONa NaH II + + CH3OH2 HCl V a) b) c) d) II, III, VI I, IV, V I, III, V II, IV, VI 12 H2 IV III CH3OH CH3CO2 + Cl VI Chapter 2 Acids and Bases 3. Identify the conjugate acids in the following reactions. (Sec. 2.3) CH3NH2 + CH3CO2H CH3NH3 + CH3CO2 I CH3OH + II CH3OH2 HCl Cl + IV III C2H5OH + C2H5ONa NaH H2 + VI V a) b) c) d) I, IV, VI I, III, VI II, IV, V I, III, V 4. Which are acid-base reactions according to Brønsted-Lowry theory? (Sec. 2.3) O O I. CH3 II. III. IV. a) b) c) d) C C + H3CNH3 AlCl3 + Cl AlCl4 CH3NH2 + HCl CH3NH3Cl CH3Li + H2O O CH3 CH4 I, III I, II, II, IV I, II, III I, III, IV 13 OH + CH3NH2 + LiOH Chapter 2 Acids and Bases 5. Which are acid-base reactions according to the Brønsted-Lowry theory? (Sec. 2.3) I. (CH3)3COH + H2SO4 (CH3)3C + H2O II. Br2 + FeBr3 FeBr4 + Br III. CH3NH2 + BF3 CH3NH2BF3 IV. CH3NH2 + HCl CH3NH3Cl a) b) c) d) + I I, III, IV II, III I, IV 6. Which are acid-base reactions according to Lewis theory but not according to the Brønsted-Lowry theory? (Sec. 2.7) O O I. H3C C CH3 + H3C CH3 C CH3 O O II. III. IV. H3C C O + H2O + BF3 + BF3 OH NH3 H3C CH3 H3C C O NH3BF3 H3C O H3C a) b) c) d) CH3 I, II III, IV I, III, IV I, II, III, IV 14 BF3 + H3O HSO4 Chapter 2 Acids and Bases 7. Which acid has the highest pKa? (Sec. 2.4) a) b) c) d) CH3COOH H2O NH4+ HCl 8. Arrange the following in order of increasing basicity (weakest to strongest). (Sec. 2.4) I. OH- II. Cla) b) c) d) III. H2O IV. NH3 II, III, IV, I III, I, IV, II IV, I, II, III III, IV, I, II 9. Arrange the following ions in the order of increasing acidity (weakest to strongest). (Sec. 2.4) II. H3O+ I. H2O a) b) c) d) III. NH4+ II, III, I I, II, III III, II, I I, III, II 10. Which ion is the strongest base? (Sec. 2.4) O a) CH3CH2O b) CH3 C O c) Cl 15 d) CH3CH2 Chapter 2 Acids and Bases 11. Which equilibria lie considerably toward the right? (Sec. 2.5) O O C I. + OH H3C NH3 C H3C O II. C H3C NH4 + O O + O C H2O H3C OH + OH III. NH4 + OH NH3 + H2O IV. HCN + OH CN + H2O a) b) c) d) II, III I, III, IV III, IV I, II, III 12. Which equilibria lie considerably toward the left? (Sec. 2.5) O O C I. OH H3C + CN C H3C O II. C H3C O + + C NH4 H3C OH a) b) c) d) C H3C OH NH3 O IV. + HCN O NH4 III. O + NH3 + H2O + CH3OH O OH + C CH3 H3C II I, IV III, IV I 16 O Chapter 2 Acids and Bases 13. List the bonds in order of increasing acidity (least to most). (Sec. 2.6) C H O H I II a) b) c) d) F H N H III IV II, III, IV, I III, I, II, IV I, IV, II, III IV, III, II, I 14. List the bonds in order of decreasing acidity (most to least). (Sec. 2.6) F Br H I a) b) c) d) H II Cl H I H III IV I, III, II, IV IV, II, III, I II, III, IV, I I, II, III, IV 15. Which substances are Lewis bases? (Sec. 2.7) H2O AlCl3 I II a) b) c) d) CH H3C F CH3 III IV I, II I, III III, IV I, IV 16. What is the stronger acid in the following reaction if the equilibrium constant is approximately 10 8. (Sec. 2.5) + NH2 HC CH a) b) HC C + c) NH3 d) 17. What is the stronger acid in the following reaction if the equilibrium constant is much less than 0.01? (Sec. 2.5) HNO3 a) + H2SO4 b) H2NO3 + c) HSO4 d) 17 Chapter 2 Acids and Bases 18. Which is the stronger base if the equilibrium lies considerably to the right? (Sec. 2.5) O O C H3C OH CN + C H3C a) b) + O c) HCN d) 19. What is the role of diethyl ether in the following reaction? (Sec. 2.7) CH3CH2OCH2CH3 a) b) c) d) + BF3 (CH3CH2)2O BF3 Lewis acid Lewis base Brønsted acid Brønsted base 20. What is the role of water in the following reaction? (Sec. 2.3) NH3 a) b) c) d) + H3O H2O + NH4 acid base conjugate acid conjugate base 21. What is the role of methanol in the following reaction? (Sec. 2.7) CH3 CH3 H3C C + CH3OH H3C C O CH3 CH3 a) b) c) d) CH3 H Lewis acid Lewis base Brønsted acid Brønsted base 22. Which statements about acid-base equilibria are true? (Sec. 2.4) I. The pKa is the negative log10 of the acid equilibrium constant. II. A stronger acid has a pKa with a smaller value than a weaker acid. III. A stronger base has a conjugate acid which has a pKa with a smaller value than a weaker base. IV. The Ka = K [HA]. a) b) c) d) I, III I, II I, II, III II, III, IV 18 Chapter 2 Acids and Bases 23. Which is the order of decreasing acid strength of the following compounds (greatest first)? (Sec. 2.6) O Cl C H2 C O Br OH C H2 C O F OH C H2 II I a) b) c) d) C O H OH C H2 III C OH IV II, I, III, IV III, IV, I, II III, I, II, IV IV, II, I, III 24. Which is the order of increasing acid strength of the following compounds (least first)? (Sec. 2.6) O O F OH O OH OH F I a) b) c) d) O F OH F II III I, III, II, IV IV, III, II, I II, I, III, IV IV, III, I, II 19 IV Chapter 2 Acids and Bases 25. Which is the proper reaction mechanism for the reaction of borontrifluoride and diethyl ether? (Sec. 2.7) a) :BF3 CH3CH2 b) BF3 CH3CH2 O + BF 3 CH2CH3 O - CH2CH3 CH3CH2 .. O .. CH CH 2 3 - BF 3 O + CH2CH3 CH3CH2 c) :BF3 d) CH3CH2 BF3 CH3CH2 O CH2CH3 CH3CH2 .. O .. CH CH 2 3 BF3 O + CH2CH3 + BF 3 CH3CH2 O - CH2CH3 Fill in the Blank 1. The weaker the acid, the ________________ the conjugate base. (Sec. 2.5) 2. The higher concentration (reactants or products) at equilibrium will lie on the side of the ___________ acid. (Sec. 2.5) 3. Complete the following reaction. (Sec. 2.3) O O CH3COH + H2O CH3CO- + 4. Complete the following reaction. (Sec. 2.3) O NH4Cl + CH3CONa NH3 + 5. Complete the following reaction. (Sec. 2.3) + H2O NH3 + H3O+ + Cl- 20 Chapter 2 Acids and Bases 6. Complete the following reaction. (Sec. 2.3) O O CH3CONa + NaHCO3 CH3COH + 7. Complete the following reaction scheme with the appropriate equilibrium arrow (indicating the higher concentrations at equilibrium). (Sec. 2.5) O O CH3CONa + HCN CH3COH + NaCN 8. Complete the following reaction scheme with the appropriate equilibrium arrow (indicating the higher concentrations at equilibrium). (Sec. 2.5) NaH2PO4 + NaHCO3 Na2PO4 + H2CO3 9. Identify the acid, base, conjugate acid, conjugate base in the following reaction. (Sec. 2.3) NaCN + NaHSO4 HCN + Na2SO4 10. Identify the acid, base, conjugate acid, conjugate base in the following reaction. (Sec. 2.3) NH4Cl + NaHCO3 NH3 + H2CO3 + NaCl True-False 1. Brønsted-Lowry acids accept protons when reacting. (Sec. 2.3) 2. Lewis bases donate electrons when reacting. (Sec. 2.7) 3. The stronger acid has the larger (more positive) pK a. (Sec. 2.4) 4. Strong acids have weak conjugate bases. (Sec. 2.4) 5. The equilibrium will lie to the right in the following reaction. (Sec. 2.5) NH4Cl + H2O NH3 + H3O+ + 6. The equilibrium will lie to the right in the following reaction. (Sec. 2.5) HCN + NaH2PO4 H3PO4 + NaCN 21 Cl- Chapter 2 Acids and Bases 7. Water acts as a base in the following reaction. (Sec. 2.3) NH3 + H2O NH4OH 8. Ammonia acts as a Brønsted-Lowry base in the following reaction. (Sec. 2.3) NH4Cl + NaHCO3 NH3 + H2CO3 + NaCl 9. The strongest acid in the following list is sodium bicarbonate. (Sec. 2.4) NH4Cl NaHCO3 H2O CH3CH2OH 10. The weakest acid in the following list is acetic acid. (Sec. 2.6) O CH3COH O ClCH2COH O O FCH2COH 22 FCH2CH2COH Chapter 2 Acids and Bases Answers Multiple Choice 1. d 2. a 3. b 4. d 5. d 6. c 7. b 8. a 9. d 10. d 11. b 12. a 13. c 14. b 15. d 16. a 17. c 18. b 19. b 20. d 21. b 22. b 23. c 24. b 25. b Fill in the Blank 1. stronger 2. weaker 3. H3O+ 4. CH3COOH + NaCl 5. NH4Cl 6. Na2CO3 7. equilibrium arrow pointed to the left 8. equilibrium arrow pointed to the left 9. base, acid, c.acid, c. base 10. acid, base, c. base, c. acid True-False 1. F 2. T 3. F 4. T 5. F 6. F 7. F 8. T 9. F 10. T 23